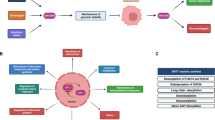
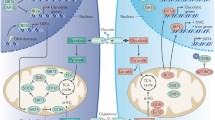
Abstract
Sirtuin 7 (SIRT7) is a member of the sirtuin family and has emerged as a key player in numerous cellular processes. It exhibits various enzymatic activities and is predominantly localized in the nucleolus, playing a role in ribosomal RNA expression, DNA damage repair, stress response and chromatin compaction. Recent studies have revealed its involvement in diseases such as cancer, cardiovascular and bone diseases, and obesity. In cancer, SIRT7 has been found to be overexpressed in multiple types of cancer, including breast cancer, clear cell renal cell carcinoma, lung adenocarcinoma, prostate adenocarcinoma, hepatocellular carcinoma, and gastric cancer, among others. In general, cancer cells exploit SIRT7 to enhance cell growth and metabolism through ribosome biogenesis, adapt to stress conditions and exert epigenetic control over cancer-related genes. The aim of this review is to provide an in-depth understanding of the role of SIRT7 in cancer carcinogenesis, evolution and progression by elucidating the underlying molecular mechanisms. Emphasis is placed on unveiling the intricate molecular pathways through which SIRT7 exerts its effects on cancer cells. In addition, this review discusses the feasibility and challenges associated with the development of drugs that can modulate SIRT7 activity.



Similar content being viewed by others
Data availability
Data sharing is not applicable to this article as no new data were created or analyzed in this study.
References
Dai Y, Faller DV (2008) Transcription Regulation by Class III Histone Deacetylases (HDACs)—Sirtuins. Transl Oncogenomics 1:53–65. https://doi.org/10.4137/TOG.S483
Lagunas-Rangel FA (2022) SIRT7 in the aging process. Cell Mol Life Sci 79:297. https://doi.org/10.1007/s00018-022-04342-x
Kiran S, Chatterjee N, Singh S et al (2013) Intracellular distribution of human SIRT7 and map** of the nuclear/nucleolar localization signal. FEBS J 280:3451–3466. https://doi.org/10.1111/febs.12346
Michishita E, Park JY, Burneskis JM et al (2005) Evolutionarily Conserved and Nonconserved Cellular Localizations and Functions of Human SIRT Proteins. Mol Biol Cell 16:4623–4635. https://doi.org/10.1091/mbc.e05-01-0033
Barber MF, Michishita-Kioi E, ** Y et al (2012) SIRT7 links H3K18 deacetylation to maintenance of oncogenic transformation. Nature 487:114–118. https://doi.org/10.1038/nature11043
Simonet NG, Thackray JK, Vazquez BN et al (2020) SirT7 auto-ADP-ribosylation regulates glucose starvation response through mH2A1. Sci Adv 6:1–17. https://doi.org/10.1126/sciadv.aaz2590
Li L, Shi L, Yang S et al (2016) SIRT7 is a histone desuccinylase that functionally links to chromatin compaction and genome stability. Nat Commun 7:12235. https://doi.org/10.1038/ncomms12235
Tanabe K, Liu J, Kato D et al (2018) LC–MS/MS-based quantitative study of the acyl group- and site-selectivity of human sirtuins to acylated nucleosomes. Sci Rep 8:2656. https://doi.org/10.1038/s41598-018-21060-2
Tong Z, Wang M, Wang Y et al (2017) SIRT7 Is an RNA-Activated Protein Lysine Deacylase. ACS Chem Biol 12:300–310. https://doi.org/10.1021/acschembio.6b00954
Yu AQ, Wang J, Jiang ST et al (2021) SIRT7-Induced PHF5A Decrotonylation Regulates Aging Progress Through Alternative Splicing-Mediated Downregulation of CDK2. Front Cell Dev Biol 9:1–15. https://doi.org/10.3389/fcell.2021.710479
Tong Z, Wang Y, Zhang X et al (2016) SIRT7 Is Activated by DNA and Deacetylates Histone H3 in the Chromatin Context. ACS Chem Biol 11:742–747. https://doi.org/10.1021/acschembio.5b01084
Lagunas-Rangel FA (2019) Current role of mammalian sirtuins in DNA repair. DNA Repair (Amst) 80:85–92. https://doi.org/10.1016/j.dnarep.2019.06.009
Lagunas-Rangel FA (2023) Role of SIRT5 in cancer. Friend or Foe? Biochimie 209:131–141. https://doi.org/10.1016/j.biochi.2023.02.008
Lin H (2018) The Enzymatic Activities of Sirtuins. In: Introductory Review on Sirtuins in Biology, Aging, and Disease. Elsevier, pp 45–62
Imai S, Guarente L (2016) It takes two to tango: NAD+ and sirtuins in aging/longevity control. npj Aging Mech Dis 2:16017. https://doi.org/10.1038/npjamd.2016.17
Tsai Y-C, Greco TM, Cristea IM (2014) Sirtuin 7 Plays a Role in Ribosome Biogenesis and Protein Synthesis. Mol Cell Proteomics 13:73–83. https://doi.org/10.1074/mcp.M113.031377
Vazquez BN, Thackray JK, Serrano L (2017) Sirtuins and DNA damage repair: SIRT7 comes to play. Nucleus 8:107–115. https://doi.org/10.1080/19491034.2016.1264552
Chistiakov DA, Sobenin IA, Revin VV et al (2014) Mitochondrial Aging and Age-Related Dysfunction of Mitochondria. Biomed Res Int 2014:1–7. https://doi.org/10.1155/2014/238463
Liu Z, Qian M, Tang X et al (2019) SIRT7 couples light-driven body temperature cues to hepatic circadian phase coherence and gluconeogenesis. Nat Metab 1:1141–1156. https://doi.org/10.1038/s42255-019-0136-6
Wu D, Li Y, Zhu KS et al (2018) Advances in Cellular Characterization of the Sirtuin Isoform, SIRT7. Front Endocrinol (Lausanne) 9:1–13. https://doi.org/10.3389/fendo.2018.00652
Voelter-Mahlknecht S, Letzel S, Mahlknecht U (2006) Fluorescence in situ hybridization and chromosomal organization of the human Sirtuin 7 gene. Int J Oncol 28:899–908. https://doi.org/10.3892/ijo.28.4.899
Varadi M, Anyango S, Deshpande M et al (2022) AlphaFold Protein Structure Database: massively expanding the structural coverage of protein-sequence space with high-accuracy models. Nucleic Acids Res 50:D439–D444. https://doi.org/10.1093/nar/gkab1061
Bateman A, Martin M-J, Orchard S et al (2021) UniProt: the universal protein knowledgebase in 2021. Nucleic Acids Res 49:D480–D489. https://doi.org/10.1093/nar/gkaa1100
Priyanka A, Solanki V, Parkesh R, Thakur KG (2016) Crystal structure of the N-terminal domain of human SIRT7 reveals a three-helical domain architecture. Proteins Struct Funct Bioinforma 84:1558–1563. https://doi.org/10.1002/prot.25085
Sanders BD, Jackson B, Marmorstein R (2010) Structural basis for sirtuin function: What we know and what we don’t. Biochim Biophys Acta - Proteins Proteomics 1804:1604–1616. https://doi.org/10.1016/j.bbapap.2009.09.009
Tsai Y-C, Greco TM, Boonmee A et al (2012) Functional Proteomics Establishes the Interaction of SIRT7 with Chromatin Remodeling Complexes and Expands Its Role in Regulation of RNA Polymerase I Transcription. Mol Cell Proteomics 11:60–76. https://doi.org/10.1074/mcp.A111.015156
Grob A, Roussel P, Wright JE et al (2009) Involvement of SIRT7 in resumption of rDNA transcription at the exit from mitosis. J Cell Sci 122:489–498. https://doi.org/10.1242/jcs.042382
Lagunas-Rangel FA (2023) Bioinformatic analysis of SIRT7 sequence and structure. J Biomol Struct Dyn 41:8081–8091. https://doi.org/10.1080/07391102.2022.2126890
Sun M, Qi S, Wu M et al (2022) Calreticulin as a prognostic biomarker and correlated with immune infiltrate in kidney renal clear cell carcinoma. Front Genet 13:3167–3182. https://doi.org/10.3389/fgene.2022.909556
Huo Q, Li Z, Cheng L, et al (2020) SIRT7 Is a Prognostic Biomarker Associated With Immune Infiltration in Luminal Breast Cancer. Front Oncol 10:. https://doi.org/10.3389/fonc.2020.00621
Tan Y, Li B, Peng F et al (2020) Integrative Analysis of Sirtuins and Their Prognostic Significance in Clear Cell Renal Cell Carcinoma. Front Oncol 10:1–10. https://doi.org/10.3389/fonc.2020.00218
Lagunas-Rangel FA (2021) KDM6B (JMJD3) and its dual role in cancer. Biochimie 184:63–71. https://doi.org/10.1016/j.biochi.2021.02.005
Skorupska A, Lenda R, Ożyhar A, Bystranowska D (2021) The Multifaceted Nature of Nucleobindin-2 in Carcinogenesis. Int J Mol Sci 22:5687. https://doi.org/10.3390/ijms22115687
Zhang L, Kim S, Ren X (2020) The Clinical Significance of SIRT2 in Malignancies: A Tumor Suppressor or an Oncogene? Front Oncol 10:. https://doi.org/10.3389/fonc.2020.01721
Navas LE, Carnero A (2021) NAD+ metabolism, stemness, the immune response, and cancer. Signal Transduct Target Ther 6:2. https://doi.org/10.1038/s41392-020-00354-w
Chen K, Li L, Yang F et al (2018) SIRT7 depletion inhibits cell proliferation, migration, and increases drug sensitivity by activating p38MAPK in breast cancer cells. J Cell Physiol 233:6767–6778. https://doi.org/10.1002/jcp.26398
Dong L, Yu L, Li H, et al (2020) An NAD+-Dependent Deacetylase SIRT7 Promotes HCC Development Through Deacetylation of USP39. iScience 23:101351. https://doi.org/10.1016/j.isci.2020.101351
Kim JK, Noh JH, Jung KH et al (2013) Sirtuin7 oncogenic potential in human hepatocellular carcinoma and its regulation by the tumor suppressors MiR-125a-5p and MiR-125b. Hepatology 57:1055–1067. https://doi.org/10.1002/hep.26101
Wu F, Xu L, Tu Y et al (2022) Sirtuin 7 super-enhancer drives epigenomic reprogramming in hepatocarcinogenesis. Cancer Lett 525:115–130. https://doi.org/10.1016/j.canlet.2021.10.039
Zhao J, Wozniak A, Adams A et al (2019) SIRT7 regulates hepatocellular carcinoma response to therapy by altering the p53-dependent cell death pathway. J Exp Clin Cancer Res 38:252. https://doi.org/10.1186/s13046-019-1246-4
Peng L-S, Duan S-L, Li R-Q et al (2023) Prognostic value of sirtuin family members and experimental verification identify SIRT5 as diagnostic biomarkers in clear cell renal cell carcinoma. PeerJ 11:e15154. https://doi.org/10.7717/peerj.15154
**ang J, Zhang N, Sun H et al (2020) Disruption of SIRT7 Increases the Efficacy of Checkpoint Inhibitor via MEF2D Regulation of Programmed Cell Death 1 Ligand 1 in Hepatocellular Carcinoma Cells. Gastroenterology 158:664-678.e24. https://doi.org/10.1053/j.gastro.2019.10.025
Yanai M, Kurata M, Muto Y et al (2020) Clinicopathological and molecular analysis of SIRT7 in hepatocellular carcinoma. Pathology 52:529–537. https://doi.org/10.1016/j.pathol.2020.03.011
Cheng Z, Li X, Hou S et al (2019) K-Ras-ERK1/2 accelerates lung cancer cell development via mediating H3 K18ac through the MDM2-GCN5-SIRT7 axis. Pharm Biol 57:701–709. https://doi.org/10.1080/13880209.2019.1672756
Jiang Y, Han Z, Wang Y, Hao W (2018) Depletion of SIRT7 sensitizes human non-small cell lung cancer cells to gemcitabine therapy by inhibiting autophagy. Biochem Biophys Res Commun 506:266–271. https://doi.org/10.1016/j.bbrc.2018.10.089
Liu X, Chen J, Long X et al (2022) RSL1D1 promotes the progression of colorectal cancer through RAN-mediated autophagy suppression. Cell Death Dis 13:43. https://doi.org/10.1038/s41419-021-04492-z
Yu H, Ye W, Wu J et al (2014) Overexpression of Sirt7 Exhibits Oncogenic Property and Serves as a Prognostic Factor in Colorectal Cancer. Clin Cancer Res 20:3434–3445. https://doi.org/10.1158/1078-0432.CCR-13-2952
Ding M, Jiang C-Y, Zhang Y et al (2020) SIRT7 depletion inhibits cell proliferation and androgen-induced autophagy by suppressing the AR signaling in prostate cancer. J Exp Clin Cancer Res 39:28. https://doi.org/10.1186/s13046-019-1516-1
Yi X, Wang H, Yang Y et al (2023) SIRT7 orchestrates melanoma progression by simultaneously promoting cell survival and immune evasion via UPR activation. Signal Transduct Target Ther 8:107. https://doi.org/10.1038/s41392-023-01314-w
Wei W, **g ZX, Ke Z, Yi P (2017) Sirtuin 7 plays an oncogenic role in human osteosarcoma via downregulating CDC4 expression. Am J Cancer Res 7:1788–1803
Lai C-C, Lin P-M, Lin S-F et al (2013) Altered expression of SIRT gene family in head and neck squamous cell carcinoma. Tumor Biol 34:1847–1854. https://doi.org/10.1007/s13277-013-0726-y
Lu W, Che X, Qu X et al (2021) Succinylation Regulators Promote Clear Cell Renal Cell Carcinoma by Immune Regulation and RNA N6-Methyladenosine Methylation. Front Cell Dev Biol 9:1–17. https://doi.org/10.3389/fcell.2021.622198
He X, Li Y, Chen Q et al (2022) O-GlcNAcylation and stablization of SIRT7 promote pancreatic cancer progression by blocking the SIRT7-REGγ interaction. Cell Death Differ 29:1970–1981. https://doi.org/10.1038/s41418-022-00984-3
Mu P, Liu K, Lin Q et al (2018) Sirtuin 7 promotes glioma proliferation and invasion through activation of the ERK/STAT3 signaling pathway. Oncol Lett 17:1445–1452. https://doi.org/10.3892/ol.2018.9800
Reed MR, Maddukuri L, Ketkar A et al (2021) Inhibition of tryptophan 2,3-dioxygenase impairs DNA damage tolerance and repair in glioma cells. NAR Cancer 3:1–20. https://doi.org/10.1093/narcan/zcab014
Li H, Tian Z, Qu Y et al (2019) SIRT7 promotes thyroid tumorigenesis through phosphorylation and activation of Akt and p70S6K1 via DBC1/SIRT1 axis. Oncogene 38:345–359. https://doi.org/10.1038/s41388-018-0434-6
Blank MF, Grummt I (2017) The seven faces of SIRT7. Transcription 8:67–74. https://doi.org/10.1080/21541264.2016.1276658
Lee Y, Shin MH, Kim M-K et al (2021) Increased Histone Acetylation and Decreased Expression of Specific Histone Deacetylases in Ultraviolet-Irradiated and Intrinsically Aged Human Skin In Vivo. Int J Mol Sci 22:2032. https://doi.org/10.3390/ijms22042032
Chen S, Seiler J, Santiago-Reichelt M et al (2013) Repression of RNA Polymerase I upon Stress Is Caused by Inhibition of RNA-Dependent Deacetylation of PAF53 by SIRT7. Mol Cell 52:303–313. https://doi.org/10.1016/j.molcel.2013.10.010
Iyer-Bierhoff A, Krogh N, Tessarz P et al (2018) SIRT7-Dependent Deacetylation of Fibrillarin Controls Histone H2A Methylation and rRNA Synthesis during the Cell Cycle. Cell Rep 25:2946-2954.e5. https://doi.org/10.1016/j.celrep.2018.11.051
Chen S, Blank MF, Iyer A et al (2016) SIRT7-dependent deacetylation of the U3–55k protein controls pre-rRNA processing. Nat Commun 7:10734. https://doi.org/10.1038/ncomms10734
Ryu D, Jo YS, Lo Sasso G et al (2014) A SIRT7-Dependent Acetylation Switch of GABPβ1 Controls Mitochondrial Function. Cell Metab 20:856–869. https://doi.org/10.1016/j.cmet.2014.08.001
Mohrin M, Shin J, Liu Y, et al (2015) A mitochondrial UPR-mediated metabolic checkpoint regulates hematopoietic stem cell aging. Science (80- ) 347:1374–1377. https://doi.org/10.1126/science.aaa2361
Mohrin M, Widjaja A, Liu Y, et al (2018) The mitochondrial unfolded protein response is activated upon hematopoietic stem cell exit from quiescence. Aging Cell 17:. https://doi.org/10.1111/acel.12756
Lagunas-Rangel FA (2020) Circular RNAs and their participation in stemness of cancer. Med Oncol 37:42. https://doi.org/10.1007/s12032-020-01373-x
Peiris-Pagès M, Martinez-Outschoorn UE, Pestell RG et al (2016) Cancer stem cell metabolism. Breast Cancer Res 18:55. https://doi.org/10.1186/s13058-016-0712-6
Liu H, Hu L, Yu G et al (2021) LncRNA, PLXDC2-OT Promoted the Osteogenesis Potentials of MSCs by Inhibiting the Deacetylation Function of RBM6/SIRT7 Complex and OSX Specific Isoform. Stem Cells 39:1049–1066. https://doi.org/10.1002/stem.3362
Vazquez BN, Thackray JK, Simonet NG, et al (2016) SIRT7 promotes genome integrity and modulates non-homologous end joining DNA repair. EMBO J 35:1488–503. https://doi.org/10.15252/embj.201593499
Tang M, Li Z, Zhang C, et al (2019) SIRT7-mediated ATM deacetylation is essential for its deactivation and DNA damage repair. Sci Adv 5:eaav1118. https://doi.org/10.1126/sciadv.aav1118
Paredes S, Angulo-Ibanez M, Tasselli L et al (2018) The epigenetic regulator SIRT7 guards against mammalian cellular senescence induced by ribosomal DNA instability. J Biol Chem 293:11242–11250. https://doi.org/10.1074/jbc.AC118.003325
Burgess RC, Burman B, Kruhlak MJ, Misteli T (2014) Activation of DNA Damage Response Signaling by Condensed Chromatin. Cell Rep 9:1703–1717. https://doi.org/10.1016/j.celrep.2014.10.060
Zhang P-Y, Li G, Deng Z-J et al (2016) Dicer interacts with SIRT7 and regulates H3K18 deacetylation in response to DNA damaging agents. Nucleic Acids Res 44:3629–3642. https://doi.org/10.1093/nar/gkv1504
Zhao X, Zhang M, Wang J et al (2022) NMN ameliorated radiation induced damage in NRF2-deficient cell and mice via regulating SIRT6 and SIRT7. Free Radic Biol Med 193:342–353. https://doi.org/10.1016/j.freeradbiomed.2022.10.267
Bai W, Cheng L, **ong L et al (2022) Protein succinylation associated with the progress of hepatocellular carcinoma. J Cell Mol Med 26:5702–5712. https://doi.org/10.1111/jcmm.17507
Ru B, Sun J, Kang Q et al (2019) A framework for identifying dysregulated chromatin regulators as master regulators in human cancer. Bioinformatics 35:1805–1812. https://doi.org/10.1093/bioinformatics/bty836
Malik S, Villanova L, Tanaka S et al (2015) SIRT7 inactivation reverses metastatic phenotypes in epithelial and mesenchymal tumors. Sci Rep 5:9841. https://doi.org/10.1038/srep09841
Sung H, Ferlay J, Siegel RL et al (2021) Global Cancer Statistics 2020: GLOBOCAN Estimates of Incidence and Mortality Worldwide for 36 Cancers in 185 Countries. CA Cancer J Clin 71:209–249. https://doi.org/10.3322/caac.21660
Yang Y, Wang Z, He M et al (2023) NAD+ biosynthesis metabolism predicts prognosis and indicates immune microenvironment for breast cancer. Pathol Oncol Res 29:1–12. https://doi.org/10.3389/pore.2023.1610956
Li D, Li L (2017) MicroRNA-3666 inhibits breast cancer cell proliferation by targeting sirtuin 7. Mol Med Rep 16:8493–8500. https://doi.org/10.3892/mmr.2017.7603
Su Y, Wu C, Chang Y et al (2022) USP17L2-SIRT7 axis regulates DNA damage repair and chemoresistance in breast cancer cells. Breast Cancer Res Treat 196:31–44. https://doi.org/10.1007/s10549-022-06711-3
Tang X, Li G, Shi L et al (2021) Combined intermittent fasting and ERK inhibition enhance the anti-tumor effects of chemotherapy via the GSK3β-SIRT7 axis. Nat Commun 12:5058. https://doi.org/10.1038/s41467-021-25274-3
Aljada A, Saleh AM, Alkathiri M, et al (2015) Altered Sirtuin 7 Expression is Associated with Early Stage Breast Cancer. Breast Cancer Basic Clin Res 9:BCBCR.S23156. https://doi.org/10.4137/BCBCR.S23156
Tang X, Shi L, **e N et al (2017) SIRT7 antagonizes TGF-β signaling and inhibits breast cancer metastasis. Nat Commun 8:318. https://doi.org/10.1038/s41467-017-00396-9
Yang F, Hu Y, Shao L et al (2021) SIRT7 interacts with TEK (TIE2) to promote adriamycin induced metastasis in breast cancer. Cell Oncol 44:1405–1424. https://doi.org/10.1007/s13402-021-00649-2
Huo Q, Chen S, Zhuang J et al (2023) SIRT7 Downregulation Promotes Breast Cancer Metastasis Via LAP2α-Induced Chromosomal Instability. Int J Biol Sci 19:1528–1542. https://doi.org/10.7150/ijbs.75340
Forner A, Reig M, Bruix J (2018) Hepatocellular carcinoma. Lancet 391:1301–1314. https://doi.org/10.1016/S0140-6736(18)30010-2
Liu M, Yu J, ** H et al (2021) Bioinformatics Analysis of the SIRT Family Members and Assessment of Their Potential Clinical Value. Onco Targets Ther 14:2635–2649. https://doi.org/10.2147/OTT.S298616
Zhang C, Zhao J, Zhao J et al (2022) CYP2E1-dependent upregulation of SIRT7 is response to alcohol mediated metastasis in hepatocellular carcinoma. Cancer Gene Ther 29:1961–1974. https://doi.org/10.1038/s41417-022-00512-y
Coppola N, de Stefano G, Panella M, et al (2017) Lowered expression of microRNA-125a-5p in human hepatocellular carcinoma and up-regulation of its oncogenic targets sirtuin-7, matrix metalloproteinase-11, and c-Raf. Oncotarget 8:25289–25299. https://doi.org/10.18632/oncotarget.15809
Zhao L, Wang W (2015) miR-125b suppresses the proliferation of hepatocellular carcinoma cells by targeting Sirtuin7. Int J Clin Exp Med 8:18469–18475
Li Y, Shi H, Yuan J et al (2020) Downregulation of circular RNA circPVT1 restricts cell growth of hepatocellular carcinoma through downregulation of Sirtuin 7 via microRNA-3666. Clin Exp Pharmacol Physiol 47:1291–1300. https://doi.org/10.1111/1440-1681.13273
Hu Z, Zhao Y, Mang Y et al (2023) MiR-21-5p promotes sorafenib resistance and hepatocellular carcinoma progression by regulating SIRT7 ubiquitination through USP24. Life Sci 325:121773. https://doi.org/10.1016/j.lfs.2023.121773
Pandey V, Kumar V (2015) Stabilization of SIRT7 deacetylase by viral oncoprotein HBx leads to inhibition of growth restrictive RPS7 gene and facilitates cellular transformation. Sci Rep 5:14806. https://doi.org/10.1038/srep14806
Pang N, Hu Q, Zhou Y et al (2023) Nicotinamide Adenine Dinucleotide Precursor Suppresses Hepatocellular Cancer Progression in Mice. Nutrients 15:1447. https://doi.org/10.3390/nu15061447
Guo X, Tan S, Wang T et al (2022) NAD + salvage governs mitochondrial metabolism, invigorating natural killer cell antitumor immunity. Hepatology n/a-n/a. https://doi.org/10.1002/hep.32658
Wang Y, Zhu Q, Hu H et al (2021) Upregulation of histone acetylation reverses organic anion transporter 2 repression and enhances 5-fluorouracil sensitivity in hepatocellular carcinoma. Biochem Pharmacol 188:114546. https://doi.org/10.1016/j.bcp.2021.114546
Gong J, Wang H, Lou W et al (2018) Associations of sirtuins with clinicopathological parameters and prognosis in non–small cell lung cancer. Cancer Manag Res 10:3341–3356. https://doi.org/10.2147/CMAR.S166946
Shi H, Ji Y, Zhang D et al (2016) MicroRNA-3666-induced suppression of SIRT7 inhibits the growth of non-small cell lung cancer cells. Oncol Rep 36:3051–3057. https://doi.org/10.3892/or.2016.5063
Sun C, Zeng X, Guo H et al (2019) MicroRNA-125a-5p modulates radioresistance in LTEP-a2 non-small cell lung cancer cells by targeting SIRT7. Cancer Biomark 27:39–49. https://doi.org/10.3233/CBM-190381
Baidoun F, Elshiwy K, Elkeraie Y et al (2021) Colorectal Cancer Epidemiology: Recent Trends and Impact on Outcomes. Curr Drug Targets 22:998–1009. https://doi.org/10.2174/1389450121999201117115717
Tang M, Lu X, Zhang C et al (2017) Downregulation of SIRT7 by 5-fluorouracil induces radiosensitivity in human colorectal cancer. Theranostics 7:1346–1359. https://doi.org/10.7150/thno.18804
Wang D, Wei X, Chen X et al (2021) GRIM-19 inhibits proliferation and induces apoptosis in a p53-dependent manner in colorectal cancer cells through the SIRT7/PCAF/MDM2 axis. Exp Cell Res 407:112799. https://doi.org/10.1016/j.yexcr.2021.112799
Haider R, Massa F, Kaminski L, et al (2017) Sirtuin 7: a new marker of aggressiveness in prostate cancer. Oncotarget 8:77309–77316. https://doi.org/10.18632/oncotarget.20468
Chmielewski JP, Bowlby SC, Wheeler FB et al (2018) CD38 Inhibits Prostate Cancer Metabolism and Proliferation by Reducing Cellular NAD+ Pools. Mol Cancer Res 16:1687–1700. https://doi.org/10.1158/1541-7786.MCR-17-0526
Mottahedeh J, Haffner MC, Grogan TR et al (2018) CD38 is methylated in prostate cancer and regulates extracellular NAD+. Cancer Metab 6:13. https://doi.org/10.1186/s40170-018-0186-3
Liu Q, Cui W, Yang C, Du L-P (2021) Circular RNA ZNF609 drives tumor progression by regulating the miR-138–5p/SIRT7 axis in melanoma. Aging (Albany NY) 13:19822–19834. https://doi.org/10.18632/aging.203394
Sun R, Guo M, Fan X et al (2020) MicroRNA-148b Inhibits the Malignant Biological Behavior of Melanoma by Reducing Sirtuin 7 Expression Levels. Biomed Res Int 2020:1–13. https://doi.org/10.1155/2020/9568976
Gill J, Gorlick R (2021) Advancing therapy for osteosarcoma. Nat Rev Clin Oncol 18:609–624. https://doi.org/10.1038/s41571-021-00519-8
Dang Y, Zhou Y, Ou X et al (2021) lncRNA AC007207.2 Promotes Malignant Properties of Osteosarcoma via the miR-1306-5p/SIRT7 Axis. Cancer Manag Res 13:7277–7288. https://doi.org/10.2147/CMAR.S318975
Yu W, Cui X, Wan Z et al (2018) Silencing forkhead box M1 promotes apoptosis and autophagy through SIRT7/mTOR/IGF2 pathway in gastric cancer cells. J Cell Biochem 119:9090–9098. https://doi.org/10.1002/jcb.27168
Hedayati MA, Khani D, Sheikhesmaeili F (2022) Sirt 3, 6, and 7 Genes Expression in Gastric Antral Epithelial Cells of Patients with Helicobacter pylori Infection. Curr Microbiol 79:114. https://doi.org/10.1007/s00284-022-02775-y
Shen X, Li P, Xu Y, et al (2017) Association of sirtuins with clinicopathological parameters and overall survival in gastric cancer. Oncotarget 8:74359–74370. https://doi.org/10.18632/oncotarget.20799
Linehan WM, Ricketts CJ (2019) The Cancer Genome Atlas of renal cell carcinoma: findings and clinical implications. Nat Rev Urol 16:539–552. https://doi.org/10.1038/s41585-019-0211-5
Qi H, Shi X, Yu M et al (2018) Sirtuin 7–mediated deacetylation of WD repeat domain 77 (WDR77) suppresses cancer cell growth by reducing WDR77/PRMT5 transmethylase complex activity. J Biol Chem 293:17769–17779. https://doi.org/10.1074/jbc.RA118.003629
Johnson DE, Burtness B, Leemans CR et al (2020) Head and neck squamous cell carcinoma. Nat Rev Dis Prim 6:92. https://doi.org/10.1038/s41572-020-00224-3
Miao P, Sheng S, Sun X et al (2013) Lactate dehydrogenase a in cancer: A promising target for diagnosis and therapy. IUBMB Life 65:904–910. https://doi.org/10.1002/iub.1216
Li W, Zhu D, Qin S (2018) SIRT7 suppresses the epithelial-to-mesenchymal transition in oral squamous cell carcinoma metastasis by promoting SMAD4 deacetylation. J Exp Clin Cancer Res 37:148. https://doi.org/10.1186/s13046-018-0819-y
Tang X, Li G, Su F et al (2020) HDAC8 cooperates with SMAD3/4 complex to suppress SIRT7 and promote cell survival and migration. Nucleic Acids Res 48:2912–2923. https://doi.org/10.1093/nar/gkaa039
Dong Z, Yang L, Lu J et al (2022) Downregulation of LINC00886 facilitates epithelial–mesenchymal transition through SIRT7/ELF3/miR-144 pathway in esophageal squamous cell carcinoma. Clin Exp Metastasis 39:661–677. https://doi.org/10.1007/s10585-022-10171-w
Rizvi S, Khan SA, Hallemeier CL et al (2018) Cholangiocarcinoma — evolving concepts and therapeutic strategies. Nat Rev Clin Oncol 15:95–111. https://doi.org/10.1038/nrclinonc.2017.157
Li W, Sun Z, Chen C et al (2018) Sirtuin7 has an oncogenic potential via promoting the growth of cholangiocarcinoma cells. Biomed Pharmacother 100:257–266. https://doi.org/10.1016/j.biopha.2018.02.007
Dhillon J, Betancourt M (2020) Pancreatic Ductal Adenocarcinoma. pp 74–91
Ostrom QT, Bauchet L, Davis FG et al (2014) The epidemiology of glioma in adults: a “state of the science” review. Neuro Oncol 16:896–913. https://doi.org/10.1093/neuonc/nou087
Monteiro-Reis S, Lameirinhas A, Miranda-Gonçalves V et al (2020) Sirtuins’ Deregulation in Bladder Cancer: SIRT7 Is Implicated in Tumor Progression through Epithelial to Mesenchymal Transition Promotion. Cancers (Basel) 12:1066. https://doi.org/10.3390/cancers12051066
Han Y, Liu Y, Zhang H et al (2013) Hsa-miR-125b suppresses bladder cancer development by down-regulating oncogene SIRT7 and oncogenic long non-coding RNA MALAT1. FEBS Lett 587:3875–3882. https://doi.org/10.1016/j.febslet.2013.10.023
Villalba JM, Alcaín FJ (2012) Sirtuin activators and inhibitors. BioFactors 38:349–359. https://doi.org/10.1002/biof.1032
Dai H, Sinclair DA, Ellis JL, Steegborn C (2018) Sirtuin activators and inhibitors: Promises, achievements, and challenges. Pharmacol Ther 188:140–154. https://doi.org/10.1016/j.pharmthera.2018.03.004
Lagunas-Rangel FA (2023) Prediction of resveratrol target proteins: a bioinformatics analysis. J Biomol Struct Dyn 0:1–10. https://doi.org/10.1080/07391102.2023.2196698
Li S, Wu B, Zheng W (2019) Cyclic tripeptide-based potent human SIRT7 inhibitors. Bioorg Med Chem Lett 29:461–465. https://doi.org/10.1016/j.bmcl.2018.12.023
Zhang C, Li Y, Liu B et al (2022) Discovery of SIRT7 Inhibitor as New Therapeutic Options Against Liver Cancer. Front Cell Dev Biol 9:1–14. https://doi.org/10.3389/fcell.2021.813233
Zhang C, Zhong W, Cao Y et al (2023) Sorafenib/2800Z Co-Loaded into Cholesterol and PEG Grafted Polylysine NPs for Liver Cancer Treatment. Pharmaceuticals 16:119. https://doi.org/10.3390/ph16010119
Kim J-H, Kim D, Cho SJ et al (2019) Identification of a novel SIRT7 inhibitor as anticancer drug candidate. Biochem Biophys Res Commun 508:451–457. https://doi.org/10.1016/j.bbrc.2018.11.120
Guo X, Chen R, Cao L (2023) Molecular docking-based virtual screening and dynamics simulation study of novel and potential SIRT7 inhibitors. Chem Biol Drug Des. https://doi.org/10.1111/cbdd.14277
Acknowledgements
No acknowledgments
Funding
This research did not receive any specific grant from funding agencies in the public, commercial, or not‐for‐profit sectors.
Author information
Authors and Affiliations
Contributions
FALR: Conceptualized, researched, analyzed, drafted, revised and edited the manuscript.
Corresponding author
Ethics declarations
Competing interests
The author declares no conflict of interest.
Ethical approval
Not applicable.
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Lagunas-Rangel, F.A. The dark side of SIRT7. Mol Cell Biochem (2023). https://doi.org/10.1007/s11010-023-04869-y
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11010-023-04869-y