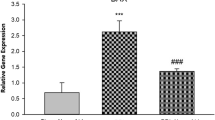
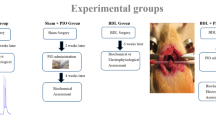
Abstract
Bile secretion is a physiological function that is disrupted following Bile Duct Ligation (BDL) and induces cholestasis. Cholestasis is a bile flow reduction that induces apoptosis, oxidative stress, and inflammation, and alters the expression of genes. Evidence shows the relationship between cholestasis and neuroinflammation. Cholestasis via attenuating mitochondrial biogenesis and anti-oxidant activity can induce neuroinflammation and apoptosis. Mitochondrial transcriptional factor A (TFAM) and Peroxisome proliferator-activated receptor γ coactivator-1α (PGC-1α) are involved in mitochondrial biogenesis, and TFAM, PGC-1α, Catalase (CAT), and Superoxide dismutase (SOD) have a role in upregulating antioxidant pathways. On the other hand, many studies have shown the neuroprotective effects of Crocin, the water-soluble carotenoid of Saffron (Crocus sativus L.). In this study, we aimed to investigate the effect of Crocin on the level of TFAM, PGC-1α, CAT, and SOD following cholestasis-induced neuroinflammation in the rat’s striatum. Cholestasis was induced by BDL surgery and administration of Crocin was intraperitoneal, at the dose of 30 mg/kg every day, 24 h after BDL surgery up to thirty days. The results showed that TFAM, PGC-1α, and SOD were decreased following cholestasis; while, CAT was increased. In addition, Crocin restored the effects of cholestasis on the level of TFAM, PGC-1α, and SOD. In conclusion, Crocin may have improvement effects on cholestasis-induced neuroinflammation in the rat’s striatum.



Similar content being viewed by others
Data availability
Data will be made available on a reasonable request.
References
Abd El Motteleb DM, Ibrahim I, Elshazly SM (2017) Sildenafil protects against bile duct ligation induced hepatic fibrosis in rats: Potential role for silent information regulator 1 (SIRT1). Toxicol Appl Pharmacol 335:64–71. https://doi.org/10.1016/j.taap.2017.09.021
Abe T et al (2004) Kupffer cell-derived interleukin 10 is responsible for impaired bacterial clearance in bile duct-ligated mice. Hepatology 40:414–423. https://doi.org/10.1002/hep.20301
Ahmadi M, Rajaei Z, Hadjzadeh MA, Nemati H, Hosseini M (2017) Crocin improves spatial learning and memory deficits in the Morris water maze via attenuating cortical oxidative damage in diabetic rats. Neurosci Lett 642:1–6. https://doi.org/10.1016/j.neulet.2017.01.049
Aktas C, Kanter M, Erboga M, Mete R, Oran M (2014) Melatonin attenuates oxidative stress, liver damage and hepatocyte apoptosis after bile-duct ligation in rats. Toxicol Ind Health 30:835–844. https://doi.org/10.1177/0748233712464811
Alawdi SH, El-Denshary ES, Safar MM, Eidi H, David MO, Abdel-Wahhab MA (2017) Neuroprotective effect of nanodiamond in alzheimer’s disease rat model: a Pivotal role for modulating NF-kappaB and STAT3 signaling. Mol Neurobiol 54:1906–1918. https://doi.org/10.1007/s12035-016-9762-0
Arduini A et al (2011) Mitochondrial biogenesis fails in secondary biliary cirrhosis in rats leading to mitochondrial DNA depletion and deletions. Am J Physiol Gastrointest Liver Physiol 301:G119-127. https://doi.org/10.1152/ajpgi.00253.2010
Bandegi AR, Rashidy-Pour A, Vafaei AA, Ghadrdoost B (2014) Protective effects of crocus sativus L extract and crocin against chronic-stress induced oxidative damage of brain, liver and kidneys in rats. Adv Pharm Bull 4:493–499. https://doi.org/10.5681/apb.2014.073
Bolhassani A, Khavari A, Bathaie SZ (2014) Saffron and natural carotenoids: Biochemical activities and anti-tumor effects. Biochim Biophys Acta 1845:20–30. https://doi.org/10.1016/j.bbcan.2013.11.001
Bouitbir J et al (2012) Opposite effects of statins on mitochondria of cardiac and skeletal muscles: a “mitohormesis” mechanism involving reactive oxygen species and PGC-1. Eur Heart J 33:1397–1407. https://doi.org/10.1093/eurheartj/ehr224
Boyer JL (2013) Bile Formation and Secretion Compr Physiol 3:1035–1078. https://doi.org/10.1002/cphy.c120027
Broide E et al (2000) Superoxide dismutase activity in children with chronic liver diseases. J Hepatol 32:188–192
Bu X, Wu LuX, Yang L, Xu X, Wang J, Tang J (2017) Role of SIRT1/PGC-1alpha in mitochondrial oxidative stress in autistic spectrum disorder. Neuropsychiatr Dis Treat 13:1633–1645. https://doi.org/10.2147/NDT.S129081
Campbell CT, Kolesar JE, Kaufman BA (2012) Mitochondrial transcription factor A regulates mitochondrial transcription initiation DNA packaging, and genome copy number. Biochim Biophys Acta 1819:921–929. https://doi.org/10.1016/j.bbagrm.2012.03.002
Cerda-Bernad D, Valero-Cases E, Pastor JJ, Frutos MJ (2020) Saffron bioactives crocin, crocetin and safranal: effect on oxidative stress and mechanisms of action. Crit Rev Food Sci Nutr 1–18. https://doi.org/10.1080/10408398.2020.1864279
Chen S et al (2015) Crocin inhibits cell proliferation and enhances cisplatin and pemetrexed chemosensitivity in lung cancer cells. Cancer Res 4:775–783. https://doi.org/10.3978/j.issn.2218-6751.2015.11.03
Chenari S, Safari F, Moradi A (2017) Curcumin enhances liver SIRT3 expression in the rat model of cirrhosis. Iran J Basic Med Sci 20:1306–1311. https://doi.org/10.22038/IJBMS.2017.9609
Coskun P, Wyrembak J, Schriner SE, Chen HW, Marciniack C, Laferla F, Wallace DC (2012) A mitochondrial etiology of alzheimer and parkinson disease. Biochim Biophys Acta 1820:553–564. https://doi.org/10.1016/j.bbagen.2011.08.008
Ebrahimzadeh A, Moghadam SY, Rahimi H, Motaghinejad M, Motevalian M, Safari S, Mesrabadi MA (2019) Crocin acts as a neuroprotective mediator against methylphenidateinduced neurobehavioral and neurochemical sequelae: possible role of the CREB-BDNF signaling pathway. Acta Neurobiol Exp (Wars) 79:352–366
El-Shabrawi MH, Kamal NM, Halawa FA, El-Guindi MA, Sobhy GA (2014) Serum superoxide dismutase activity in acute and chronic paediatric liver diseases. Arab J Gastroenterol 15:72–75. https://doi.org/10.1016/j.ajg.2014.04.002
Elchuri S et al (2005) CuZnSOD deficiency leads to persistent and widespread oxidative damage and hepatocarcinogenesis later in life. Oncogene 24:367–380. https://doi.org/10.1038/sj.onc.1208207
Fahmy SR (2015) Anti-fibrotic effect of Holothuria arenicola extract against bile duct ligation in rats. BMC Complement Altern Med 15:14. https://doi.org/10.1186/s12906-015-0533-7
Fahmy SR, Sayed DA, Soliman AM, Almortada NY, Aal WEA (2019) Protective effect of echinochrome against intrahepatic cholestasis induced by alpha-naphthylisothiocyanate in rats. Braz J Biol. https://doi.org/10.1590/1519-6984.192697
Farina A, Delhaye M, Lescuyer P, Dumonceau JM (2014) Bile proteome in health and disease. Compr Physiol 4:91–108. https://doi.org/10.1002/cphy.c130016
Finley JW, Gao S (2017) A Perspective on Crocus sativus L. (Saffron) constituent crocin: a potent water-soluble antioxidant and potential therapy for alzheimer’s disease. J Agric Food Chem 65:1005–1020. https://doi.org/10.1021/acs.jafc.6b04398
Fridovich I (1995) Superoxide radical and superoxide dismutases. Annu Rev Biochem 64:97–112. https://doi.org/10.1146/annurev.bi.64.070195.000525
Georgiev P, Jochum W, Heinrich S, Jang JH, Nocito A, Dahm F, Clavien PA (2008) Characterization of time-related changes after experimental bile duct ligation. Br J Surg 95:646–656. https://doi.org/10.1002/bjs.6050
Ghonem NS, Assis DN, Boyer JL (2015) Fibrates and Cholestasis Hepatology 62:635–643. https://doi.org/10.1002/hep.27744
Glorieux C, Calderon PB (2017) Catalase, a remarkable enzyme: targeting the oldest antioxidant enzyme to find a new cancer treatment approach. Biol Chem 398:1095–1108. https://doi.org/10.1515/hsz-2017-0131
Halliwell B (2007) Oxidative stress and cancer: have we moved forward? Biochem J 401:1–11. https://doi.org/10.1042/BJ20061131
Herzig S et al (2001) CREB regulates hepatic gluconeogenesis through the coactivator PGC-1. Nature 413:179–183. https://doi.org/10.1038/35093131
Hosseinzadeh H, Sadeghnia HR, Ghaeni FA, Motamedshariaty VS, Mohajeri SA (2012) Effects of saffron (Crocus sativus L.) and its active constituent, crocin, on recognition and spatial memory after chronic cerebral hypoperfusion in rats. Phytother Res 26:381–386. https://doi.org/10.1002/ptr.3566
Ismail NA, Okasha SH, Dhawan A, Abdel-Rahman AO, Shaker OG, Sadik NA (2010) Antioxidant enzyme activities in hepatic tissue from children with chronic cholestatic liver disease Saudi. J Gastroenterol 16:90–94. https://doi.org/10.4103/1319-3767.61234
Javad-Moosavi BZ, Nasehi M, Vaseghi S, Jamaldini SH, Zarrindast MR (2020) Activation and inactivation of nicotinic receptnors in the dorsal hippocampal region restored negative effects of total (TSD) and REM sleep deprivation (RSD) on memory acquisition, locomotor activity and pain perception. Neuroscience 433:200–211. https://doi.org/10.1016/j.neuroscience.2020.03.006
Kim JH, Park GY, Bang SY, Park SY, Bae SK, Kim Y (2014) Crocin suppresses LPS-stimulated expression of inducible nitric oxide synthase by upregulation of heme oxygenase-1 via calcium/calmodulin-dependent protein kinase 4. Mediators Inflamm 2014:728709. https://doi.org/10.1155/2014/728709
Kong LY, Li GP, Yang P, ** Z (2015) Protective effect of thymoquinone on cholestatic rats with liver injury. Genet Mol Res 14:12247–12253. https://doi.org/10.4238/2015.October.9.13
Krishnaswamy VKD, Alugoju P, Periyasamy L (2020) Effect of short-term oral supplementation of crocin on age-related oxidative stress, cholinergic, and mitochondrial dysfunction in rat cerebral cortex. Life Sci 263:118545. https://doi.org/10.1016/j.lfs.2020.118545
Kunkel GH, Chaturvedi P, Thelian N, Nair R, Tyagi SC (2018) Mechanisms of TFAM-mediated cardiomyocyte protection. Can J Physiol Pharmacol 96:173–181. https://doi.org/10.1139/cjpp-2016-0718
Kunkel GH, Kunkel CJ, Ozuna H, Miralda I, Tyagi SC (2019) TFAM overexpression reduces pathological cardiac remodeling. Mol Cell Biochem 454:139–152. https://doi.org/10.1007/s11010-018-3459-9
Lei Y, Wang K, Deng L, Chen Y, Nice EC, Huang C (2015) Redox regulation of inflammation: old elements, a new story. Med Res Rev 35:306–340. https://doi.org/10.1002/med.21330
Lewandowski L, Kepinska M, Milnerowicz H (2018) Inhibition of copper-zinc superoxide dismutase activity by selected environmental xenobiotics. Environ Toxicol Pharmacol 58:105–113. https://doi.org/10.1016/j.etap.2017.12.022
Li K, Li Y, Ma Z, Zhao J (2015) Crocin exerts anti-inflammatory and anti-catabolic effects on rat intervertebral discs by suppressing the activation of JNK. Int J Mol Med 36:1291–1299. https://doi.org/10.3892/ijmm.2015.2359
Linardaki ZI, Orkoula MG, Kokkosis AG, Lamari FN, Margarity M (2013) Investigation of the neuroprotective action of saffron (Crocus sativus L.) in aluminum-exposed adult mice through behavioral and neurobiochemical assessment. Food Chem Toxicol 52:163–170. https://doi.org/10.1016/j.fct.2012.11.016
Liu KZ, Man A, Shaw RA, Liang B, Xu Z, Gong Y (2006) Molecular determination of liver fibrosis by synchrotron infrared microspectroscopy. Biochim Biophys Acta 1758:960–967. https://doi.org/10.1016/j.bbamem.2006.05.006
Liu SG, Wang YM, Zhang YJ, He XJ, Ma T, Song W, Zhang YM (2017) ZL006 protects spinal cord neurons against ischemia-induced oxidative stress through AMPK-PGC-1alpha-Sirt3 pathway. Neurochem Int 108:230–237. https://doi.org/10.1016/j.neuint.2017.04.005
Loew O (1900) A new enzyme of general occurrence in organismis. Science 11:701–702. https://doi.org/10.1126/science.11.279.701
Lu Z et al (2010) PGC-1 alpha regulates expression of myocardial mitochondrial antioxidants and myocardial oxidative stress after chronic systolic overload. Antioxid Redox Signal 13:1011–1022. https://doi.org/10.1089/ars.2009.2940
Ma X et al (2015) Paeonia lactiflora Pall. protects against ANIT-induced cholestasis by activating Nrf2 via PI3K/Akt signaling pathway. Drug Des Devel Ther 9:5061–5074. https://doi.org/10.2147/DDDT.S90030
Mahdavi MS, Nasehi M, Vaseghi S, Mousavi Z, Zarrindast MR (2020) The effect of alpha lipoic acid on passive avoidance and social interaction memory, pain perception, and locomotor activity in REM sleep-deprived rats. Pharmacol Rep. https://doi.org/10.1007/s43440-020-00161-8
Malboosi N, Nasehi M, Hashemi M, Vaseghi S, Zarrindast MR (2020) The neuroprotective effect of NeuroAid on morphine-induced amnesia with respect to the expression of TFAM, PGC-1alpha, DeltafosB and CART genes in the hippocampus of male Wistar rats. Gene 742:144601. https://doi.org/10.1016/j.gene.2020.144601
Margaritis I et al (2020) Effect of crocin on antioxidant gene expression, fibrinolytic parameters, redox status and blood biochemistry in nicotinamide-streptozotocin-induced diabetic rats. J Biol Res (Thessalon) 27:4. https://doi.org/10.1186/s40709-020-00114-5
McCord JM, Fridovich I (1969) Superoxide dismutase. An enzymic function for erythrocuprein (hemocuprein). J Biol Chem 244:6049–6055
Milnerowicz H, Jablonowska M, Bizon A (2009) Change of zinc, copper, and metallothionein concentrations and the copper-zinc superoxide dismutase activity in patients with pancreatitis. Pancreas 38:681–688. https://doi.org/10.1097/MPA.0b013e3181a53d1
Montilla P et al (2001) Melatonin versus vitamin E as protective treatment against oxidative stress after extra-hepatic bile duct ligation in rats. J Pineal Res 31:138–144
Mousavi SH, Tayarani NZ, Parsaee H (2010) Protective effect of saffron extract and crocin on reactive oxygen species-mediated high glucose-induced toxicity in PC12 cells. Cell Mol Neurobiol 30:185–191. https://doi.org/10.1007/s10571-009-9441-z
Nam KN et al (2010) Anti-inflammatory effects of crocin and crocetin in rat brain microglial cells. Eur J Pharmacol 648:110–116. https://doi.org/10.1016/j.ejphar.2010.09.003
Ochiai T et al (2007) Protective effects of carotenoids from saffron on neuronal injury in vitro and in vivo. Biochim Biophys Acta 1770:578–584. https://doi.org/10.1016/j.bbagen.2006.11.012
Ochiai T, Soeda S, Ohno S, Tanaka H, Shoyama Y, Shimeno H (2004) Crocin prevents the death of PC-12 cells through sphingomyelinase-ceramide signaling by increasing glutathione synthesis. Neurochem Int. 44:321–330
Ongwijitwat S, Liang HL, Graboyes EM, Wong-Riley MT (2006) Nuclear respiratory factor 2 senses changing cellular energy demands and its silencing down-regulates cytochrome oxidase and other target gene mRNAs. Gene 374:39–49. https://doi.org/10.1016/j.gene.2006.01.009
Onyango IG, Khan SM, Bennett JP Jr (2017) Mitochondria in the pathophysiology of Alzheimer’s and Parkinson’s diseases. Front Biosci (Landmark Ed) 22:854–872
Ota F, Kizuka Y, Kitazume S, Adachi T, Taniguchi N (2016) N-Glycosylation is essential for the secretion of extracellular superoxide dismutase. FEBS Lett 590:3357–3367. https://doi.org/10.1002/1873-3468.12378
Park YS et al (2016) Eccentric localization of catalase to protect chromosomes from oxidative damages during meiotic maturation in mouse oocytes. Histochem Cell Biol 146:281–288. https://doi.org/10.1007/s00418-016-1446-3
Parola M, Leonarduzzi G, Robino G, Albano E, Poli G, Dianzani MU (1996) On the role of lipid peroxidation in the pathogenesis of liver damage induced by long-standing cholestasis. Free Radic Biol Med 20:351–359
Peng K et al (2017) The interaction of mitochondrial biogenesis and fission/fusion mediated by PGC-1alpha regulates rotenone-induced dopaminergic neurotoxicity. Mol Neurobiol 54:3783–3797. https://doi.org/10.1007/s12035-016-9944-9
Prieto MA, Vazquez JA, Murado MA (2015) Crocin bleaching antioxidant assay revisited: application to microplate to analyse antioxidant and pro-oxidant activities. Food Chem 167:299–310. https://doi.org/10.1016/j.foodchem.2014.06.114
Puigserver P, Spiegelman BM (2003) Peroxisome proliferator-activated receptor-gamma coactivator 1 alpha (PGC-1 alpha): transcriptional coactivator and metabolic regulator. Endocr Rev 24:78–90. https://doi.org/10.1210/er.2002-0012
Qi Y, Chen L, Zhang L, Liu WB, Chen XY, Yang XG (2013) Crocin prevents retinal ischaemia/reperfusion injury-induced apoptosis in retinal ganglion cells through the PI3K/AKT signalling pathway. Exp Eye Res 107:44–51. https://doi.org/10.1016/j.exer.2012.11.011
Rajaei Z, Hosseini M, Alaei H (2016) Effects of crocin on brain oxidative damage and aversive memory in a 6-OHDA model of Parkinson’s disease. Arq Neuropsiquiatr 74:723–729. https://doi.org/10.1590/0004-282X20160131
Rao SV, Hemalatha P, Yetish S, Muralidhara M, Ra**i PS (2019) Prophylactic neuroprotective propensity of Crocin, a carotenoid against rotenone induced neurotoxicity in mice: behavioural and biochemical evidence. Metab Brain Dis 34:1341–1353. https://doi.org/10.1007/s11011-019-00451-y
Rasouri S, Lagouge M, Auwerx J (2007) SIRT1/PGC-1: a neuroprotective axis? Med Sci (Paris) 23:840–844. https://doi.org/10.1051/medsci/20072310840
Salama RM, Abdel-Latif GA, Abbas SS, El Magdoub HM, Schaalan MF (2020) Neuroprotective effect of crocin against rotenone-induced Parkinson’s disease in rats: Interplay between PI3K/Akt/mTOR signaling pathway and enhanced expression of miRNA-7 and miRNA-221. Neuropharmacology 164:107900. https://doi.org/10.1016/j.neuropharm.2019.107900
Salem TA, El-Refaei MF, Badra GA (2003) Study of antioxidant enzymes level and phagocytic activity in chronic liver disease patients. Egypt J Immunol 10:37–45
Samsel A, Seneff S (2015) Glyphosate, pathways to modern diseases III: Manganese, neurological diseases, and associated pathologies. Surg Neurol Int 6:45. https://doi.org/10.4103/2152-7806.153876
Santamaria E et al (2018) The epidermal growth factor receptor ligand amphiregulin protects from cholestatic liver injury and regulates bile acids synthesis. Hepatology. https://doi.org/10.1002/hep.30348
Schriner SE et al (2005) Extension of murine life span by overexpression of catalase targeted to mitochondria. Science 308:1909–1911. https://doi.org/10.1126/science.1106653
Serviddio G et al (2004) Ursodeoxycholic acid protects against secondary biliary cirrhosis in rats by preventing mitochondrial oxidative stress. Hepatology 39:711–720. https://doi.org/10.1002/hep.20101
Shahidani S, Rajaei Z, Alaei H (2019) Pretreatment with crocin along with treadmill exercise ameliorates motor and memory deficits in hemiparkinsonian rats by anti-inflammatory and antioxidant mechanisms. Metab Brain Dis 34:459–468. https://doi.org/10.1007/s11011-018-0379-z
Sies H (2017) Hydrogen peroxide as a central redox signaling molecule in physiological oxidative stress: Oxidative eustress. Redox Biol 11:613–619. https://doi.org/10.1016/j.redox.2016.12.035
Singh I, Gulati S, Orak JK, Singh AK (1993) Expression of antioxidant enzymes in rat kidney during ischemia-reperfusion injury. Mol Cell Biochem 125:97–104
Soeda S, Ochiai T, Paopong L, Tanaka H, Shoyama Y, Shimeno H (2001) Crocin suppresses tumor necrosis factor-alpha-induced cell death of neuronally differentiated PC-12 cells. Life Sci 69:2887–2898
Sun J, Wang J, Zhang N, Yang R, Chen K, Kong D (2019) Identification of global mRNA expression profiles and comprehensive bioinformatic analyses of abnormally expressed genes in cholestatic liver disease. Gene. https://doi.org/10.1016/j.gene.2019.04.078
Tiao MM et al (2011) Dexamethasone decreases cholestatic liver injury via inhibition of intrinsic pathway with simultaneous enhancement of mitochondrial biogenesis. Steroids 76:660–666. https://doi.org/10.1016/j.steroids.2011.03.002
Tiao MM et al (2009) Early transcriptional deregulation of hepatic mitochondrial biogenesis and its consequent effects on murine cholestatic liver injury. Apoptosis 14:890–899. https://doi.org/10.1007/s10495-009-0357-3
Tilg H, Wilmer A, Vogel W, Herold M, Nolchen B, Judmaier G, Huber C (1992) Serum levels of cytokines in chronic liver diseases. Gastroenterology 103:264–274
Tokac M et al (2015) Hepatoprotective and antioxidant effects of lycopene in acute cholestasis . Turk J Med Sci 45:857–864
Valentine JS, Doucette PA, Zittin Potter S (2005) Copper-zinc superoxide dismutase and amyotrophic lateral sclerosis. Annu Rev Biochem 74:563–593. https://doi.org/10.1146/annurev.biochem.72.121801.161647
Vartak N, Damle-Vartak A, Richter B, Dirsch O, Dahmen U, Hammad S, Hengstler JG (2016) Cholestasis-induced adaptive remodeling of interlobular bile ducts. Hepatology 63:951–964. https://doi.org/10.1002/hep.28373
Virbasius JV, Scarpulla RC (1994) Activation of the human mitochondrial transcription factor A gene by nuclear respiratory factors: a potential regulatory link between nuclear and mitochondrial gene expression in organelle biogenesis. Proc Natl Acad Sci U S A 91:1309–1313. https://doi.org/10.1073/pnas.91.4.1309
Wang K (2015) Molecular mechanisms of hepatic apoptosis regulated by nuclear factors. Cell Signal 27:729–738. https://doi.org/10.1016/j.cellsig.2014.11.038
Wang K et al (2015) Neuroprotective effects of crocin against traumatic brain injury in mice: involvement of notch signaling pathway. Neurosci Lett 591:53–58. https://doi.org/10.1016/j.neulet.2015.02.016
Wang P et al (2019) PGC-1alpha/SNAI1 axis regulates tumor growth and metastasis by targeting miR-128b in gastric cancer. J Cell Physiol. https://doi.org/10.1002/jcp.28193
Xu J, Lee G, Wang H, Vierling JM, Maher JJ (2004) Limited role for CXC chemokines in the pathogenesis of alpha-naphthylisothiocyanate-induced liver injury. Am J Physiol Gastrointest Liver Physiol 287:G734-741. https://doi.org/10.1152/ajpgi.00300.2003
Yang X et al (2017) Crocin inhibits oxidative stress and pro-inflammatory response of microglial cells associated with diabetic retinopathy through the activation of PI3K/Akt signaling pathway. J Mol Neurosci 61:581–589. https://doi.org/10.1007/s12031-017-0899-8
Young RP et al (2006) Functional variants of antioxidant genes in smokers with COPD and in those with normal lung function. Thorax 61:394–399. https://doi.org/10.1136/thx.2005.048512
Zelko IN, Mariani TJ, Folz RJ (2002) Superoxide dismutase multigene family: a comparison of the CuZn-SOD (SOD1), Mn-SOD (SOD2), and EC-SOD (SOD3) gene structures, evolution, and expression. Free Radic Biol Med 33:337–349
Zhang R, Wang J (2018) HuR stabilizes TFAM mRNA in an ATM/p38-dependent manner in ionizing irradiated cancer cells. Cancer Sci 109:2446–2457. https://doi.org/10.1111/cas.13657
Zhang X, Zhang X, Dang Z, Su S, Li Z, Lu D (2020) Cognitive protective mechanism of crocin pretreatment in rat submitted to acute high-altitude hypoxia exposure. Biomed Res Int 2020:3409679. https://doi.org/10.1155/2020/3409679
Zhang XY et al (2018) Crocin attenuates acute hypobaric hypoxia-induced cognitive deficits of rats. Eur J Pharmacol 818:300–305. https://doi.org/10.1016/j.ejphar.2017.10.042
Zhao SS et al (2018) D-chiro-inositol effectively attenuates cholestasis in bile duct ligated rats by improving bile acid secretion and attenuating oxidative stress. Acta Pharmacol Sin 39:213–221. https://doi.org/10.1038/aps.2017.98
Author information
Authors and Affiliations
Contributions
MR. Eteghadi conducted the tests. M. Nasehi analyzed data and prepared figures. S. Vaseghi wrote the manuscript. S. Hesami-Tackallou designed the study.
Corresponding author
Ethics declarations
Consent for publish and to participate
Not applicable.
Research involving human participants and/or animals
Our experimental protocol was done in accord with Institute for Cognitive Science Studies (ICSS) guidelines, and with the National Institutes of Health Guide for the Care and Use of Laboratory Animals (NIH publications No. 80–23).
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Eteghadi, MR., Nasehi, M., Vaseghi, S. et al. The effect of Crocin on TFAM and PGC-1α expression and Catalase and Superoxide dismutase activities following cholestasis-induced neuroinflammation in the striatum of male Wistar rats. Metab Brain Dis 36, 1791–1801 (2021). https://doi.org/10.1007/s11011-021-00748-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11011-021-00748-x