Abstract
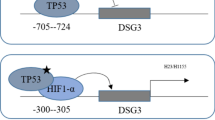
Cancer-associated fibroblasts (CAFs) have been proved to facilitate colorectal cancer (CRC) development, either with boosting chemo-resistance by communicating with CRC cells in the tumor microenvironment. However, the underlying molecular mechanisms remain largely unclear. Relative expressions of FOSL1 and ITGB4, either with their correlations in CRC tissues, were assessed using qRT-PCR analysis. Also, Kaplan–Meier survival analysis was employed for evaluating the prognosis. Identification of CAFs was determined by the detection of specific makers (α-SMA, FAP, and FSP1) using western blot and immunofluorescence staining. Cell proliferation, self-renewal capacity, and cell apoptosis were estimated by CCK-8, sphere-formation, and flow cytometry assays. Transcriptional regulation of FOSL1 on integrin β4 (ITGB4) was confirmed using ChIP and dual-luciferase reporter assays. Increased FOSL1 and ITGB4 in CRC tissues were both positively correlated with the poor prognosis of CRC patients. Interestingly, FOSL1 was enriched in the CAFs isolated from CRC stroma, instead of ITGB4. CRC cells under a co-culture system with CAFs-conditioned medium (CAFs-CM) exhibited increased FOSL1, promotive cell proliferation, and reduced apoptosis, while these effects could be blocked by exosome inhibitor (GW4869). Moreover, CAFs-derived exosomal FOSL1 was validated to enhance proliferative ability and oxaliplatin resistance of CRC cells. Our results uncovered that CAFs-derived exosomes could transfer FOSL1 to CRC cells, thereby promoting CRC cell proliferation, stemness, and oxaliplatin resistance by transcriptionally activating ITGB4.






Similar content being viewed by others
Data availability
The data underlying this article will be shared on reasonable request to the corresponding author.
References
Brenner H, Kloor M, Pox CP (2014) Colorectal cancer. Lancet 383(9927):1490–1502
Punt CJ, Tol J (2009) More is less – combining targeted therapies in metastatic colorectal cancer. Nat Rev Clin Oncol 6(12):731–733
Dekker E, Tanis PJ, Vleugels JLA, Kasi PM, Wallace MB (2019) Colorectal cancer. Lancet 394(10207):1467–1480
Van der Jeught K, Xu HC, Li YJ, Lu XB, Ji G (2018) Drug resistance and new therapies in colorectal cancer. World J Gastroenterol 24(34):3834–3848
Arneth, B., Tumor Microenvironment. Medicina (Kaunas), 2019. 56(1).
**ao Y, Yu D (2021) Tumor microenvironment as a therapeutic target in cancer. Pharmacol Ther 221:107753
Biffi G, Tuveson DA (2021) Diversity and biology of cancer-associated fibroblasts. Physiol Rev 101(1):147–176
Chen Y, McAndrews KM, Kalluri R (2021) Clinical and therapeutic relevance of cancer-associated fibroblasts. Nat Rev Clin Oncol 18(12):792–804
Li C, Teixeira AF, Zhu HJ, Ten Dijke P (2021) Cancer associated-fibroblast-derived exosomes in cancer progression. Mol Cancer 20(1):154
Zhang L, Yu D (2019) Exosomes in cancer development, metastasis, and immunity. Biochim Biophys Acta Rev Cancer 1871(2):455–468
Sobolev VV, Khashukoeva AZ, Evina OE, Geppe NA, Chebysheva SN, Korsunskaya IM et al (2022) Role of the Transcription Factor FOSL1 in Organ Development and Tumorigenesis. Int J Mol Sci 23(3):1521
Zhu X, Liu H, Xu Z, Zhang Y (2019) Expression and clinical significance of FOS-like antigen 1 in gastric adenocarcinoma. Pathol Res Pract 215(6):152394
Duan L, Zhao M, Ang L, Hu H, Wu Z, Wang J et al (2020) Down-regulation of FOS-like antigen 1 enhances drug sensitivity in breast cancer. Int J Clin Exp Pathol 13(8):2092–2099
Liu Y, Yue M, Li Z (2021) FOSL1 promotes tumorigenesis in colorectal carcinoma by mediating the FBXL2/Wnt/beta-catenin axis via Smurf1. Pharmacol Res 165:105405
Li L, Zhang W, Zhao S, Sun M (2019) FOS-like antigen 1 is a prognostic biomarker in hepatocellular carcinoma. Saudi J Gastroenterol 25(6):369–376
Kim JE, Kim BG, Jang Y, Kang S, Lee JH, Cho NH (2020) The stromal loss of miR-4516 promotes the FOSL1-dependent proliferation and malignancy of triple negative breast cancer. Cancer Lett 469:256–265
Zhou L, Li J, Tang Y, Yang M (2021) Exosomal LncRNA LINC00659 transferred from cancer-associated fibroblasts promotes colorectal cancer cell progression via miR-342-3p/ANXA2 axis. J Transl Med 19(1):8
Ren J, Ding L, Zhang D, Shi G, Xu Q, Shen S et al (2018) Carcinoma-associated fibroblasts promote the stemness and chemoresistance of colorectal cancer by transferring exosomal lncRNA H19. Theranostics 8(14):3932–3948
Liao Z, Tan ZW, Zhu P, Tan NS (2019) Cancer-associated fibroblasts in tumor microenvironment - Accomplices in tumor malignancy. Cell Immunol 343:103729
Yang X, Li Y, Zou L, Zhu Z (2019) Role of exosomes in crosstalk between cancer-associated fibroblasts and cancer cells. Front Oncol 9:356
von Ahrens D, Bhagat TD, Nagrath D, Maitra A, Verma A (2017) The role of stromal cancer-associated fibroblasts in pancreatic cancer. J Hematol Oncol 10(1):76
Affo S, Yu LX, Schwabe RF (2017) The Role of Cancer-Associated Fibroblasts and Fibrosis in Liver Cancer. Annu Rev Pathol 12:153–186
Yoshida GJ (2020) Regulation of heterogeneous cancer-associated fibroblasts: the molecular pathology of activated signaling pathways. J Exp Clin Cancer Res 39(1):112
Hu JL, Wang W, Lan XL, Zeng ZC, Liang YS, Yan YR et al (2019) CAFs secreted exosomes promote metastasis and chemotherapy resistance by enhancing cell stemness and epithelial-mesenchymal transition in colorectal cancer. Mol Cancer 18(1):91
Chen X, Liu Y, Zhang Q, Liu B, Cheng Y, Zhang Y et al (2021) Exosomal miR-590-3p derived from cancer-associated fibroblasts confers radioresistance in colorectal cancer. Mol Ther Nucleic Acids 24:113–126
Gu C, Lu H, Qian Z (2020) Matrine reduces the secretion of exosomal circSLC7A6 from cancer-associated fibroblast to inhibit tumorigenesis of colorectal cancer by regulating CXCR5. Biochem Biophys Res Commun 527(3):638–645
Dai C, Rennhack JP, Arnoff TE, Thaker M, Younger ST, Doench JG et al (2021) SMAD4 represses FOSL1 expression and pancreatic cancer metastatic colonization. Cell Rep 36(4):109443
Vallejo A, Erice O, Entrialgo-Cadierno R, Feliu I, Guruceaga E, Perugorria MJ et al (2021) FOSL1 promotes cholangiocarcinoma via transcriptional effectors that could be therapeutically targeted. J Hepatol 75(2):363–376
Zhang M, Hoyle RG, Ma Z, Sun B, Cai W, Cai H et al (2021) FOSL1 promotes metastasis of head and neck squamous cell carcinoma through super-enhancer-driven transcription program. Mol Ther 29(8):2583–2600
Jiang X, Wang J, Wang M, Xuan M, Han S, Li C et al (2021) ITGB4 as a novel serum diagnosis biomarker and potential therapeutic target for colorectal cancer. Cancer Med 10(19):6823–6834
Chen B, Ma Y, Bi J, Wang W, He A, Su G et al (2021) Regulation network of colorectal-cancer-specific enhancers in the progression of colorectal cancer. Int J Mol Sci 22(15):8337
Zhu X, Jiang X, Zhang Q, Huang H, Shi X, Hou D et al (2022) TCN1 Deficiency Inhibits the Malignancy of Colorectal Cancer Cells by Regulating the ITGB4 Pathway. Gut Liver. https://doi.org/10.5009/gnl210494
Sung JS, Kang CW, Kang S, Jang Y, Chae YC, Kim BG et al (2020) ITGB4-mediated metabolic reprogramming of cancer-associated fibroblasts. Oncogene 39(3):664–676
Leng C, Zhang ZG, Chen WX, Luo HP, Song J, Dong W et al (2016) An integrin beta4-EGFR unit promotes hepatocellular carcinoma lung metastases by enhancing anchorage independence through activation of FAK-AKT pathway. Cancer Lett 376(1):188–196
Acknowledgements
The authors thanked the participants for their contribution to the study. We also thanked Prof. Hu for her assistance during data analysis and manuscript revision.
Funding
This work was supported by the Medical Research Foundation of Guangdong Province (No.B2022159); Research Project of Jiangmen Central Hospital (No.J202104) and Medical and Health Science and Technology Project of Jiangmen (No.2022YL01065, No.2022YL01089).
Author information
Authors and Affiliations
Contributions
SSL—acquisition of data; analysis and interpretation of data; drafting the manuscript. BZ—conception and design of study; acquisition of data; revising the manuscript critically for important intellectual content.
Corresponding author
Ethics declarations
Competing interests
The authors have no commercial or other associations that might pose a conflict of interest.
Ethical approval
This study has obtained approval of the Ethics Committee of Zhongshan City People’s Hospital.
Consent to participate
Written informed consent was obtained from the patients.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Lin, S., Zhu, B. Exosome-transmitted FOSL1 from cancer-associated fibroblasts drives colorectal cancer stemness and chemo-resistance through transcriptionally activating ITGB4. Mol Cell Biochem 479, 665–677 (2024). https://doi.org/10.1007/s11010-023-04737-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11010-023-04737-9