Abstract
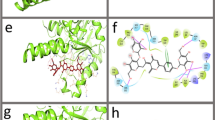
Tuberculosis (TB), an infectious disease caused by the Mycobacterium tuberculosis (Mtb), has been responsible for the deaths of millions of individuals around the globe. A vital protein in viral pathogenesis known as resuscitation promoting factor (RpfB) has been identified as a potential therapeutic target of anti-tuberculosis drugs. This study offered an in silico process to examine possible RpfB inhibitors employing a computational drug design pipeline. In this study, a total of 1228 phytomolecules were virtually tested against the RpfB of Mtb. These phytomolecules were sourced from the NP-lib database of the MTi-OpenScreen server, and five top hits (ZINC000044404209, ZINC000059779788, ZINC000001562130, ZINC000014766825, and ZINC000043552589) were prioritized for compute intensive docking with dock score ≤ − 8.5 kcal/mole. Later, molecular dynamics (MD) simulation and principal component analysis (PCA) were used to validate these top five hits. In the list of these top five hits, the ligands ZINC000044404209, ZINC000059779788, and ZINC000043552589 showed hydrogen bond formation with the functional residue Glu292 of the RpfB protein suggesting biological significance of the binding. The RMSD study showed stable protein–ligand complexes and higher conformational consistency for the ligands ZINC000014766825, and ZINC000043552589 with RMSD 3–4 Å during 100 ns MD simulation. The overall analysis performed in the study suggested promising binding of these compounds with the RpfB protein of the Mtb at its functional site, further experimental investigation is needed to validate the computational finding.







Similar content being viewed by others
Data availability
All data generated or analysed during this study are included in this published article.
References
World Health Organization (2021) Global tuberculosis report 2021. World Health Organization, Geneva
(2020) Indonesia Tuberculosis Roadmap Overview, Fiscal Year 2021. https://www.usaid.gov/global-health/health-areas/tuberculosis/resources/news-and-updates/global-accelerator-end-tb/tb-roadmaps/indonesia. Accessed 28 Nov 2022
(2018) Tuberculosis (TB)—Treatment. In: nhs.uk. https://www.nhs.uk/conditions/tuberculosis-tb/treatment/. Accessed 30 Nov 2022
Bajrai LH, Khateb AM, Alawi MM et al (2022) Glycosylated flavonoid compounds as potent CYP121 inhibitors of Mycobacterium tuberculosis. Biomolecules 12:1356
Singh K, Pandey N, Ahmad F et al (2022) Identification of novel inhibitor of Enoyl-acyl Carrier protein reductase (InhA) enzyme in Mycobacterium tuberculosis from plant-derived metabolites: an in silico study. Antibiotics 11:1038
Kell DB, Young M (2000) Bacterial dormancy and culturability: the role of autocrine growth factors. Curr Opin Microbiol 3:238–243. https://doi.org/10.1016/s1369-5274(00)00082-5
Squeglia F, Marchetti R, Ruggiero A et al (2011) Chemical basis of peptidoglycan discrimination by PrkC, a key kinase involved in bacterial resuscitation from dormancy. J Am Chem Soc 133:20676–20679. https://doi.org/10.1021/ja208080r
Ruggiero A, Marasco D, Squeglia F et al (2010) Structure and functional regulation of RipA, a mycobacterial enzyme essential for daughter cell separation. Structure 18:1184–1190. https://doi.org/10.1016/j.str.2010.06.007
Ruggiero A, Squeglia F, Marasco D et al (2011) X-ray structural studies of the entire extracellular region of the serine/threonine kinase PrkC from Staphylococcus aureus. Biochem J 435:33–41. https://doi.org/10.1042/BJ20101643
Kaufmann SHE, McMichael AJ (2005) Annulling a dangerous liaison: vaccination strategies against AIDS and tuberculosis. Nat Med 11:S33-44. https://doi.org/10.1038/nm1221
Zhang X, Guo J (2012) Advances in the treatment of pulmonary tuberculosis. J Thorac Dis. https://doi.org/10.3978/j.issn.2072-1439.2012.10.10
Dye C, Watt CJ, Bleed D (2002) Low access to a highly effective therapy: a challenge for international tuberculosis control. Bull World Health Organ 80:437–444
Latent Tuberculosis Infection: updated and consolidated guidelines for programmatic management.
Corbett EL, Watt CJ, Walker N et al (2003) The growing burden of tuberculosis: global trends and interactions with the HIV epidemic. Arch Intern Med 163:1009–1021. https://doi.org/10.1001/archinte.163.9.1009
Velayati AA, Masjedi MR, Farnia P et al (2009) Emergence of new forms of totally drug-resistant tuberculosis bacilli: super extensively drug-resistant tuberculosis or totally drug-resistant strains in iran. Chest 136:420–425. https://doi.org/10.1378/chest.08-2427
Rowland K (2012) Totally drug-resistant TB emerges in India. Nature. https://doi.org/10.1038/nature.2012.9797
Migliori GB, De Iaco G, Besozzi G et al (2007) First tuberculosis cases in Italy resistant to all tested drugs. Euro Surveill 12(E070517):1. https://doi.org/10.2807/esw.12.20.03194-en
Mylliemngap BJ, Borthakur A, Velmurugan D, Bhattacharjee A (2012) Insilico analysis and molecular docking of resuscitation promoting factor B (RpfB) protein of Mycobacterium tuberculosis. Bioinformation 8:646–651. https://doi.org/10.6026/97320630008646
Mukamolova GV, Turapov OA, Young DI et al (2002) A family of autocrine growth factors in Mycobacterium tuberculosis. Mol Microbiol 46:623–635. https://doi.org/10.1046/j.1365-2958.2002.03184.x
Muñoz-Elías EJ, Timm J, Botha T et al (2005) Replication dynamics of Mycobacterium tuberculosis in chronically infected mice. Infect Immun 73:546–551. https://doi.org/10.1128/IAI.73.1.546-551.2005
Tufariello JM, Mi K, Xu J et al (2006) Deletion of the Mycobacterium tuberculosis resuscitation-promoting factor Rv1009 gene results in delayed reactivation from Chronic Tuberculosis. Infect Immun 74:2985–2995. https://doi.org/10.1128/IAI.74.5.2985-2995.2006
Cole ST, Brosch R, Parkhill J et al (1998) Deciphering the biology of Mycobacterium tuberculosis from the complete genome sequence. Nature 393:537–544. https://doi.org/10.1038/31159
Hett EC, Chao MC, Deng LL, Rubin EJ (2008) A mycobacterial enzyme essential for cell division synergizes with resuscitation-promoting factor. PLoS Pathog 4:e1000001. https://doi.org/10.1371/journal.ppat.1000001
Fan A, Jian W, Shi C et al (2010) Production and characterization of monoclonal antibody against Mycobacterium tuberculosis RpfB domain. Hybridoma 29:327–332. https://doi.org/10.1089/hyb.2010.0007
Dwivedi VD, Arya A, Sharma T et al (2020) Computational investigation of phytomolecules as resuscitation-promoting factor B (RpfB) inhibitors for clinical suppression of Mycobacterium tuberculosis dormancy reactivation. Infect Genet Evol 83:104356
Dwivedi VD, Arya A, Sharma T et al (2020) Computational investigation of phytomolecules as resuscitation-promoting factor B (RpfB) inhibitors for clinical suppression of Mycobacterium tuberculosis dormancy reactivation. Infect, Genet Evol 83:104356. https://doi.org/10.1016/j.meegid.2020.104356
Demina GR, Makarov VA, Nikitushkin VD et al (2009) Finding of the Low molecular weight inhibitors of resuscitation promoting factor enzymatic and resuscitation activity. PLoS ONE 4:e8174. https://doi.org/10.1371/journal.pone.0008174
Gupta VK, Kumar MM, Singh D et al (2018) Drug targets in dormant Mycobacterium tuberculosis: can the conquest against tuberculosis become a reality? Infect Dis 50:81–94. https://doi.org/10.1080/23744235.2017.1377346
Gupta M, Sharma R, Kumar A (2018) Docking techniques in pharmacology: how much promising? Comput Biol Chem 76:210–217. https://doi.org/10.1016/j.compbiolchem.2018.06.005
Kumar S, Paul P, Yadav P et al (2022) A multi-targeted approach to identify potential flavonoids against three targets in the SARS-CoV-2 life cycle. Comput Biol Med 142:105231. https://doi.org/10.1016/j.compbiomed.2022.105231
Berman HM, Westbrook J, Feng Z et al (2000) The protein data bank. Nucleic Acids Res 28:235–242. https://doi.org/10.1093/nar/28.1.235
Ruggiero A, Marchant J, Squeglia F et al (2013) Molecular determinants of inactivation of the resuscitation promoting factor B from Mycobacterium tuberculosis. J Biomol Struct Dyn 31:195–205. https://doi.org/10.1080/07391102.2012.698243
Pettersen EF, Goddard TD, Huang CC et al (2004) UCSF Chimera—a visualization system for exploratory research and analysis. J Comput Chem 25:1605–1612
Labbé CM, Rey J, Lagorce D et al (2015) MTiOpenScreen: a web server for structure-based virtual screening. Nucleic Acids Res 43:W448-454. https://doi.org/10.1093/nar/gkv306
Lin K, Li D, Ye Y et al (2022) Friction behavior of 2D hydrogenated diamond-like films and bilayer graphene. Diam Relat Mater 127:109179. https://doi.org/10.1016/j.diamond.2022.109179
Eberhardt J, Santos-Martins D, Tillack AF, Forli S (2021) AutoDock Vina 1.2.0: New docking methods, expanded force field, and Python bindings. J Chem Inf Model 61:3891–3898. https://doi.org/10.1021/acs.jcim.1c00203
Trott O, Olson AJ (2010) AutoDock Vina: improving the speed and accuracy of docking with a new scoring function, efficient optimization and multithreading. J Comput Chem 31:455–461. https://doi.org/10.1002/jcc.21334
Schrödinger Release 2022–3: Maestro, Schrödinger, LLC, New York, 2021.
Bowers KJ, Chow DE, Xu H, et al (2006) Scalable Algorithms for Molecular Dynamics Simulations on Commodity Clusters. In: SC ’06: Proceedings of the 2006 ACM/IEEE Conference on Supercomputing. pp 43–43
Schrödinger Release 2022–3: Desmond Molecular Dynamics System, D. E. Shaw Research, New York, NY, 2021. Maestro-Desmond Interoperability Tools, Schrödinger, New York, 2021.
Grant BJ, Rodrigues APC, ElSawy KM et al (2006) Bio3d: an R package for the comparative analysis of protein structures. Bioinformatics 22:2695–2696. https://doi.org/10.1093/bioinformatics/btl461
Jolliffe I, Lovric M (2011) International encyclopedia of statistical science. Principal Component Analysis. Springer, Berlin, pp 1094–1096
Bao L, Yu J, Morigentu M, et al (2022) A computer simulation-based study on the intervention of active ingredients of Mongolian medicine into the S-protein of SARS-CoV-2-associated variant Omicron. Chemistry
Bhowmik D, Jagadeesan R, Rai P et al (2021) Evaluation of potential drugs against leishmaniasis targeting catalytic subunit of Leishmania donovani nuclear DNA primase using ligand based virtual screening, docking and molecular dynamics approaches. J Biomol Struct Dyn 39:1838–1852. https://doi.org/10.1080/07391102.2020.1739557
Squeglia F, Romano M, Ruggiero A et al (2013) Carbohydrate recognition by RpfB from Mycobacterium tuberculosis unveiled by crystallographic and molecular dynamics analyses. Biophys J 104:2530–2539. https://doi.org/10.1016/j.bpj.2013.04.040
Singh A, Kumar S, Gupta VK et al (2022) (2022) Computational assessment of Withania somnifera phytomolecules as putative inhibitors of Mycobacterium tuberculosis CTP synthase PyrG. J Biomol Struct Dyn. https://doi.org/10.1080/073911022074142
Funding
This study is supported via funding from Prince Sattam bin Abdulaziz University project number (PSAU/2023/R/1444).
Author information
Authors and Affiliations
Contributions
Conceptualization, A.A.R. and R.K.M; Data curation, M.G., M.A. and B.R.A.S.; Formal analysis, A.A.R., M.G., M.A., B.R.A.S., A.A., M.A.N., M.A., Y.A., M.Ali, M.M.M., A.S.S.A., M.A., S.M.A., R.A.F.; Methodology, , A.A.R., M.G., M.A., B.R.A.S., A.A., M.A.N., M.A., Y.A., M.Ali, M.M.M., A.S.S.A., M.A., S.M.A., R.A.F.; Supervision, A.A.R. and R.K.M.; Validation, , A.A.R., M.G., M.A., B.R.A.S., A.A., M.A.N., M.A., Y.A., M.Ali, M.M.M., A.S.S.A., M.A., S.M.A., R.A.F.; Methodology, , A.A.R., M.G., M.A., B.R.A.S., A.A., M.A.N., M.A., Y.A., M. Ali, M.M.M., A.S.S.A., M.A., S.M.A., R.A.F., R.K.M.; Writing—original draft, A.A.R. and R.K.M.; All authors reviewed the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Rabaan, A.A., Garout, M., Aljeldah, M. et al. Anti-tubercular activity evaluation of natural compounds by targeting Mycobacterium tuberculosis resuscitation promoting factor B inhibition: An in silico study. Mol Divers (2023). https://doi.org/10.1007/s11030-023-10632-8
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11030-023-10632-8