Abstract
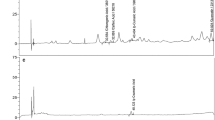
Marine algae are an auspicious source of innovative bioactive compounds containing possible therapeutic agents against mammalian cancers. However, the mechanism by which bioactive algal compounds exhibit anticancer activity against oral squamous cell carcinoma (OSCC) is scant. The main objective of the current study was to explore the properties of the Enteromorpha compressa solvent extracts that induced autophagy and apoptosis with reference to their potent phytochemical and antioxidant properties. The presence of bioactive compounds were confirmed by UV and FT-IR spectroscopy. The free radical scavenging activity were analyzed by evaluating H2O2, DPPH, superoxide and hydroxyl activity. The anticancer activities of the extracts were investigated by employing clonogenic and scratch assay. The apoptosis potential was evaluated by DAPI and MMP by Rh123 fluorescence assay. Moreover, the CAT, SOD, GPX, APX, and GR activities were measured. The autophagy potential was evaluated by LC3 puncta formation, acridine orange in addition to LysoTracker staining. The present investigation revealed that the methanolic extract of E. compressa elicited robust free radical scavenging activity that discerns its antiproliferative potency. Moreover, the methanolic algal extract boosted intrinsic apoptosis against OSCC by downregulating protective antioxidant enzymes. Furthermore, it also revealed induction of autophagy to promote cell death in oral cancer cells. The presence of novel bioactive compounds in E. compressa has uncovered possible therapeutic value against OSCC by modulating antioxidant defense system, apoptosis and autophagy that could be used to explore very competent algal candidates for the development of potential alternative anticancer drugs.





Similar content being viewed by others
References
Kurt O, Özdal-Kurt F, Tuğlu M, Akçora C (2014) The cytotoxic, neurotoxic, apoptotic and antiproliferative activities of extracts of some marine algae on the MCF-7 cell line. Biotechnic Histochem 89(8):568–576
Namvar F, Baharara J, Mahdi AA (2014) Antioxidant and anticancer activities of selected persian gulf algae. Indian J Clin Biochem 29(1):13–20. https://doi.org/10.1007/s12291-013-0313-4
Pradhan B, Patra S, Nayak R, Behera C, Dash SR, Nayak S, Sahu BB, Bhutia SK, Jena M (2020) Multifunctional role of fucoidan, sulfated polysaccharides in human health and disease: a journey under the sea in pursuit of potent therapeutic agents. Int J Biol Macromol. https://doi.org/10.1016/j.ijbiomac.2020.09.019
Mohanty S, Pradhan B, Patra S, Behera C, Nayak R, Jena M (2020) Screening for nutritive bioactive compounds in some algal strains isolated from coastal Odisha. J Adv Plant Sci 10(2):1–8
Murugan K, Iyer VV (2013) Differential growth inhibition of cancer cell lines and antioxidant activity of extracts of red, brown, and green marine algae. In Vitro Cell Dev Biol Anim 49(5):324–334. https://doi.org/10.1007/s11626-013-9603-7
Paul S, Kundu R (2013) Antiproliferative activity of methanolic extracts from two green algae, Enteromorpha intestinalis and Rizoclonium riparium on HeLa cells. DARU 21(1):72. https://doi.org/10.1186/2008-2231-21-72
Xu N, Fan X, Yan X, Tseng CK (2004) Screening marine algae from China for their antitumor activities. J Appl Phycol 16(6):451–456. https://doi.org/10.1007/s10811-004-5508-x
Mridha A, Paul S (2017) Algae as potential repository of anti cancerous natural compounds. Int J Phytomed. https://doi.org/10.5138/09750185.2023
Rath J, Adhikary S (2008) Biodiversity assessment of algae in Chilika Lake, east coast of India. In: Mohanty PK (ed) Monitoring and modelling lakes and coastal environments. Springer, Dordrecht, pp 22–33
Mirza MB, Elkady AI, Al-Attar AM, Syed FQ, Mohammed FA, Hakeem KR (2018) Induction of apoptosis and cell cycle arrest by ethyl acetate fraction of Phoenix dactylifera L. (Ajwa dates) in prostate cancer cells. J Ethnopharmacol 218:35–44. https://doi.org/10.1016/j.jep.2018.02.030
Patra S, Mishra SR, Behera BP, Mahapatra KK, Panigrahi DP, Bhol CS, Praharaj PP, Sethi G, Patra SK, Bhutia SK (2020) Autophagy-modulating phytochemicals in cancer therapeutics: current evidences and future perspectives. Semin Cancer Biol. https://doi.org/10.1016/j.semcancer.2020.05.008
Patra S, Praharaj PP, Panigrahi DP, Panda B, Bhol CS, Mahapatra KK, Mishra SR, Behera BP, Jena M, Sethi G, Patil S, Patra SK, Bhutia SK (2020) Bioactive compounds from marine invertebrates as potent anticancer drugs: the possible pharmacophores modulating cell death pathways. Mol Biol Rep. https://doi.org/10.1007/s11033-020-05709-8
Sinha N, Mukhopadhyay S, Das DN, Panda PK, Bhutia SK (2013) Relevance of cancer initiating/stem cells in carcinogenesis and therapy resistance in oral cancer. Oral Oncol 49(9):854–862. https://doi.org/10.1016/j.oraloncology.2013.06.010
Sinha N, Panda PK, Naik PP, Das DN, Mukhopadhyay S, Maiti TK, Shanmugam MK, Chinnathambi A, Zayed ME, Alharbi SA, Sethi G, Agarwal R, Bhutia SK (2017) Abrus agglutinin promotes irreparable DNA damage by triggering ROS generation followed by ATM-p73 mediated apoptosis in oral squamous cell carcinoma. Mol Carcinog 56(11):2400–2413. https://doi.org/10.1002/mc.22679
Panigrahi GK, Yadav A, Mandal P, Tripathi A, Das M (2016) Immunomodulatory potential of rhein, an anthraquinone moiety of Cassia occidentalis seeds. Toxicol Lett 245:15–23
Riboli E, Norat T (2003) Epidemiologic evidence of the protective effect of fruit and vegetables on cancer risk. Am J Clin Nutr 78(3 Suppl):559s–569s. https://doi.org/10.1093/ajcn/78.3.559S
Salehi B, Zucca P (2018) Phytotherapeutics in cancer invasion and metastasis. Phytother Res 32(8):1425–1449. https://doi.org/10.1002/ptr.6087
Danaei G, Vander Hoorn S, Lopez AD, Murray CJ, Ezzati M, group CRAc (2005) Causes of cancer in the world: comparative risk assessment of nine behavioural and environmental risk factors. The Lancet 366(9499):1784–1793
Patra S, Panda PK, Naik PP, Panigrahi DP, Praharaj PP, Bhol CS, Mahapatra KK, Padhi P, Jena M, Patil S, Patra SK, Bhutia SK (2020) Terminalia bellirica extract induces anticancer activity through modulation of apoptosis and autophagy in oral squamous cell carcinoma. Food Chem Toxicol 136:111073. https://doi.org/10.1016/j.fct.2019.111073
Usman H, Abdulrahman F, Usman A (2009) Qualitative phytochemical screening and in vitro antimicrobial effects of methanol stem bark extract of Ficus thonningii (Moraceae). Afr J Tradit Complement Altern Med 6(3):289–295
Kabir MF, Mohd Ali J (2017) Melicope ptelefolia leaf extracts exhibit antioxidant activity and exert anti-proliferative effect with apoptosis induction on four different cancer cell lines. BMC Complement Altern Med 17(1):252. https://doi.org/10.1186/s12906-017-1761-9
Quettier-Deleu C, Gressier B, Vasseur J, Dine T, Brunet C, Luyckx M, Cazin M, Cazin JC, Bailleul F, Trotin F (2000) Phenolic compounds and antioxidant activities of buckwheat (Fagopyrum esculentum Moench) hulls and flour. J Ethnopharmacol 72(1–2):35–42. https://doi.org/10.1016/s0378-8741(00)00196-3
Pradhan B, Baral S, Patra S, Behera C, Nayak R, MubarakAli D, Jena M (2020) Delineation of gamma irradiation (60Co) induced oxidative stress by decrypting antioxidants and biochemical responses of microalga Chlorella sp. . Biocatal Agric Biotechnol 25:101595. https://doi.org/10.1016/j.bcab.2020.101595
Prieto P, Pineda M, Aguilar M (1999) Spectrophotometric quantitation of antioxidant capacity through the formation of a phosphomolybdenum complex: specific application to the determination of vitamin E. Anal Biochem 269(2):337–341. https://doi.org/10.1006/abio.1999.4019
Nabavi S, Ebrahimzadeh M, Nabavi S, Hamidinia A, Bekhradnia A (2008) Determination of antioxidant activity, phenol and flavonoids content of Parrotia persica Mey. Pharmacologyonline 2(9):560–567
Pradhan B, Patra S, Maharana S, Behera C, Dash SR, Jena M (2020) Demarcating antioxidant response against aluminum induced oxidative stress in Westiellopsis prolifica Janet. Int J Phytobioremed 1941:1–14. https://doi.org/10.1080/15226514.2020.1807906
Liu F, Ooi V, Chang S (1997) Free radical scavenging activities of mushroom polysaccharide extracts. Life Sci 60(10):763–771
Weydert CJ, Cullen JJ (2010) Measurement of superoxide dismutase, catalase and glutathione peroxidase in cultured cells and tissue. Nat Protoc 5(1):51–66. https://doi.org/10.1038/nprot.2009.197
Pradhan B, Patra S, Dash SR, Maharana S, Behera C, Jena M (2020) Antioxidant responses against aluminum metal stress in Geitlerinema amphibium. SN Appl Sci 2(5):800. https://doi.org/10.1007/s42452-020-2599-1
Patra S, Bhol CS, Panigrahi DP, Praharaj PP, Pradhan B, Jena M, Bhutia SK (2020) Gamma irradiation promotes chemo-sensitization potential of gallic acid through attenuation of autophagic flux to trigger apoptosis in an NRF2 inactivation signalling pathway. Free Radical Biol Med 160:111–124. https://doi.org/10.1016/j.freeradbiomed.2020.06.022
Kosanić M, Ranković B, Stanojković T (2015) Biological activities of two macroalgae from Adriatic coast of Montenegro. Saudi J Biol Sci 22(4):390–397
Narasimhan MK, Pavithra SK, Krishnan V, Chandrasekaran M (2013) In vitro analysis of antioxidant, antimicrobial and antiproliferative activity of Enteromorpha antenna, Enteromorpha linza and Gracilaria corticata extracts. Jundishapur J Nat Pharm Prod 8(4):151
Hiqashi-Okaj K, Otani S, Okai Y (1999) Potent suppressive effect of a Japanese edible seaweed, Enteromorpha prolifera (Sujiao-nori) on initiation and promotion phases of chemically induced mouse skin tumorigenesis. Cancer Lett 140(1–2):21–25
Panigrahi DP, Bhol CS, Niveta R, Nagini S, Patil S, Maiti TK, Bhutia SK (2019) Abrus agglutinin inhibits oral carcinogenesis through inactivation of NRF2 signaling pathway. Int J Biol Macromol. https://doi.org/10.1016/j.ijbiomac.2019.11.079
Panda PK, Patra S, Naik PP, Praharaj PP, Mukhopadhyay S, Meher BR, Gupta PK, Verma RS, Maiti TK, Bhutia SK (2020) Deacetylation of LAMP1 drives lipophagy-dependent generation of free fatty acids by Abrus agglutinin to promote senescence in prostate cancer. J Cell Physiol 235(3):2776–2791. https://doi.org/10.1002/jcp.29182
Lobo V, Patil A, Phatak A, Chandra N (2010) Free radicals, antioxidants and functional foods: impact on human health. Pharmacognosy Rev 4(8):118–126. https://doi.org/10.4103/0973-7847.70902
Perez-Perez ME, Couso I, Crespo JL (2017) The TOR signaling network in the model unicellular green alga Chlamydomonas reinhardtii. Biomolecules 7(3):54. https://doi.org/10.3390/biom7030054
Perez-Perez ME, Couso I (2017) Monitoring autophagy in the model green microalga Chlamydomonas reinhardtii. Cells 6(4):36. https://doi.org/10.3390/cells6040036
Jiang Q, Zhao L, Dai J, Wu Q (2012) Analysis of autophagy genes in microalgae: chlorella as a potential model to study mechanism of autophagy. PLoS ONE 7(7):e41826. https://doi.org/10.1371/journal.pone.0041826
Acknowledgements
The authors are thankful to Berhampur University and the National Institute of Technology Rourkela for providing the necessary facilities. We acknowledge S&T Department, Govt. of Odisha and MoEF & CC, Govt. of India for funding.
Funding
The authors kindly thank S&T Department, Govt. of Odisha and MoEF & CC, Govt. of India for funding to carry out this research work.
Author information
Authors and Affiliations
Contributions
BP, SP, CB and RN have prepared the manuscript and table. BP, SP and RN have contributed towards the preparation of figure. MJ, SKB and SGP have done the proof reading of the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors have no conflicts of interest to disclose.
Research involving human participants and/or animals
No Human participation and/or Animal have been used in this study.
Informed consent
The corresponding author on behalf of all coauthors agree to accept the informed consent of compliance with ethical standard.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Rights and permissions
About this article
Cite this article
Pradhan, B., Patra, S., Behera, C. et al. Enteromorpha compressa extract induces anticancer activity through apoptosis and autophagy in oral cancer. Mol Biol Rep 47, 9567–9578 (2020). https://doi.org/10.1007/s11033-020-06010-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11033-020-06010-4