Abstract
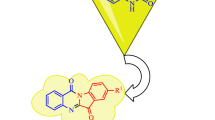
An ultrasound-assisted green protocol for one-pot synthesis of a new series of pharmaceutically relevant pyrazolo quinoline derivatives (4a–t) were synthesized, characterized, and evaluated using DFT and biological activities. Pyrazolo quinoline derivatives (4a–t) were synthesized via a three-component tandem reaction of 1,3-dicarbonyl compound (1a–b), substituted aromatic aldehyde (2a–o), and 5-amino indazole (3a) in the presence of 1-butyl-3-methylimidazolium tetrafluoroborate [BMIM]BF4 ionic liquid in ethanol at ambient conditions. The main purpose of the present work is selective functionalization of pyrazolo quinoline (4a–t) core excluding another potential parallel reaction under environmentally benign reaction conditions. The present protocol shows features such as amphiphilic behavior of ionic liquid during reaction transformation, and reusability of the [BMIM]BF4 ionic liquid under mild reaction condition. All newly derived compounds were evaluated for their in vitro anti-inflammatory and antioxidant activity. Among them, compound 4c showed encouraging antioxidant activity compared with standard antioxidant ascorbic acid, and compounds 4n and 4r displayed very good anti-inflammatory activity compared with a standard drug. In this study, a theoretical computational density functional study was also executed to perform the geometry optimizations, frontier molecular orbital approach, and molecular electrostatic potential (MESP). The DFT study was carried out with the basis set DFT/B3LYP/6-31+G (d, p) level of theory. The quantum chemical descriptors (QCDS) and MESP diagrams were plotted to examine the biological reactivities of representative pyrazolo quinolines (4a–t).
Graphical abstract







Similar content being viewed by others
References
Dömling A, Wang W, Wang K (2012) Chemistry and biology of multicomponent reactions. Chem Rev 112(6):3083–3135. https://doi.org/10.1021/cr100233r
Rotstein BH, Zaretsky S, Rai V, Yudin AK (2014) Small heterocycles in multicomponent reactions. Chem Rev 114(16):8323–8359. https://doi.org/10.1021/cr400615v
Yu J, Shi F, Gong L-Z (2011) Brønsted-acid-catalyzed asymmetric multicomponent reactions for the facile synthesis of highly enantioenriched structurally diverse nitrogenous heterocycles. Acc Chem Res 44(11):1156–1171. https://doi.org/10.1021/ar2000343
Shi T, Zerio CJ, Sivinski J, Ambrose AJ, Moore KT, Buckley T, Kaneko L, Zhang M, Zhang T, Chapman E (2019) A one-step, atom economical synthesis of thieno[2,3-d]pyrimidin-4-amine derivatives via a four-component reaction. Eur J Org Chem 20(2):3269–3272. https://doi.org/10.1002/ejoc.201900414
Liu P, Hao J-W, Mo L-P, Zhang Z-H (2015) Recent advances in the application of deep eutectic solvents as sustainable media as well as catalysts in organic reactions. RSC Adv 5(60):48675–48704. https://doi.org/10.1039/C5RA05746A
Sunderhaus JD, Martin SF (2009) Applications of multicomponent reactions to the synthesis of diverse heterocyclic scaffolds. Chemistry 15(6):1300–1308. https://doi.org/10.1002/chem.200802140
Abbaraju S, Zhao JC-G (2014) Asymmetric aldol reaction of 3-acetyl-2H-chromen-2-ones and isatins catalyzed by a bifunctional quinidine urea catalyst. Adv Synth Catal 356(1):237–241. https://doi.org/10.1002/adsc.201300623
Sun X-X, Li C, He Y-Y, Zhu Z-Q, Mei G-J, Shi F (2017) Catalytic asymmetric [3+3] cycloaddition of azomethine ylides with C3-substituted 2-indolylmethanols. Adv Synth Catal 359(15):2660–2670. https://doi.org/10.1002/adsc.201700203
Patel DM, Sharma MG, Vala RM, Lagunes I, Puerta A, Padrón JM, Rajani DP, Patel HM (2019) Hydroxyl alkyl ammonium ionic liquid assisted green and one-pot regioselective access to functionalized pyrazolodihydropyridine core and their pharmacological evaluation. Bioorg Chem 86:137–150. https://doi.org/10.1016/j.bioorg.2019.01.029
**e L-Y, Jiang L-L, Tan J-X, Wang Y, Xu X-Q, Zhang B, Cao Z, He W-M (2019) Visible-light-initiated decarboxylative alkylation of quinoxalin-2(1H)-ones with phenyliodine(III) dicarboxylates in recyclable ruthenium(II) catalytic system. ACS Sustain Chem Eng 7(16):14153–14160. https://doi.org/10.1021/acssuschemeng.9b02822
Ma N, Zhang M-M, Wang X-S (2016) Parallel synthesis of pyrrolo[3,2-f]quinolines (PQQ skeleton) library via a one-pot three-component reaction under catalyst-free conditions. Polycyclic Aromat Compd 36(5):683–696. https://doi.org/10.1080/10406638.2015.1046608
Shi F, Zhang S, Wu S-S, Gao Y, Tu S-J (2011) A diversity-oriented synthesis of pyrazolo[4,3-f]quinoline derivatives with potential bioactivities via microwave-assisted multi-component reactions. Mol Diversity 15(2):497–505. https://doi.org/10.1007/s11030-010-9272-3
Zhang M-M, Chen D-S, Wang X-S (2014) A three-component domino reaction for efficient synthesis of functionalized pyrazolo[3,4-f]quinolines under catalyst-free conditions. Res Chem Intermed. https://doi.org/10.1007/s11164-014-1743-1
Damavandi S, Sandaroos R, Mohammadi A (2013) Ultrasonic-assisted Cu-catalyzed multicomponent synthesis of furo[3,4-b]pyrazolo[4,3-f]quinolinones. Heterocycl Commun. https://doi.org/10.1515/hc-2011-0086
Sandaroos R, Damavandi S (2013) Synthesis of 10-aryl-7,9-dihydro-3H-furo[3,4-b]pyrazolo[4,3-f]quinolin-9-one using supported ionic liquid. Res Chem Intermed. https://doi.org/10.1007/s11164-013-1289-7
Xu H, Li L, Dai L, Mao K, Kou W, Lin C, Rong L (2018) The efficient in-situ reduction and cyclization reaction of aromatic aldehyde, 1,3-cyclopentanedione (tetronic acid), and nitro-compound under SnCl2·2H2O-THF medium. Appl Organomet Chem 32(3):e4194. https://doi.org/10.1002/aoc.4194
Brahmachari G, Mandal M, Karmakar I, Nurjamal K, Mandal B (2019) Ultrasound-promoted expedient and green synthesis of diversely functionalized 6-amino-5-((4-hydroxy-2-oxo-2H-chromen-3-yl)(aryl)methyl)pyrimidine-2,4(1H,3H)-diones via one-pot multicomponent reaction under sulfamic acid catalysis at ambient conditions. ACS Sustain Chem Eng 7(6):6369–6380. https://doi.org/10.1021/acssuschemeng.9b00133
Frindy S, Primo A, Lahcini M, Bousmina M, Garcia H, El Kadib A (2015) Pd embedded in chitosan microspheres as tunable soft-materials for Sonogashira cross-coupling in water–ethanol mixture. Green Chem 17(3):1893–1898. https://doi.org/10.1039/C4GC02175D
Martins MAP, Frizzo CP, Moreira DN, Zanatta N, Bonacorso HG (2008) Ionic liquids in heterocyclic synthesis. Chem Rev 108(6):2015–2050. https://doi.org/10.1021/cr078399y
Brahmachari G, Karmakar I, Nurjamal K (2018) Ultrasound-assisted expedient and green synthesis of a new series of diversely functionalized 7-aryl/heteroarylchromeno[4,3-d]pyrido[1,2-a]pyrimidin-6(7H)-ones via one-pot multicomponent reaction under sulfamic acid catalysis at ambient conditions. ACS Sustain Chem Eng 6(8):11018–11028. https://doi.org/10.1021/acssuschemeng.8b02448
Baig RBN, Varma RS (2012) Alternative energy input: mechanochemical, microwave and ultrasound-assisted organic synthesis. Chem Soc Rev 41(4):1559–1584. https://doi.org/10.1039/C1CS15204A
Puri S, Kaur B, Parmar A, Chopra H (2013) Applications of ultrasound in organic synthesis—a green approach. Curr Org Chem 17:1790–1828. https://doi.org/10.2174/13852728113179990018
Soldo L, Ruggieri A, Milanese C, Pinza M, Guglielmotti A (2004) Bendazac lysine inhibition of human lens epithelial cell adhesion to polymethylmethacrylate intraocular lenses. Ophthalmic Res 36(3):145–150. https://doi.org/10.1159/000077327
Dayal N, Wang M, Sintim HO (2020) HSD1787, a tetrahydro-3H-pyrazolo[4,3-f]quinoline compound synthesized via povarov reaction, potently inhibits proliferation of cancer cell lines at nanomolar concentrations. ACS Omega 5(37):23799–23807. https://doi.org/10.1021/acsomega.0c03001
Denya I, Malan SF, Joubert J (2018) Indazole derivatives and their therapeutic applications: a patent review (2013–2017). Expert Opin Ther Pat 28(6):441–453. https://doi.org/10.1080/13543776.2018.1472240
Chung T-W, Narhe BD, Lin C-C, Sun C-M (2015) Multicomponent synthesis of functionalized tetrahydroacridinones: insights into a mechanistic route. Org Lett 17(21):5368–5371. https://doi.org/10.1021/acs.orglett.5b02705
Khaligh NG (2017) Synthesis of tetrahydrotriazoloacridines: a synergistic effect of microwave irradiation and Brönsted acidic ionic liquids. J Heterocycl Chem 54(6):3350–3357. https://doi.org/10.1002/jhet.2955
Xu H, Li L, Lin C, Kou W, Ling Z, Xu Z, Rong L (2018) An efficient Fe/H2O medium in situ reduction and cyclization reaction for the synthesis of pyrazolo[3,4-a]acridin-10-one and pyrazolo[4,3-a]acridin-10-one derivatives. Synthesis 50(03):583–592. https://doi.org/10.1055/s-0036-1589124;ArtID:ss-2017-h0529-op
Xavier S, Periandy S, Ramalingam S (2015) NBO, conformational, NLO, HOMO–LUMO, NMR and electronic spectral study on 1-phenyl-1-propanol by quantum computational methods. Spectrochim Acta Part A Mol Biomol Spectrosc 137:306–320. https://doi.org/10.1016/j.saa.2014.08.039
Vengatesh G, Sundaravadivelu M (2021) Synthesis, 2D NMR, crystal structure, Hirshfeld surface, stereochemical and DFT studies of 4,8,9,10-tetraaryl-1,3-diazaadamantan-6-one O-methoxy oximes. J Mol Struct 1229:129653. https://doi.org/10.1016/j.molstruc.2020.129653
Chouchène N, Toumi A, Boudriga S, Edziri H, Sobeh M, Abdelfattah MAO, Askri M, Knorr M, Strohmann C, Brieger L, Soldera A (2022) Antimicrobial activity and DFT studies of a novel set of spiropyrrolidines tethered with thiochroman-4-one/chroman-4-one scaffolds. Molecules. https://doi.org/10.3390/molecules27030582
Wu Q, Chen Y, Chen D, Zhou Z (2021) Synthesis, crystal structure and vibrational properties studies of 2-((4-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)phenoxy)methyl) benzonitrile and N-(3-bromobenzyl)-4-(4,4,5,5-tetramethyl-1,3,2- dioxaborolan-2-yl)aniline. J Mol Struct 1229:129782. https://doi.org/10.1016/j.molstruc.2020.129782
Erkan S (2019) Activity of the rocuronium molecule and its derivatives: a theoretical calculation. J Mol Struct 1189:257–264. https://doi.org/10.1016/j.molstruc.2019.04.042
Sayin K, Üngördü A (2019) Investigations of structural, spectral and electronic properties of enrofloxacin and boron complexes via quantum chemical calculation and molecular docking. Spectrochim Acta Part A Mol Biomol Spectrosc 220:117102. https://doi.org/10.1016/j.saa.2019.05.007
Tadić JD, Lađarević JM, Vitnik ŽJ, Vitnik VD, Stanojković TP, Matić IZ, Mi** DZ (2021) Novel azo pyridone dyes based on dihydropyrimidinone skeleton: Synthesis, DFT study and anticancer activity. Dyes Pigm 187:109123. https://doi.org/10.1016/j.dyepig.2020.109123
Kökbudak Z, Saracoglu M, Akkoç S, Çimen Z, Yilmazer MI, Kandemirli F (2020) Synthesis, cytotoxic activity and quantum chemical calculations of new 7-thioxopyrazolo[1,5-f]pyrimidin-2-one derivatives. J Mol Struct 1202:127261. https://doi.org/10.1016/j.molstruc.2019.127261
Talu M, Demiroğlu EU, Yurdakul Ş, Badoğlu S (2015) FTIR, Raman and NMR spectroscopic and DFT theoretical studies on poly(N-vinylimidazole). Spectrochim Acta Part A Mol Biomol Spectrosc 134:267–275. https://doi.org/10.1016/j.saa.2014.06.101
Ghareeb EA, Mahmoud NFH, El-Bordany EA, El-Helw EAE (2021) Synthesis, DFT, and eco-friendly insecticidal activity of some N-heterocycles derived from 4-((2-oxo-1,2-dihydroquinolin-3-yl)methylene)-2-phenyloxazol-5(4H)-one. Bioorg Chem 112:104945. https://doi.org/10.1016/j.bioorg.2021.104945
Rafique R, Khan KM, Arshia CS, Wadood A, Rehman AU, Salar U, Venugopal V, Shamim S, Taha M, Perveen S (2020) Synthesis, in vitro α-amylase inhibitory, and radicals (DPPH & ABTS) scavenging potentials of new N-sulfonohydrazide substituted indazoles. Bioorg Chem 94:103410. https://doi.org/10.1016/j.bioorg.2019.103410
Chen H, Zhang X, Zhang X, Fan Z, Liu W, Lei Y, Zhu C, Ma B (2020) Dihydrobenzoxazinone derivatives as aldose reductase inhibitors with antioxidant activity. Bioorg Med Chem 28(20):115699. https://doi.org/10.1016/j.bmc.2020.115699
Najda A, Bains A, Chawla P, Kumar A, Balant S, Walasek-Janusz M, Wach D, Kaushik R (2021) Assessment of anti-inflammatory and antimicrobial potential of ethanolic extract of Woodfordia fruticosa flowers: GC-MS analysis. Molecules. https://doi.org/10.3390/molecules26237193
Dennington R, Keith TA, Millam JMJSI (2016). GaussView, Version 6.1. Semichem Inc., Shawnee Mission
Frisch MJ, Trucks GW, Schlegel HB, Scuseria GE, Robb MA, Cheeseman JR, Scalmani G, Barone V, Petersson GA, Nakatsuji H, Li X, Caricato M, Marenich AV, Bloino J, Janesko BG, Gomperts R, Mennucci B, Hratchian HP, Ortiz JV, Izmaylov AF, Sonnenberg JL, Williams, Ding F, Lipparini F, Egidi F, Goings J, Peng B, Petrone A, Henderson T, Ranasinghe D, Zakrzewski VG, Gao J, Rega N, Zheng G, Liang W, Hada M, Ehara M, Toyota K, Fukuda R, Hasegawa J, Ishida M, Nakajima T, Honda Y, Kitao O, Nakai H, Vreven T, Throssell K, Jr Montgomery JA, Peralta JE, Ogliaro F, Bearpark MJ, Heyd JJ, Brothers EN, Kudin KN, Staroverov VN, Keith TA, Kobayashi R, Normand J, Raghavachari K, Rendell AP, Burant JC, Iyenger SS, Tomasi J, Cossi M, Millam JM, Klene M, Adamo C, Cammi R, Ochterski JW, Martin RL, Morokuma K, Farkas O, Foresman JB, Fox DJ (2016) Gaussian 16 Rev. C.01. Gaussian, Wallingford
Karabacak M, Kose E, Babur Şaş E, Kurt M, Asiri AM, Atac A (2015) DFT calculations and experimental FT-IR, FT-Raman, NMR, UV–Vis spectral studies of 3-fluorophenylboronic acid. Spectrochim Acta Part A Mol Biomol Spectrosc 136:306–320. https://doi.org/10.1016/j.saa.2014.08.141
Bansal G, Singh S, Monga V, Thanikachalam PV, Chawla P (2019) Synthesis and biological evaluation of thiazolidine-2,4-dione-pyrazole conjugates as antidiabetic, anti-inflammatory and antioxidant agents. Bioorg Chem 92:103271. https://doi.org/10.1016/j.bioorg.2019.103271
Jiang Y, Rakesh KP, Alharbi NS, Vivek HK, Manukumar HM, Mohammed YHE, Qin H-L (2019) Radical scavenging and anti-inflammatory activities of (hetero)arylethenesulfonyl fluorides: Synthesis and structure-activity relationship (SAR) and QSAR studies. Bioorg Chem 89:103015. https://doi.org/10.1016/j.bioorg.2019.103015
Yesmin S, Paul A, Naz T, Rahman ABMA, Akhter SF, Wahed MII, Emran TB, Siddiqui SA (2020) Membrane stabilization as a mechanism of the anti-inflammatory activity of ethanolic root extract of Choi (Piper chaba). Clin Phytosci 6(1):59. https://doi.org/10.1186/s40816-020-00207-7
Malik A, Najda A, Bains A, Nurzyńska-Wierdak R, Chawla P (2021) Characterization of Citrus nobilis peel methanolic extract for antioxidant, antimicrobial, and anti-inflammatory activity. Molecules. https://doi.org/10.3390/molecules26144310
Acknowledgements
This research was supported by the Department of Chemistry, Sardar Patel University, Vallabh Vidyanagar, Gujarat, India. All the authors are grateful to UGC, New Delhi for UGC-CPEPA Phase-II program sponsored under award letter no. F. No. 1-14/2002-2016 (NS/PE) dated 28 April, 2016, as well as UGC-CAS, Phase-II sponsored under award latter no. F-540/5/CASII/2018 (SAP-I) dated 25 July, 2018 for the assistance in general. We also thankful to Microbiology Department of R. C. Patel A. S. C College, Shirpur, Maharashtra, India for the biological screening of the compounds reported herein. The authors are also thankful to SynZeal Research Pvt. Ltd. Ahmedabad for analytical testing. One of the authors (Dipakkumar D. Chudasama) is grateful to the SHODH-ScHeme of Develo** High quality research, student Ref No: 202001640021, Education Department, Gujarat State.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Chudasama, D.D., Patel, M.S., Parekh, J.N. et al. Ultrasound-promoted convenient and ionic liquid [BMIM]BF4 assisted green synthesis of diversely functionalized pyrazolo quinoline core via one-pot multicomponent reaction, DFT study and pharmacological evaluation. Mol Divers 27, 1409–1425 (2023). https://doi.org/10.1007/s11030-022-10498-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11030-022-10498-2