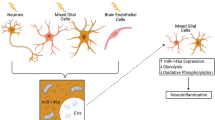
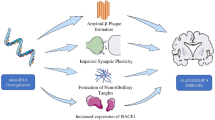
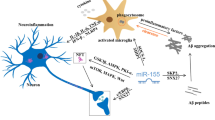
Abstract
Small non-coding RNAs (miRNAs) regulate gene expression by binding to mRNA and mediating its degradation or inhibiting translation. Since miRNAs can regulate the expression of several genes, they have multiple roles to play in biological processes and human diseases. The majority of miRNAs are known to be expressed in the brain and are involved in synaptic functions, thus marking their presence and role in major neurodegenerative disorders, including Alzheimer’s disease (AD). In AD, amyloid beta (Aβ) plaques and neurofibrillary tangles (NFTs) are known to be the major hallmarks. The clearance of Aβ and tau is known to be associated with miRNA dysregulation. In addition, the β-site APP cleaving enzyme (BACE 1), which cleaves APP to form Aβ, is also found to be regulated by miRNAs, thus directly affecting Aβ accumulation. Growing evidences suggest that neuroinflammation can be an initial event in AD pathology, and miRNAs have been linked with the regulation of neuroinflammation. Inflammatory disorders have also been associated with AD pathology, and exosomes associated with miRNAs are known to regulate brain inflammation, suggesting for the role of systemic miRNAs in AD pathology. Several miRNAs have been related in AD, years before the clinical symptoms appear, most of which are associated with regulating the cell cycle, immune system, stress responses, cellular senescence, nerve growth factor (NGF) signaling, and synaptic regulation. Phytochemicals, especially polyphenols, alter the expression of various miRNAs by binding to miRNAs or binding to the transcriptional activators of miRNAs, thus control/alter various metabolic pathways. Awing to the sundry biological processes being regulated by miRNAs in the brain and regulation of expression of miRNAs via phytochemicals, miRNAs and the regulatory bioactive phytochemicals can serve as therapeutic agents in the treatment and management of AD.





Similar content being viewed by others
Data availability
The MS being a review article, no data to be shared, not applicable.
Code availability
Not applicable.
References
Abuelezz NZ, Nasr FE, AbdulKader MA, Bassiouny AR, Zaky A (2021) MicroRNAs as potential orchestrators of Alzheimer’s disease-related pathologies: insights on current status and future possibilities. Front Aging Neurosci 13:743573
Akter R, Rahman H, Behl T, Chowdhury MA, Manirujjaman M, Bulbul IJ, Bungau S (2021) Prospective role of polyphenolic compounds in the treatment of neurodegenerative diseases. CNS Neurol Disord-Drug Targets 20(5):430–450
An F, Gong G, Wang Y, Bian M, Yu L, Wei C (2017) MiR-124 acts as a target for Alzheimer’s disease by regulating BACE1. Oncotarget 8(69):114065
An J, Cai T, Che H, Yu T, Cao Z, Liu X, ..., Luo W (2014) The changes of miRNA expression in rat hippocampus following chronic lead exposure. Toxicol Lett 229(1):158–166
Angelucci F, Cechova K, Valis M, Kuca K, Zhang B, Hort J (2019) MicroRNAs in Alzheimer’s Disease: diagnostic markers or therapeutic agents? Front Pharmacol 10:665
Arnold SE, Arvanitakis Z, Macauley-Rambach SL, Koenig AM, Wang HY, Ahima RS, ..., Nathan DM (2018) Brain insulin resistance in type 2 diabetes and Alzheimer disease: concepts and conundrums. Nat Rev Neurol 14(3):168–181
Arora T, Prashar V, Singh R, Barwal TS, Changotra H, Sharma A, Parkash J (2022) Dysregulated miRNAs in progression and pathogenesis of Alzheimer’s disease. Mol Neurobiol 59(10):6107–6124
Aschrafi A, Schwechter AD, Mameza MG, Natera-Naranjo O, Gioio AE, Kaplan BB (2008) MicroRNA-338 regulates local cytochrome c oxidase IV mRNA levels and oxidative phosphorylation in the axons of sympathetic neurons. J Neurosci 28(47):12581–12590
Balakrishnan R, Azam S, Cho DY, Su-Kim I, Choi DK (2021) Natural phytochemicals as novel therapeutic strategies to prevent and treat Parkinson’s disease: current knowledge and future perspectives. Oxid Med Cell Longev 2021:6680935–6680967
Banerjee N, Talcott S, Safe S, Mertens-Talcott SU (2012) Cytotoxicity of pomegranate polyphenolics in breast cancer cells in vitro and vivo: potential role of miRNA-27a and miRNA-155 in cell survival and inflammation. Breast Cancer Research and Ttreatment 136(1):21–34
Banzhaf-Strathmann J, Benito E, May S, Arzberger T, Tahirovic S, Kretzschmar H, Edbauer D (2014) Micro RNA-125b induces tau hyperphosphorylation and cognitive deficits in Alzheimer’s disease. EMBO J 33(15):1667–1680
Barta T, Peskova L, Hampl A (2016) miRNA song: a web-based tool for generation and testing of miRNA sponge constructs in silico. Sci Rep 6:36625
Bazrgar M, Khodabakhsh P, Prudencio M, Mohagheghi F, Ahmadiani A (2021) The role of microRNA-34 family in Alzheimer’s disease: a potential molecular link between neurodegeneration and metabolic disorders. Pharmacol Res 172:105805
Braak H, Braak E (1995) Staging of Alzheimer’s disease-related neurofibrillary changes. Neurobiol Aaging 16(3):271–284
Brites D, Fernandes A (2015) Neuroinflammation and depression: microglia activation, extracellular microvesicles and microRNA dysregulation. Front Cell Neurosci 2015(9):476
Bryniarski K, Ptak W, Martin E, Nazimek K, Szczepanik M, Sanak M, Askenase PW (2015) Free extracellular miRNA functionally targets cells by transfecting exosomes from their companion cells. PLoS ONE 10(4):e0122991
Buciuc M, Botha H, Murray ME, Schwarz CG, Senjem ML, Jones DT, ..., Josephs KA (2020) Utility of FDG-PET in diagnosis of Alzheimer-related TDP-43 proteinopathy. Neurology 95(1):e23-e34
Buzas EI, György B, Nagy G, Falus A, Gay S (2014) Emerging role of extracellular vesicles in inflammatory diseases. Nat Rev Rheumatol 10(6):356–364
Chaudhary A, Jaswal VS, Choudhary S, Sharma A, Beniwal V, Tuli HS, Sharma S (2019) Ferulic acid: a promising therapeutic phytochemical and recent patents advances. Recent Pat Inflammation Allergy Drug Discovery 13(2):115–123
Chaudhuri AD, Yelamanchili SV, Marcondes MCG, Fox HS (2013) Up-regulation of microRNA-142 in simian immunodeficiency virus encephalitis leads to repression of sirtuin1. FASEB J 27(9):3720–3729
Chen JJ, Zhao B, Zhao J, Li S (2017) Potential roles of exosomal MicroRNAs as diagnostic biomarkers and therapeutic application in Alzheimer’s disease. Neural Plast 2017:7027380–7027392
Chen J, Ning R, Zacharek A, Cui C, Cui X, Yan T, Venkat P, Zhang Y, Chopp M (2016) MiR-126 contributes to human umbilical cord blood cell-induced neurorestorative effects after stroke in type-2 diabetic mice. Stem Cells (Dayton, Ohio) 34(1):102–113
Chen L, Na R, Boldt E, Ran Q (2015) NLRP3 inflammasome activation by mitochondrial reactive oxygen species plays a key role in long-term cognitive impairment induced by paraquat exposure. Neurobiol Aging 36(9):2533–2543
Cheng C, Li W, Zhang Z, Yoshimura S, Hao Q, Zhang C, Wang Z (2013) MicroRNA-144 is regulated by activator protein-1 (AP-1) and decreases expression of Alzheimer disease-related a disintegrin and metalloprotease 10 (ADAM10). J Biol Chem 288(19):13748–13761
Cho S, Lee H, Seo J (2021) Impact of genetic risk factors for Alzheimer’s disease on brain glucose metabolism. Mol Neurobiol 58(6):2608–2619
Chu AJ, Williams JM (2022) Astrocytic MicroRNA in ageing, inflammation, and neurodegenerative disease. Front Physiol 12:826697
Collotta M, Bertazzi PA, Bollati V (2013) Epigenetics and pesticides. Toxicology 307:35–41
Cosín-Tomás M, Antonell A, Lladó A, Alcolea D, Fortea J, Ezquerra M, Lleó A, Martí MJ, Pallàs M, Sanchez-Valle R, Molinuevo JL, Sanfeliu C, Kaliman P (2017) Plasma miR-34a-5p and miR-545-3p as early biomarkers of Alzheimer’s disease: potential and limitations. Mol Neurobiol 54(7):5550–5562
Costa C, Teodoro M, Rugolo CA, Alibrando C, Giambò F, Briguglio G, Fenga C (2020) MicroRNAs alteration as early biomarkers for cancer and neurodegenerative diseases: new challenges in pesticides exposure. Toxicol Rep 7:759–767
Croce N, Gelfo F, Ciotti MT, Federici G, Caltagirone C, Bernardini S, Angelucci F (2013) NPY modulates miR-30a-5p and BDNF in opposite direction in an in vitro model of Alzheimer disease: a possible role in neuroprotection? Mol Cell Biochem 376(1):189–195
Dash M, Eid A, Subaiea G, Chang J, Deeb R, Masoud A, ..., Zawia NH (2016) Developmental exposure to lead (Pb) alters the expression of the human tau gene and its products in a transgenic animal model. Neurotoxicology 55:154–159
Davis TH, Cuellar TL, Koch SM, Barker AJ, Harfe BD, McManus MT, Ullian EM (2008) Conditional loss of Dicer disrupts cellular and tissue morphogenesis in the cortex and hippocampus. J Neurosci 28(17):4322–4330
De La Parra C, Castillo-Pichardo L, Cruz-Collazo A, Cubano L, Redis R, Calin GA, Dharmawardhane S (2016) Soy isoflavone genistein-mediated downregulation of miR-155 contributes to the anticancer effects of genistein. Nutr Cancer 68(1):154–164
De Leon MJ, Convit A, Wolf OT, Tarshish CY, DeSanti S, Rusinek H, ..., Fowler J (2001) Prediction of cognitive decline in normal elderly subjects with 2-[18F] fluoro-2-deoxy-D-glucose/positron-emission tomography (FDG/PET). Proc Natl Acad Sci 98(19):10966–10971
Deisseroth K, Bito H, Tsien RW (1996) Signaling from synapse to nucleus: postsynaptic CREB phosphorylation during multiple forms of hippocampal synaptic plasticity. Neuron 16(1):89–101
Denk J, Oberhauser F, Kornhuber J, Wiltfang J, Fassbender K, Schroeter ML, FTLDc study group (2018) Specific serum and CSF microRNA profiles distinguish sporadic behavioural variant of frontotemporal dementia compared with Alzheimer patients and cognitively healthy controls. PLoS ONE 13(5):e0197329
Di Meco A, Praticò D (2016) MicroRNAs as therapeutic targets for Alzheimer’s disease. J Alzheimer’s Dis 53(2):367–372
Doxakis E (2010) Post-transcriptional regulation of α-synuclein expression by mir-7 and mir-153. J Biol Chem 285(17):12726–12734
Dutta S, Hornung S, Taha HB, Bitan G (2023) Biomarkers for parkinsonian disorders in CNS-originating EVs: promise and challenges. Acta Neuropathol 145(5):515–540
Edbauer D, Neilson JR, Foster KA, Wang CF, Seeburg DP, Batterton MN, Tada T, Dolan BM, Sharp PA, Sheng M (2010) Regulation of synaptic structure and function by FMRP-associated microRNAs miR-125b and miR-132. Neuron 65(3):373–384
Eid A, Bihaqi SW, Renehan WE, Zawia NH (2016) Developmental lead exposure and lifespan alterations in epigenetic regulators and their correspondence to biomarkers of Alzheimer’s disease. Alzheimers Dementia 2:123–131
Fagiani F, Govoni S, Racchi M, Lanni C (2021) The peptidyl-prolyl isomerase Pin1 in neuronal signaling: from neurodevelopment to neurodegeneration. Mol Neurobiol 58(3):1062–1073
Femminella GD, Ferrara N, Rengo G (2015) The emerging role of microRNAs in Alzheimer’s disease. Front Physiol 6:40
Fernández-de Frutos M, Galán-Chilet I, Goedeke L, Kim B, Pardo-Marqués V, Pérez-García A, Herrero JI, Fernández-Hernando C, Kim J, Ramírez CM (2019) MicroRNA 7 impairs insulin signaling and regulates Aβ levels through posttranscriptional regulation of the insulin receptor substrate 2, insulin receptor, insulin-degrading enzyme, and liver X receptor pathway. Mol Cell Biol 39(22):e00170-e219
Fiore R, Khudayberdiev S, Christensen M, Siegel G, Flavell SW, Kim TK, Schratt G (2009) Mef2-mediated transcription of the miR379–410 cluster regulates activity-dependent dendritogenesis by fine-tuning Pumilio2 protein levels. EMBO J 28(6):697–710
Forner S, Baglietto-Vargas D, Martini AC, Trujillo-Estrada L, LaFerla FM (2017) Synaptic impairment in Alzheimer’s disease: a dysregulated symphony. Trends Neurosci 40(6):347–357
Fukazawa Y, Saitoh Y, Ozawa F, Ohta Y, Mizuno K, Inokuchi K (2003) Hippocampal LTP is accompanied by enhanced F-actin content within the dendritic spine that is essential for late LTP maintenance in vivo. Neuron 38(3):447–460
Garland EF, Hartnell IJ, Boche D (2022) Microglia and astrocyte function and communication: what do we know in humans? Front Neurosci 16:824888
Gautam V, D’Avanzo C, Berezovska O, Tanzi RE, Kovacs DM (2015) Synaptotagmins interact with APP and promote Aβ generation. Mol Neurodegener 10(1):1–15
Geekiyanage H, Chan C (2011) MicroRNA-137/181c regulates serine palmitoyltransferase and in turn amyloid β, novel targets in sporadic Alzheimer’s disease. J Neurosci 31(41):14820–14830
Geng L, Zhang T, Liu W, Chen Y (2018) Inhibition of miR-128 abates Aβ-mediated cytotoxicity by targeting PPAR-γ via NF-κB inactivation in primary mouse cortical neurons and Neuro2a cells. Yonsei Med J 59(9):1096–1106
Gerasymchuk D, Hubiernatorova A, Domanskyi A (2020) MicroRNAs regulating cytoskeleton dynamics, endocytosis, and cell motility—a link between neurodegeneration and cancer? Front Neurol 11:1–13
Gharibzadeh S, Hoseini SS (2008) Arsenic exposure may be a risk factor for Alzheimer’s disease. J Neuropsychiatry Clin Neurosci 20(4):501–501
Ghafouri-Fard S, Shoorei H, Dong P, Poornajaf Y, Hussen BM, Taheri M, Dilmaghani NA (2023) Emerging functions and clinical applications of exosomal microRNAs in diseases. Non-coding RNA Research 8(3):350–362
Giambò F, Leone GM, Gattuso G, Rizzo R, Cosentino A, Cinà D, ..., Falzone L (2021) Genetic and epigenetic alterations induced by pesticide exposure: integrated analysis of gene expression, microRNA expression, and DNA methylation datasets. Int J Environ Res Public Health 18(16):8697
Guan Y, Rao Z, Chen C (2018) miR-30a suppresses lung cancer progression by targeting SIRT1. Oncotarget 9(4):4924–4934
Guedes JR, Santana I, Cunha C, Duro D, Almeida MR, Cardoso AM, Cardoso AL (2016) MicroRNA deregulation and chemotaxis and phagocytosis impairment in Alzheimer’s disease. Alzheimers Dementia 3:7–17
Hannun YA, Obeid LM (2008) Principles of bioactive lipid signalling: lessons from sphingolipids. Nat Rev Mol Cell Biol 9(2):139–150
Hara N, Kikuchi M, Miyashita A, Hatsuta H, Saito Y, Kasuga K, Kuwano R (2017) Serum microRNA miR-501-3p as a potential biomarker related to the progression of Alzheimer’s disease. Acta Neuropathol Commun 5(1):1–9
Hernandez-Rapp J, Rainone S, Hébert SS (2017) MicroRNAs underlying memory deficits in neurodegenerative disorders. Progress Nneuro-Psychopharmacol Biol Psychiatry 73:79–86
He D, Tan J, Zhang J (2017) miR-137 attenuates Abeta-induced neurotoxicity through inactivation of NF-kappaB pathway by targeting TNFAIP1 in Neuro2a cells. Biochem Biophys Res Commun 490:941–947
Hong H, Li Y, Su B (2017) Identification of circulating miR-125b as a potential biomarker of Alzheimer’s disease in APP/PS1 transgenic mouse. J Alzheimers Dis 59(4):1449–1458
Horgan D, Nobili F, Teunissen C, Grimmer T, Mitrecic D, Ris L, Scarmeas N (2020) Biomarker testing: piercing the fog of Alzheimer’s and related dementia. Biomedicine Hub 5(3):1–22
Hou Q, Ruan H, Gilbert J, Wang G, Ma Q, Yao WD, Man HY (2015) MicroRNA miR124 is required for the expression of homeostatic synaptic plasticity. Nat Commun 6(1):1–12
Hou TY, Zhou Y, Zhu LS, Wang X, Pang P, Wang DQ, Liuyang ZY, Man H, Lu Y, Zhu LQ, Liu D (2020) Correcting abnormalities in miR-124/PTPN1 signaling rescues tau pathology in Alzheimer’s disease. J Neurochem 154(4):441–457
Hu G, Yao H, Chaudhuri AD, Duan M, Yelamanchili SV, Wen H, Buch S (2012) Exosome-mediated shuttling of microRNA-29 regulates HIV Tat and morphine-mediated neuronal dysfunction. Cell Death Dis 3(8):e381–e381
Ismail N, Wang Y, Dakhlallah D, Moldovan L, Agarwal K, Batte K, Marsh CB (2013) Macrophage microvesicles induce macrophage differentiation and miR-223 transfer. Blood J Am Soc Hematol 121(6):984–995
Jeena GS, Singh N, Shikha, Shukla RK (2022) An insight into microRNA biogenesis and its regulatory role in plant secondary metabolism. Plant Cell Rep 41(8):1651–1671
Ji H, Xu L, Wang Z, Fan X, Wu L (2018) Differential microRNA expression in the prefrontal cortex of mouse offspring induced by glyphosate exposure during pregnancy and lactation. Exp Ther Med 15(3):2457–2467
Jian C, Lu M, Zhang Z, Liu L, Li X, Huang F, Zou D (2017) miR-34a knockout attenuates cognitive deficits in APP/PS1 mice through inhibition of the amyloidogenic processing of APP. Life Sci 182:104–111
**g X, Yang J, Jiang L, Chen J, Wang H (2018) MicroRNA-29b regulates the mitochondria-dependent apoptotic pathway by targeting Bax in doxorubicin cardiotoxicity. Cell Physiol Biochem 48(2):692–704
Kang H (2019) MicroRNA-mediated health-promoting effects of phytochemicals. Int J Mol Sci 20(10):2535–2546
Kanthasamy A, ** H, Anantharam V, Sondarva G, Rangasamy V, Rana A, Kanthasamy A (2012) Emerging neurotoxic mechanisms in environmental factors-induced neurodegeneration. Neurotoxicology 33(4):833–837
Karki HP, Jang Y, Jung J, Oh J (2021) Advances in the development paradigm of biosample-based biosensors for early ultrasensitive detection of Alzheimer’s disease. J Nanobiotechnol 19(1):1–24
Kaur N, Kaur S, Saini M, Dhiman M, Mantha AK (2020) Phytochemical ginkgolide b protectscultured neuroblastoma sh-sy5y cells against aβ (25–35) induced oxidative stress responses bymaintaining the mitochondrial integrity. Rasayan J Chem 13(1):306–321
Kaur S, Verma H, Kaur S, Singh S, Mantha AK, Dhiman M (2022) Herbal Remedies for Improving Cancer Treatment Through Modulation of Redox Balance. In: Chakraborti S (eds) Handbook of Oxidative Stress in Cancer: Therapeutic Aspects. Springer, Singapore. https://doi.org/10.1007/978-981-16-1247-3_270-1.
Kaur N, Dhiman M, Perez-Polo JR, Mantha AK (2015a) Ginkgolide B revamps neuroprotective role of apurinic/apyrimidinic endonuclease 1 and mitochondrial oxidative phosphorylation against Aβ25–35-induced neurotoxicity in human neuroblastoma cells. J Neurosci Res 93(6):938–947
Kaur N, Sarkar B, Mittal S, Dhiman M, Taglialatela G, Perez-Polo RJ, Mantha AK (2015b) Oxidative stress events and neuronal dysfunction in Alzheimer’s disease: focus on APE1/Ref-1-mediated survival strategies. Free Radicals in Human health and Disease. Springer, New Delhi, p 175–207
Kaur S, Dhiman M, Mantha AK (2018) Ferulic Acid: A Natural Antioxidant with Application towards Neuroprotection against Alzheimer’s Disease. Functional Food and Human Health. Springer, Singapore, p 575–586
Kaur S, Verma H, Dhiman M, Tell G, Gigli GL, Janes F, Mantha AK (2021) Brain exosomes: friend or foe in Alzheimer’s disease? Mol Neurobiol 58(12):6610–6624
Khodabakhsh P, Bazrgar M, Mohagheghi F, Parvardeh S, Ahmadiani A (2022) MicroRNA-140-5p inhibitor attenuates memory impairment induced by amyloid-ß oligomer in vivo possibly through Pin1 regulation. CNS Neurosci Ther 29(1):91–103
Kiko T, Nakagawa K, Tsuduki T, Furukawa K, Arai H, Miyazawa T (2014) MicroRNAs in plasma and cerebrospinal fluid as potential markers for Alzheimer’s disease. J Alzheimers Dis 39(2):253–259
Komatsu S, Kitai H, Suzuki HI (2023) Network regulation of microRNA biogenesis and target interaction. Cells 12(2):306
Kong WQ, Bai R, Liu T, Cai CL, Liu M, Li X, Tang H (2012) MicroRNA-182 targets cAMP-responsive element-binding protein 1 and suppresses cell growth in human gastric adenocarcinoma. FEBS J 279(7):1252–1260
Kumar S, Reddy PH (2018) MicroRNA-455-3p as a potential biomarker for Alzheimer’s disease: an update. Frontiers in Aging Neuroscience 10:41
Kumar S, Reddy PH (2020) The role of synaptic microRNAs in Alzheimer’s disease. BBA-Mol Basis Dis 1866(12):165937
Kumar S, Orlov E, Gowda P, Bose C, Swerdlow RH, Lahiri DK, Reddy PH (2022) Synaptosome microRNAs regulate synapse functions in Alzheimer’s disease. NPJ Genom Med 7(1):47
Kuzuya A, Zoltowska KM, Post KL, Arimon M, Li X, Svirsky S, ..., Berezovska O (2016) Identification of the novel activity-driven interaction between synaptotagmin 1 and presenilin 1 links calcium, synapse, and amyloid beta. BMC Biol 14(1):1–24
Kye MJ, Liu T, Levy SF, Xu NL, Groves BB, Bonneau R, Kosik KS (2007) Somatodendritic microRNAs identified by laser capture and multiplex RT-PCR. RNA 13(8):1224–1234
Lahiri DK, Chen D, Vivien D, Ge YW, Greig NH, Rogers JT (2003) Role of cytokines in the gene expression of amyloid β–protein precursor: identification of a 5’-UTR-Binding nuclear factor and its implications in Alzheimer’s disease. J Alzheimers Dis 5(2):81–90
Lam JK, Chow MY, Zhang Y, Leung SW (2015) siRNA versus miRNA as therapeutics for gene silencing. Mol Ther-Nucleic Acids 4:e252
Lee ST, Chu K, Jung KH, Kim JH, Huh JY, Yoon H, Roh JK (2012) miR-206 regulates brain-derived neurotrophic factor in Alzheimer disease model. Ann Neurol 72(2):269–277
Lee YS, Lewis JA, Ippolito DL, Hussainzada N, Lein PJ, Jackson DA, Stallings JD (2016) Repeated exposure to neurotoxic levels of chlorpyrifos alters hippocampal expression of neurotrophins and neuropeptides. Toxicology 340:53–62
Lehmann SM, Krüger C, Park B, Derkow K, Rosenberger K, Baumgart J, Lehnardt S (2012) An unconventional role for miRNA: let-7 activates Toll-like receptor 7 and causes neurodegeneration. Nat Neurosci 15(6):827–835
Lei X, Lei L, Zhang Z, Zhang Z, Cheng Y (2015) Downregulated miR-29c correlates with increased BACE1 expression in sporadic Alzheimer’s disease. Int J Clin Exp Pathol 8(2):1565–1574
Leitão AL, Enguita FJ (2022) A structural view of miRNA biogenesis and function. Non-Coding RNA 8(1):10
Li HH, Lin SL, Huang CN, Lu FJ, Chiu PY, Huang WN et al (2016) miR-302 attenuates Amyloid-beta-induced neurotoxicity through activation of Akt signaling. J Alzheimers Dis 50:1083–1098
Li JJ, Wang B, Kodali MC, Chen C, Kim E, Patters BJ, Liao FF (2018) In vivo evidence for the contribution of peripheral circulating inflammatory exosomes to neuroinflammation. J Neuroinflammation 15(1):1–16
Li J, Wang H (2018) miR-15b reduces amyloid-β accumulation in SH-SY5Y cell line through targetting NF-κB signaling and BACE1. Biosci Rep 38(6):BSR20180051
Li SH, Gao P, Wang LT, Yan YH, **a Y, Song J, Yang JX (2017) Osthole stimulated neural stem cells differentiation into neurons in an Alzheimer’s disease cell model via upregulation of microRNA-9 and rescued the functional impairment of hippocampal neurons in APP/PS1 transgenic mice. Front Neurosci 11:340–355
Li X, **e W, **e C, Huang C, Zhu J, Liang Z, Zhong C (2014) Curcumin modulates miR-19/PTEN/AKT/p53 axis to suppress bisphenol A-induced MCF-7 breast cancer cell proliferation. Phytother Res 28(10):1553–1560
Lim AC, Qi RZ (2003) Cyclin-dependent kinases in neural development and degeneration. J Alzheimers Dis 5(4):329–335
Liu CG, Song J, Zhang YQ, Wang PC (2014) MicroRNA-193b is a regulator of amyloid precursor protein in the blood and cerebrospinal fluid derived exosomal microRNA-193b is a biomarker of Alzheimer’s disease. Mol Med Rep 10(5):2395–2400
Liu J, Ye T, Zhang Y, Zhang R, Kong Y, Zhang Y, Sun J (2021) Protective effect of Ginkgolide B against cognitive impairment in mice via regulation of gut microbiota. J Agric Food Chem 69(41):12230–12240
Liu Y, Xu Y, Yu M (2022) MicroRNA-4722-5p and microRNA-615-3p serve as potential biomarkers for Alzheimer’s disease. Exp Ther Med 23(3):1–10
Long JM, Lahiri DK (2011) MicroRNA-101 downregulates Alzheimer’s amyloid-β precursor protein levels in human cell cultures and is differentially expressed. Biochem Biophys Res Commun 404(4):889–895
Long JM, Maloney B, Rogers JT, Lahiri DK (2019) Novel upregulation of amyloid-β precursor protein (APP) by microRNA-346 via targeting of APP mRNA 5′-untranslated region: implications in Alzheimer’s disease. Mol Psychiatry 24(3):345–363
Long JM, Ray B, Lahiri DK (2012) MicroRNA-153 physiologically inhibits expression of amyloid-β precursor protein in cultured human fetal brain cells and is dysregulated in a subset of Alzheimer disease patients. J Biol Chem 287(37):31298–31310
Long JM, Ray B, Lahiri DK (2014) MicroRNA-339-5p down-regulates protein expression of β-site amyloid precursor protein-cleaving enzyme 1 (BACE1) in human primary brain cultures and is reduced in brain tissue specimens of Alzheimer disease subjects. J Biol Chem 289(8):5184–5198
Macfarlane LA, Murphy PR (2010) MicroRNA: biogenesis, function and role in cancer. Curr Genomics 11(7):537–561
Maldonado-Lasuncion I, Atienza M, Sanchez-Espinosa MP, Cantero JL (2019) Aging-related changes in cognition and cortical integrity are associated with serum expression of candidate microRNAs for Alzheimer disease. Cereb Cortex 29(10):4426–4437
Manzine PR, Pelucchi S, Horst MA, Vale FA, Pavarini SC, Audano M, ..., Cominetti MR (2018) microRNA 221 Targets ADAM10 mRNA and is downregulated in Alzheimer’s disease. J Alzheimers Dis 61(1):113–123
Marcus C, Mena E, Subramaniam RM (2014) Brain PET in the diagnosis of Alzheimer’s disease. Clin Nucl Med 39(10):e413–e426
McKeever PM, Schneider R, Taghdiri F, Weichert A, Multani N, Brown RA, ..., Tartaglia MC (2018) MicroRNA expression levels are altered in the cerebrospinal fluid of patients with young-onset Alzheimer’s disease. Mol Neurobiol 55(12):8826–8841
Michán S, Li Y, Chou MMH, Parrella E, Ge H, Long JM, Longo VD (2010) SIRT1 is essential for normal cognitive function and synaptic plasticity. J Neurosci 30(29):9695–9707
Momin MY, Gaddam RR, Kravitz M, Gupta A, Vikram A (2021) The challenges and opportunities in the development of MicroRNA Therapeutics: a multidisciplinary viewpoint. Cells 10(11):3097–3118
Moraghebi M, Maleki R, Ahmadi M, Negahi AA, Abbasi H, Mousavi P (2021) In silico analysis of polymorphisms in microRNAs deregulated in Alzheimer disease. Front Neurosci 15:631852
Mosconi L (2005) Brain glucose metabolism in the early and specific diagnosis of Alzheimer’s disease. Eur J Nucl Med Mol Imaging 32(4):486–510
Munagala R, Aqil F, Vadhanam MV, Gupta RC (2013) MicroRNA ‘signature’during estrogen-mediated mammary carcinogenesis and its reversal by ellagic acid intervention. Cancer Lett 339(2):175–184
Nagaraj S, Want A, Laskowska-Kaszub K, Fesiuk A, Vaz S, Logarinho E, Wojda U (2021) Candidate Alzheimer’s disease biomarker miR-483-5p lowers TAU phosphorylation by direct ERK1/2 repression. Int J Mol Sci 22(7):3653
Nagaraj S, Zoltowska KM, Laskowska-Kaszub K, Wojda U (2019) microRNA diagnostic panel for Alzheimer’s disease and epigenetic trade-off between neurodegeneration and cancer. Ageing Res Rev 49:125–143
Narasimhan M, Riar AK, Rathinam ML, Vedpathak D, Henderson G, Mahimainathan L (2014) Hydrogen peroxide responsive miR153 targets Nrf2/ARE cytoprotection in paraquat induced dopaminergic neurotoxicity. Toxicol Lett 228(3):179–191
Navas-Carrillo D, Rivera-Caravaca JM, Sampedro-Andrada A, Orenes-Piñero E (2021) Novel biomarkers in Alzheimer’s disease using high resolution proteomics and metabolomics: miRNAS, proteins and metabolites. Crit Rev Clin Lab Sci 58(3):167–179
O’Brien J, Hayder H, Zayed Y, Peng C (2018) Overview of microRNA biogenesis, mechanisms of actions, and circulation. Front Endocrinol 9:402–414
Otoukesh B, Abbasi M, Gorgani HO, Farahini H, Moghtadaei M, Boddouhi B, Kaghazian P, Hosseinzadeh S, Alaee A (2020) MicroRNAs signatures, bioinformatics analysis of miRNAs, miRNA mimics and antagonists, and miRNA therapeutics in osteosarcoma. Cancer Cell Int 20(1):1–20
Pallocca G, Fabbri M, Sacco MG, Gribaldo L, Pamies D, Laurenza I, Bal-Price A (2013) miRNA expression profiling in a human stem cell-based model as a tool for developmental neurotoxicity testing. Cell Biol Toxicol 29(4):239–257
Pegtel DM, Peferoen L, Amor S (2014) Extracellular vesicles as modulators of cell-to-cell communication in the healthy and diseased brain. Phil Trans R Soc B Biol Sci 369(1652):20130516
Pena-Bautista C, Tarazona-Sánchez A, Braza-Boils A, Balaguer A, Ferré-González L, Cañada-Martínez AJ, ..., Cháfer-Pericás C (2022) Plasma microRNAs as potential biomarkers in early Alzheimer disease expression. Sci Rep 12(1):15589
Phinney DG, Di Giuseppe M, Njah J, Sala E, Shiva S, St Croix CM, Ortiz LA (2015) Mesenchymal stem cells use extracellular vesicles to outsource mitophagy and shuttle microRNAs. Nat Commun 6(1):1–15
Piscopo P, Grasso M, Manzini V, Zeni A, Castelluzzo M, Fontana F, Talarico G, Castellano AE, Rivabene R, Crestini A, Bruno G, Ricci L, Denti MA (2023) Identification of miRNAs regulating MAPT expression and their analysis in plasma of patients with dementia. Front Mol Neurosci 16:1127163
Planel E, Miyasaka T, Launey T, Chui DH, Tanemura K, Sato S, Murayama O, Ishiguro K, Tatebayashi Y, Takashima A (2004) Alterations in glucose metabolism induce hypothermia leading to tau hyperphosphorylation through differential inhibition of kinase and phosphatase activities: implications for Alzheimer’s disease. J Neurosci 24(10):2401–2411
Price NL, Ramirez CM, Fernández-Hernando C (2014) Relevance of microRNA in metabolic diseases. Crit Rev Clin Lab Sci 51(6):305–320
Qiang J, Zhu XW, He J, Tao YF, Bao JW, Zhu JH, Xu P (2020) miR-34a regulates the activity of HIF-1a and P53 signaling pathways by promoting GLUT1 in genetically improved farmed tilapia (GIFT, Oreochromis niloticus) under hypoxia stress. Front Physiol 11:670
Ramirez CM, Dávalos A, Goedeke L, Salerno AG, Warrier N, Cirera-Salinas D, ..., Fernández-Hernando C (2011) MicroRNA-758 regulates cholesterol efflux through posttranscriptional repression of ATP-binding cassette transporter A1. Arterioscler Thromb Vasc Biol 31(11):2707–2714
Ramírez CM, Goedeke L, Rotllan N, Yoon JH, Cirera-Salinas D, Mattison JA, ..., Fernández-Hernando C (2013) MicroRNA 33 regulates glucose metabolism. Mol Cell Biol 33(15):2891–2902
Rayner KJ, Suárez Y, Dávalos A, Parathath S, Fitzgerald ML, Tamehiro N, ..., Fernández-Hernando C (2010) MiR-33 contributes to the regulation of cholesterol homeostasis. Science 328(5985):1570–1573
Reza-Zaldivar EE, Hernández-Sápiens MA, Minjarez B, Gómez-Pinedo U, Sánchez-González VJ, Márquez-Aguirre AL, Canales-Aguirre AA (2020) Dendritic spine and synaptic plasticity in Alzheimer’s disease: a focus on MicroRNA. Front Cell Dev Biol 8:255–264
Riscado MSR (2019) Evaluation of the role of miR-9 and miR-29 in amyloid pathway of Alzheimer's disease (Doctoral dissertation, Universidade da Beira Interior (Portugal)
Rodriguez-Ortiz CJ, Baglietto-Vargas D, Martinez-Coria H, LaFerla FM, Kitazawa M (2014) Upregulation of miR-181 decreases c-Fos and SIRT-1 in the hippocampus of 3xTg-AD mice. J Alzheimers Dis 42(4):1229–1238
Samandari-Bahraseman MR, Jahanshahi M, Barbariha SA, Elyasi L (2018) Altered Micro-RNA regulation and neuroprotection activity of eremostachys labiosiformis in Alzheimer’s disease model. Ann Neurosci 25(3):160–165
Santa-Maria I, Alaniz ME, Renwick N, Cela C, Fulga TA, Van Vactor D, Crary JF (2015) Dysregulation of microRNA-219 promotes neurodegeneration through post-transcriptional regulation of tau. J Clin Investig 125(2):681–686
Sarkar S, Jun S, Rellick S, Quintana DD, Cavendish JZ, Simpkins JW (2016) Expression of microRNA-34a in Alzheimer’s disease brain targets genes linked to synaptic plasticity, energy metabolism, and resting state network activity. Brain Res 1646:139–151
Schonrock N, Humphreys DT, Preiss T, Götz J (2012) Target gene repression mediated by miRNAs miR-181c and miR-9 both of which are down-regulated by amyloid-β. J Mol Neurosci 46(2):324–335
Schonrock N, Ke YD, Humphreys D, Staufenbiel M, Ittner LM, Preiss T, Götz J (2010) Neuronal microRNA deregulation in response to Alzheimer’s disease amyloid-β. PLoS ONE 5(6):e11070
Schratt G (2009) microRNAs at the synapse. Nat Rev Neurosci 10(12):842–849
Schratt GM, Tuebing F, Nigh EA, Kane CG, Sabatini ME, Kiebler M, Greenberg ME (2006) A brain-specific microRNA regulates dendritic spine development. Nature 439(7074):283–289
Selkoe DJ, Hardy J (2016) The amyloid hypothesis of Alzheimer’s disease at 25 years. EMBO Mol Med 8(6):595–608
Shu B, Zhang X, Du G, Fu Q, Huang L (2018) MicroRNA-107 prevents amyloid-beta-induced neurotoxicity and memory impairment in mice. Int J Mol Med 41:1665–1672
Silvestro S, Bramanti P, Mazzon E (2019) Role of miRNAs in Alzheimer’s disease and possible fields of application. Int J Mol Sci 20(16):3979–4005
Sim SE, Lim CS, Kim JI, Seo D, Chun H, Yu NK, Kaang BK (2016) The brain-enriched microRNA miR-9-3p regulates synaptic plasticity and memory. J Neurosci 36(33):8641–8652
Smailagic N, Vacante M, Hyde C, Martin S, Ukoumunne O, Sachpekidis C (2015) 18F-FDG PET for the early diagnosis of Alzheimer’s disease dementia and other dementias in people with mild cognitive impairment (MCI). Cochrane Database Syst Rev 1(1):CD010632
Sohel MH (2016) Extracellular/circulating microRNAs: release mechanisms, functions and challenges. Achieve Life Sci 10(2):175–186
Spillantini MG, Murrell JR, Goedert M, Farlow MR, Klug A, Ghetti B (1998) Mutation in the tau gene in familial multiple system tauopathy with presenile dementia. Proc Natl Acad Sci USA 95(13):7737–7741
Srivastava SK, Arora S, Averett C, Singh S, Singh AP (2015) Modulation of microRNAs by phytochemicals in cancer: underlying mechanisms and translational significance. Biomed Res Int 2015:848710–848719
Sultana R, Butterfield DA (2009) Oxidatively modified, mitochondria-relevant brain proteins in subjects with Alzheimer disease and mild cognitive impairment. J Bioenerg Biomembr 41(5):441–446
Takousis P, Schulz J, Lill CM, Perneczky R, Bertram L (2017) [P3–196]: microRNA expression in Alzheimer disease: a systematic review of the literature and meta-analysis. Alzheimers Dementia 13(7S_Part_21):P1010
Teter B, Morihara T, Lim GP, Chu T, Jones MR, Zuo X, Cole GM (2019) Curcumin restores innate immune Alzheimer’s disease risk gene expression to ameliorate Alzheimer pathogenesis. Neurobiol Dis 127:432–448
Tili E, Michaille JJ, Adair B, Alder H, Limagne E, Taccioli C, Croce CM (2010) Resveratrol decreases the levels of miR-155 by upregulating miR-663, a microRNA targeting JunB and JunD. Carcinogenesis 31(9):1561–1566
Tiribuzi R, Crispoltoni L, Porcellati S, Di Lullo M, Florenzano F, Pirro M, Orlacchio A (2014) miR128 up-regulation correlates with impaired amyloid β (1–42) degradation in monocytes from patients with sporadic Alzheimer’s disease. Neurobiol Aging 35(2):345–356
Tognini P, Pizzorusso T (2012) MicroRNA212/132 family: molecular transducer of neuronal function and plasticity. Int J Biochem Cell Biol 44(1):6–10
Tumolo MR, Panico A, De Donno A, Mincarone P, Leo CG, Guarino R, ..., Sabina S (2022) The expression of microRNAs and exposure to environmental contaminants related to human health: a review. Int J Environ Health Res 32(2):332-354
Vannucci SJ, Maher F, Simpson IA (1997) Glucose transporter proteins in brain: delivery of glucose to neurons and glia. Glia 21(1):2–21
Varendi K, Kumar A, Härma MA, Andressoo JO (2014) miR-1, miR-10b, miR-155, and miR-191 are novel regulators of BDNF. Cell Mol Life Sci 71(22):4443–4456
Venkatadri R, Muni T, Iyer AKV, Yakisich JS, Azad N (2016) Role of apoptosis-related miRNAs in resveratrol-induced breast cancer cell death. Cell Death Dis 7(2):e2104–e2104
Vessey JP, Vaccani A, **e Y, Dahm R, Karra D, Kiebler MA, Macchi P (2006) Dendritic localization of the translational repressor Pumilio 2 and its contribution to dendritic stress granules. J Neurosci 26(24):6496–6508
Vishnoi A, Rani S (2017) MiRNA biogenesis and regulation of diseases: an overview. MicroRNA profiling: Methods and protocols 1509: 1–10
Vo N, Klein ME, Varlamova O, Keller DM, Yamamoto T, Goodman RH, Impey S (2005) A cAMP-response element binding protein-induced microRNA regulates neuronal morphogenesis. Proc Natl Acad Sci USA 102(45):16426–16431
Wahid F, Khan T, Kim YY (2014) MicroRNA and diseases: therapeutic potential as new generation of drugs. Biochimie 104:12–26
Wanet A, Tacheny A, Arnould T, Renard P (2012) miR-212/132 expression and functions: within and beyond the neuronal compartment. Nucleic Acids Res 40(11):4742–4753
Wang G, Huang Y, Wang LL, Zhang YF, Xu J, Zhou Y, Chen SD (2016) MicroRNA-146a suppresses ROCK1 allowing hyperphosphorylation of tau in Alzheimer’s disease. Sci R Rep 6(1):1–12
Wang L, Bammler TK, Beyer RP, Gallagher EP (2013) Copper-induced deregulation of microRNA expression in the zebrafish olfactory system. Environ Sci Technol 47(13):7466–7474
Wang Q, Zhan Y, Ren N, Wang Z, Zhang Q, Wu S, Li H (2018) Paraquat and MPTP alter microRNA expression profiles, and downregulated expression of miR-17-5p contributes to PQ-induced dopaminergic neurodegeneration. J Appl Toxicol 38(5):665–677
Wang R, Hu Y, Song G, Hao CJ, Cui Y, **a HF, Ma X (2012) MiR-206 regulates neural cells proliferation and apoptosis via Otx2. Cell Physiol Biochem 29(3–4):381–390
Wang WX, Rajeev BW, Stromberg AJ, Ren N, Tang G, Huang Q, Rigoutsos I, Nelson PT (2008) The expression of microRNA miR-107 decreases early in Alzheimer’s disease and may accelerate disease progression through regulation of beta-site amyloid precursor protein-cleaving enzyme 1. J Neurosci 28(5):1213–1223
Wang W, Zhao F, Ma X, Perry G, Zhu X (2020) Mitochondria dysfunction in the pathogenesis of Alzheimer’s disease: recent advances. Mol Neurodegener 15(1):1–22
Wayman GA, Davare M, Ando H, Fortin D, Varlamova O, Cheng HYM, Impey S (2008) An activity-regulated microRNA controls dendritic plasticity by down-regulating p250GAP. Proc Natl Acad Sci 105(26):9093–9098
Weise CM, Chen K, Chen Y, Kuang X, Savage CR, Reiman EM, Alzheimer’s Disease Neuroimaging Initiative (2018) Left lateralized cerebral glucose metabolism declines in amyloid-β positive persons with mild cognitive impairment. NeuroImage Clin 20:286–296
Weldon Furr J, Morales-Scheihing D, Manwani B, Lee J, McCullough LD (2019) Cerebral amyloid angiopathy, Alzheimer’s disease and microRNA: miRNA as diagnostic biomarkers and potential therapeutic targets. NeuroMol Med 21(4):369–390
Weldon BA, Shubin SP, Smith MN, Workman T, Artemenko A, Griffith WC, ..., Faustman EM (2016) Urinary microRNAs as potential biomarkers of pesticide exposure. Toxicol Appl Pharmacol 312:19–25
Wen MM (2016) Getting miRNA therapeutics into the target cells for neurodegenerative diseases: a mini-review. Front Mol Neurosci 9:129–136
Wijesinghe P, ** J, Cui J, Campbell M, Pham W, Matsubara JA (2023) MicroRNAs in tear fluids predict underlying molecular changes associated with Alzheimer’s disease. Life Sci Alliance 6(6):e202201757
Wong HK, Veremeyko T, Patel N, Lemere CA, Walsh DM, Esau C, Vanderburg C, Krichevsky AM (2013) De-repression of FOXO3a death axis by microRNA-132 and -212 causes neuronal apoptosis in Alzheimer’s disease. Hum Mol Genet 22(15):3077–3092
**n H, Li YI, Buller B, Katakowski M, Zhang Y, Wang X, Chopp M (2012) Exosome-mediated transfer of miR-133b from multipotent mesenchymal stromal cells to neural cells contributes to neurite outgrowth. Stem Cells 30(7):1556–1564
Xu XL, Li Y, Wang F, Gao FB (2008) The steady-state level of the nervous-system-specific microRNA-124a is regulated by dFMR1 in Drosophila. J Neurosci 28(46):11883–11889
Yan X, Zhu Z, Xu S, Yang LN, Liao XH, Zheng M, ..., Liu H (2017) MicroRNA-140–5p inhibits hepatocellular carcinoma by directly targeting the unique isomerase Pin1 to block multiple cancer-driving pathways. Sci Rep 7(1):1–12
Yang G, Song Y, Zhou X, Deng Y, Liu T, Weng G, Pan S (2015) MicroRNA-29c targets β-site amyloid precursor protein-cleaving enzyme 1 and has a neuroprotective role in vitro and in vivo. Mol Med Rep 12(2):3081–3088
Yang TT, Liu CG, Gao SC, Zhang Y, Wang PC (2018) The Serum Exosome Derived MicroRNA-135a, -193b, and -384 Were Potential Alzheimer’s Disease Biomarkers. Biomed Environ Sci 31(2):87–96
Yao K, Yang Q, Li Y, Lan T, Yu H, Yu Y (2020) MicroRNA-9 mediated the protective effect of ferulic acid on hypoxic-ischemic brain damage in neonatal rats. PLoS ONE 15(5):e0228825
Yin Z, Herron S, Silveira S, Kleemann K, Gauthier C, Mallah D, Cheng Y, Margeta MA, Pitts KM, Barry JL, Subramanian A, Shorey H, Brandao W, Durao A, Delpech JC, Madore C, Jedrychowski M, Ajay AK, Murugaiyan G, Hersh SW, …, Butovsky O (2023) Identification of a protective microglial state mediated by miR-155 and interferon-γ signaling in a mouse model of Alzheimer's disease. Nat Neurosci 26(7):1196–1207
Yoon H, Flores LF, Kim J (2016) MicroRNAs in brain cholesterol metabolism and their implications for Alzheimer’s disease. Biochim Biophys Acta (BBA)-Mol Cell Biol Lipids 1861(12):2139–2147
Young DD, Connelly CM, Grohmann C, Deiters A (2010) Small molecule modifiers of microRNA miR-122 function for the treatment of hepatitis C virus infection and hepatocellular carcinoma. J Am Chem Soc 132(23):7976–7981
Yu JY, Chung KH, Deo M, Thompson RC, Turner DL (2008) MicroRNA miR-124 regulates neurite outgrowth during neuronal differentiation. Exp Cell Res 314(14):2618–2633
Yuan Q, Shakhawat AM, Harley CW (2014) Mechanisms underlying early odor preference learning in rats. Prog Brain Res 208:115–156
Yuan XZ, Sun S, Tan CC, Yu JT, Tan L (2017) The role of ADAM10 in Alzheimer’s disease. J Alzheimers Dis 58(2):303–322
Zeng CW, Zhang XJ, Lin KY, Ye H, Feng SY, Zhang H, Chen YQ (2012) Camptothecin induces apoptosis in cancer cells via microRNA-125b-mediated mitochondrial pathways. Mol Pharmacol 81(4):578–586
Zhang L, Fang Y, Zhao X, Zheng Y, Ma Y, Li S, ..., Li L (2021a) miR-204 silencing reduces mitochondrial autophagy and ROS production in a murine AD model via the TRPML1-activated STAT3 pathway. Mol Ther-Nucleic Acids 24:822–831
Zhang L, Zhang Y, Wong SH, Law PT, Zhao S, Yu J, Wu WK (2018) Common deregulation of seven biological processes by MicroRNAs in gastrointestinal cancers. Sci Rep 8(1):1–11
Zhang X, Alshakhshir N, Zhao L (2021b) Glycolytic metabolism, brain resilience, and Alzheimer’s disease. Front Neurosci 15:476
Zhang Y, Liu D, Chen X, Li J, Li L, Bian Z, Zhang CY (2010) Secreted monocytic miR-150 enhances targeted endothelial cell migration. Mol Cell 39(1):133–144
Zhao MW, Yang P, Zhao LL (2019) Chlorpyrifos activates cell pyroptosis and increases susceptibility on oxidative stress-induced toxicity by miR-181/SIRT1/PGC-1α/Nrf2 signaling pathway in human neuroblastoma SH-SY5Y cells. Environ Toxicol 34(6):699–707
Zhao Y, Jaber V, Alexandrov PN, Vergallo A, Lista S, Hampel H, Lukiw WJ (2020) microRNA-based biomarkers in Alzheimer’s disease (AD). Front Neurosci 14:585432
Zhao ZB, Wu L, **ong R, Wang LL, Zhang B, Wang C, Li H, Liang L, Chen SD (2014) MicroRNA-922 promotes tau phosphorylation by downregulating ubiquitin carboxy-terminal hydrolase L1 (UCHL1) expression in the pathogenesis of Alzheimer’s disease. Neuroscience 275:232–237
Zhi Y, Pan J, Shen W, He P, Zheng J, Zhou X, ..., Zhou Z (2016) Ginkgolide B inhibits human bladder cancer cell migration and invasion through microRNA-223–3p. Cell Physiol Biochem 39(5):1787–1794
Zhou R, Wang R, Qin Y, Ji J, Xu M, Wu W, Wang X (2015) Mitochondria-related miR-151a-5p reduces cellular ATP production by targeting CYTB in asthenozoospermia. Sci Rep 5(1):1–10
Zhou XW, Li X, Bjorkdahl C, Sjogren MJ, Alafuzoff I, Soininen H, Grundke-Iqbal I, Iqbal K, Winblad B, Pei JJ (2006) Assessments of the accumulation severities of amyloid beta-protein and hyperphosphorylated tau in the medial temporal cortex of control and Alzheimer’s brains. Neurobiol Dis 22(3):657–668
Zhou Y, Zhang T, Yan Y, You B, You Y, Zhang W, Chen J (2021) MicroRNA-223-3p regulates allergic inflammation by targeting INPP4A. Braz J Otorhinolaryngol 87:591–600
Zhao Y, Zhao R, Wu J, Wang Q, Pang K, Shi Q et al (2018) Melatonin protects against Abeta-induced neurotoxicity in primary neurons via miR-132/PTEN/AKT/FOXO3a pathway. BioFactors 44:609–618
Acknowledgements
H.V., S.K., P.G. A.Y. and R.R. acknowledges financial support in the form of a senior/junior research fellowship (SRF/JRF) from the University Grants Commission (UGC), New Delhi and Dept. of Biotechnology (DBT) and Council for Scientific and Industrial Research (CSIR), Govt. of India, New Delhi, India, respectively. Because of the limited focus of the article, many relevant and appropriate references could not be included, for which the authors apologize.
Funding
A.K.M and M.D. thankfully acknowledges financial support in the form of Core Research Grant (CRG/2021/002524) from SERB, DST, Govt. of India, New Delhi, India.
Author information
Authors and Affiliations
Contributions
AKM and MD conceived the idea and outline of the manuscript. SK and HV (both of them contributed equally) performed literature review, wrote the manuscript and made the figures and tables along with SK, PG, AY, BY, and RR. All authors have read the manuscript and agreed to the final draft of the manuscript.
Corresponding author
Ethics declarations
Ethics approval
Not required.
Consent to participate
Not required.
Consent for publication
Not required.
Conflicts of interest/Competing interests
Authors do not have any conflict of interest to declare.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Kaur, S., Verma, H., Kaur, S. et al. Understanding the multifaceted role of miRNAs in Alzheimer’s disease pathology. Metab Brain Dis 39, 217–237 (2024). https://doi.org/10.1007/s11011-023-01265-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11011-023-01265-9