Abstract
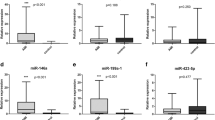
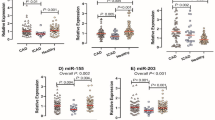
The role of inflammation has been proven in acute myocardial infarction (AMI) pathogenesis. Due to the effect of NLRP3 gene expression in the inflammation process of MI, we aimed to explore the expression changes and diagnostic power of four inflammation-related miRNAs including miR-17-3p, miR-101-3p, miR-335-3p, miR-296-3p and their potential target, NLRP3, in ST-segment elevation myocardial infarction (STEMI), and non-STEMI (NSTEMI) patients as two major classes of AMI. The expression level of these genes were evaluated in 300 participants equally divided into three groups of STEMI, NSTEMI, and control using quantitative real-time PCR. The expression level of NLRP3 was upregulated in STEMI and NSTEMI patients compared to control subjects. Besides, the expression levels of miR-17-3p, miR-101-3p, and miR-296-3p were significantly downregulated in STEMI and NSTEMI patients compared to controls. The increased expression of NLRP3 had a very strong inverse correlation with miR-17-3p in patients with STEMI and with miR-101-3p in the STEMI and NSTEMI patients. ROC curve analysis showed that the expression level of miR-17-3p had the highest diagnostic power for discrimination between STEMI patients and controls. Remarkably, the combination of all markers resulted in a higher AUC. In summary, there is a significant association between the expression levels of miR-17-3p, miR-101-3p, miR-335-3p, miR-296-3p, and NLRP3 and the incidence of AMI. Although the miR-17-3p expression level has the highest diagnostic power to distinguish between STEMI patients and control subjects, the combination of these miRNAs and NLRP3 could serve as a novel potential diagnostic biomarker of STEMI.


Similar content being viewed by others
Data availability
Data are available on request.
References
Anderson JL, Morrow DA (2017) Acute myocardial infarction. N Engl J Med 376:2053–2064. https://doi.org/10.1056/NEJMra1606915
Bhatt DL, Lopes RD, Harrington RA (2022) Diagnosis and treatment of acute coronary syndromes: a review. JAMA 327:662–675. https://doi.org/10.1001/jama.2022.0358
Daga LC, Kaul U, Mansoor A (2011) Approach to STEMI and NSTEMI. J Assoc Physicians India 59(Suppl):19–25
de Bliek EC (2018) ST elevation: differential diagnosis and caveats. A comprehensive review to help distinguish ST elevation myocardial infarction from nonischemic etiologies of ST elevation. Turk J Emerg Med 18:1–10. https://doi.org/10.1016/j.tjem.2018.01.008
Wu C, Liu B, Wang R, Li G (2021) The regulation mechanisms and clinical application of MicroRNAs in myocardial infarction: a review of the recent 5 years. Front Cardiovasc Med 8:580. https://doi.org/10.3389/fcvm.2021.809580
Feinberg MW, Moore KJ (2016) MicroRNA regulation of atherosclerosis. Circ Res 118:703–720. https://doi.org/10.1161/CIRCRESAHA.115.306300
Mahjoob G, Ahmadi Y, Fatima Rajani H, Khanbabaei N, Abolhasani S (2022) Circulating microRNAs as predictive biomarkers of coronary artery diseases in type 2 diabetes patients. J Clin Lab Anal 36:e24380. https://doi.org/10.1002/jcla.24380
Ruparelia N, Godec J, Lee R, Chai JT, Dall’Armellina E, McAndrew D, Digby JE, Forfar JC, Prendergast BD, Kharbanda RK, Banning AP, Neubauer S, Lygate CA, Channon KM, Haining NW, Choudhury RP (2015) Acute myocardial infarction activates distinct inflammation and proliferation pathways in circulating monocytes, prior to recruitment, and identified through conserved transcriptional responses in mice and humans. Eur Heart J 36:1923–1934. https://doi.org/10.1093/eurheartj/ehv195
Westman PC, Lipinski MJ, Luger D, Waksman R, Bonow RO, Wu E, Epstein SE (2016) Inflammation as a driver of adverse left ventricular remodeling after acute myocardial infarction. J Am Coll Cardiol 67:2050–2060. https://doi.org/10.1016/j.jacc.2016.01.073
Everett BM, Cornel JH, Lainscak M, Anker SD, Abbate A, Thuren T, Libby P, Glynn RJ, Ridker PM (2019) Anti-inflammatory therapy with canakinumab for the prevention of hospitalization for heart failure. Circulation 139:1289–1299. https://doi.org/10.1161/CIRCULATIONAHA.118.038010
Ridker PM, Everett BM, Thuren T, MacFadyen JG, Chang WH, Ballantyne C, Fonseca F, Nicolau J, Koenig W, Anker SD, Kastelein JJP, Cornel JH, Pais P, Pella D, Genest J, Cifkova R, Lorenzatti A, Forster T, Kobalava Z, Vida-Simiti L, Flather M, Shimokawa H, Ogawa H, Dellborg M, Rossi PRF, Troquay RPT, Libby P, Glynn RJ, Group CT (2017) Antiinflammatory therapy with canakinumab for atherosclerotic disease. N Engl J Med 377:1119–1131. https://doi.org/10.1056/NEJMoa1707914
Houshmandfar S, Saeedi-Boroujeni A, Rashno M, Khodadadi A, Mahmoudian-Sani MR (2021) miRNA-223 as a regulator of inflammation and NLRP3 inflammasome, the main fragments in the puzzle of immunopathogenesis of different inflammatory diseases and COVID-19. Naunyn Schmiedebergs Arch Pharmacol 394:2187–2195. https://doi.org/10.1007/s00210-021-02163-6
Haneklaus M, O’Neill LA (2015) NLRP3 at the interface of metabolism and inflammation. Immunol Rev 265:53–62. https://doi.org/10.1111/imr.12285
Zhang WJ, Chen SJ, Zhou SC, Wu SZ, Wang H (2021) Inflammasomes and fibrosis. Front Immunol 12:649. https://doi.org/10.3389/fimmu.2021.643149
Afrasyab A, Qu P, Zhao Y, Peng K, Wang H, Lou D, Niu N, Yuan D (2016) Correlation of NLRP3 with severity and prognosis of coronary atherosclerosis in acute coronary syndrome patients. Heart Vessels 31:1218–1229. https://doi.org/10.1007/s00380-015-0723-8
Peng H, Wu H, Zhang G, Zhang W, Guo Y, Chang L, Chen S, Xue R, Zhang S (2020) Expression and clinical prognostic value of platelet NLRP3 in acute coronary syndrome. Int J Gen Med 13:791–802. https://doi.org/10.2147/IJGM.S275481
Zamani P, Oskuee RK, Atkin SL, Navashenaq JG, Sahebkar A (2020) MicroRNAs as important regulators of the NLRP3 inflammasome. Prog Biophys Mol Biol 150:50–61. https://doi.org/10.1016/j.pbiomolbio.2019.05.004
Benjamin EJ, Muntner P, Alonso A, Bittencourt MS, Callaway CW, Carson AP, Chamberlain AM, Chang AR, Cheng S, Das SR, Delling FN, Djousse L, Elkind MSV, Ferguson JF, Fornage M, Jordan LC, Khan SS, Kissela BM, Knutson KL, Kwan TW, Lackland DT, Lewis TT, Lichtman JH, Longenecker CT, Loop MS, Lutsey PL, Martin SS, Matsushita K, Moran AE, Mussolino ME, O’Flaherty M, Pandey A, Perak AM, Rosamond WD, Roth GA, Sampson UKA, Satou GM, Schroeder EB, Shah SH, Spartano NL, Stokes A, Tirschwell DL, Tsao CW, Turakhia MP, VanWagner LB, Wilkins JT, Wong SS, Virani SS, American Heart Association Council on Epidemiology and Prevention Statistics Committee and Stroke Statistics Subcommittee (2019) Heart disease and stroke statistics-2019 update: a report from the American Heart Association. Circulation 139:e56–e528. https://doi.org/10.1161/CIR.0000000000000659
Adachi T, Nakanishi M, Otsuka Y, Nishimura K, Hirokawa G, Goto Y, Nonogi H, Iwai N (2010) Plasma microRNA 499 as a biomarker of acute myocardial infarction. Clin Chem 56:1183–1185. https://doi.org/10.1373/clinchem.2010.144121
He F, Lv P, Zhao X, Wang X, Ma X, Meng W, Meng X, Dong S (2014) Predictive value of circulating miR-328 and miR-134 for acute myocardial infarction. Mol Cell Biochem 394:137–144. https://doi.org/10.1007/s11010-014-2089-0
Xue S, Liu D, Zhu W, Su Z, Zhang L, Zhou C, Li P (2019) Circulating MiR-17-5p, MiR-126-5p and MiR-145-3p are novel biomarkers for diagnosis of acute myocardial infarction. Front Physiol 10:123. https://doi.org/10.3389/fphys.2019.00123
Zhu J, Wu S, Hu S, Li H, Li M, Geng X, Wang H (2019) NLRP3 inflammasome expression in peripheral blood monocytes of coronary heart disease patients and its modulation by rosuvastatin. Mol Med Rep 20:1826–1836. https://doi.org/10.3892/mmr.2019.10382
Sandanger O, Ranheim T, Vinge LE, Bliksoen M, Alfsnes K, Finsen AV, Dahl CP, Askevold ET, Florholmen G, Christensen G, Fitzgerald KA, Lien E, Valen G, Espevik T, Aukrust P, Yndestad A (2013) The NLRP3 inflammasome is up-regulated in cardiac fibroblasts and mediates myocardial ischaemia-reperfusion injury. Cardiovasc Res 99:164–174. https://doi.org/10.1093/cvr/cvt091
Chen H, Qi J, Bi Q, Zhang S (2017) Expression profile of long noncoding RNA (HOTAIR) and its predicted target miR-17-3p in LPS-induced inflammatory injury in human articular chondrocyte C28/I2 cells. Int J Clin Exp Pathol 10:9146–9157
Du WW, Li X, Li T, Li H, Khorshidi A, Liu F, Yang BB (2015) The microRNA miR-17-3p inhibits mouse cardiac fibroblast senescence by targeting Par4. J Cell Sci 128:293–304. https://doi.org/10.1242/jcs.158360
Du W, Pan Z, Chen X, Wang L, Zhang Y, Li S, Liang H, Xu C, Zhang Y, Wu Y, Shan H, Lu Y (2014) By targeting Stat3 microRNA-17-5p promotes cardiomyocyte apoptosis in response to ischemia followed by reperfusion. Cell Physiol Biochem 34:955–965. https://doi.org/10.1159/000366312
Li W, ** LY, Cui YB, **e N (2021) Human umbilical cord mesenchymal stem cells-derived exosomal microRNA-17–3p ameliorates inflammatory reaction and antioxidant injury of mice with diabetic retinopathy via targeting STAT1. Int Immunopharmacol 90:107010. https://doi.org/10.1016/j.intimp.2020.107010
Shi J, Bei Y, Kong X, Liu X, Lei Z, Xu T, Wang H, Xuan Q, Chen P, Xu J, Che L, Liu H, Zhong J, Sluijter JP, Li X, Rosenzweig A, **ao J (2017) miR-17-3p contributes to exercise-induced cardiac growth and protects against myocardial ischemia-reperfusion injury. Theranostics 7:664–676. https://doi.org/10.7150/thno.15162
Faltejskova P, Bocanek O, Sachlova M, Svoboda M, Kiss I, Vyzula R, Slaby O (2012) Circulating miR-17-3p, miR-29a, miR-92a and miR-135b in serum: evidence against their usage as biomarkers in colorectal cancer. Cancer Biomark 12:199–204. https://doi.org/10.3233/CBM-130308
Hu G, Lv Q, Yan J, Chen L, Du J, Zhao K, Xu W (2018) MicroRNA-17 as a promising diagnostic biomarker of gastric cancer: an investigation combining TCGA, GEO, meta-analysis, and bioinformatics. FEBS Open Bio 8:1508–1523. https://doi.org/10.1002/2211-5463.12496
Yang F, Li Y, Xu L, Zhu Y, Gao H, Zhen L, Fang L (2017) miR-17 as a diagnostic biomarker regulates cell proliferation in breast cancer. Onco Targets Ther 10:543–550. https://doi.org/10.2147/OTT.S127723
Shaker OG, Abdelaleem OO, Mahmoud RH, Abdelghaffar NK, Ahmed TI, Said OM, Zaki OM (2019) Diagnostic and prognostic role of serum miR-20b, miR-17-3p, HOTAIR, and MALAT1 in diabetic retinopathy. IUBMB Life 71:310–320. https://doi.org/10.1002/iub.1970
Li X, Zhang S, Wa M, Liu Z, Hu S (2019) MicroRNA-101 protects against cardiac remodeling following myocardial infarction via downregulation of runt-related transcription factor 1. J Am Heart Assoc 8:e013112. https://doi.org/10.1161/JAHA.119.013112
Pan Z, Sun X, Shan H, Wang N, Wang J, Ren J, Feng S, **e L, Lu C, Yuan Y, Zhang Y, Wang Y, Lu Y, Yang B (2012) MicroRNA-101 inhibited postinfarct cardiac fibrosis and improved left ventricular compliance via the FBJ osteosarcoma oncogene/transforming growth factor-beta1 pathway. Circulation 126:840–850. https://doi.org/10.1161/CIRCULATIONAHA.112.094524
Zhao X, Li S, Wang Z, Bai N, Feng Y (2021) miR-101-3p negatively regulates inflammation in systemic lupus erythematosus via MAPK1 targeting and inhibition of the NF-kappaB pathway. Mol Med Rep. https://doi.org/10.3892/mmr.2021.11998
Zheng Y, Wang Z, Tu Y, Shen H, Dai Z, Lin J, Zhou Z (2015) miR-101a and miR-30b contribute to inflammatory cytokine-mediated beta-cell dysfunction. Lab Investig 95:1387–1397. https://doi.org/10.1038/labinvest.2015.112
Funding
None.
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Material preparation, data collection and analysis were performed by [AB], [MHN], [AM], [PB], [LH], and [NS]. The first draft of the manuscript was written by [NS] and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
There is no conflict of interest.
Ethics approval
This study was conducted in line with the principles of the Declaration of Helsinki and according to the accepted regulations of the Shahid Beheshti University of Medical Sciences ethical committee (IR.SBMU.RETECH.REC.1400.892).
Consent to participate
Informed consent was obtained from all participants.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Bakhshi, A., Khani, M., Alipour Parsa, S. et al. Investigating the expression level of miR-17-3p, miR-101-3p, miR-335-3p, and miR-296-3p in the peripheral blood of patients with acute myocardial infarction. Mol Cell Biochem 479, 859–868 (2024). https://doi.org/10.1007/s11010-023-04766-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11010-023-04766-4