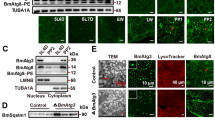
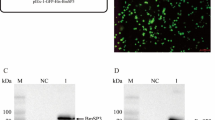
Abstract
Silkworm (Bombyx mori) is not only a model organism for scientific studies, but also a commercial insect for agricultural production. BmAtg8 (a B. mori homolog of yeast Atg8) plays crucial roles in macroautophagy (hereafter referred to autophagy), which is helpful for silkworm metamorphosis. Relevant mechanism about BmAtg8 currently remains ambiguous. Based on our previous acetylome of B. mori after BmNPV infection, we focused on that acetylation of BmAtg8 K13 was changed upon virus challenge. Subsequently, anti-BmAtg8 antibody was generated, and EBSS-induced BmN cellular autophagy model was established. Next, by constructing acetylation-mimic K13Q or deacetylation-mimic K13R mutant BmAtg8, we further examined that K13 of BmAtg8 was acetylated after BmNPV infection and chose 3 h as an appropriate point after EBSS treatment for autophagy initiation. Furthermore, acetylation of BmAtg8 K13 significantly reduced BmAtg8-PE formation in the presence of EBSS, thereby interfering autophagy initiation. Interestingly, acetylated K13 of BmAtg8 contributed to weaken interaction with Atg7, which may influence BmAtg8-PE conjugation. Eventually, acetylation of BmAtg8 K13 is critical for attenuating cell rescue through impaired autophagy initiation. Taken together, our data support an acetylated molecular function for BmAtg8 during starvation-induced autophagy, and provide insights into the modulating mechanisms that potentially reveal the LC3 (a mammalian homolog of Atg8) function in mammal.






Similar content being viewed by others
References
Sun W, Yu H, Shen Y, Banno Y, **ang Z, Zhang Z (2012) Phylogeny and evolutionary history of the silkworm. Sci China Life Sci 55:483–496. https://doi.org/10.1007/s11427-012-4334-7
Mizushima N (2007) Autophagy: process and function. Genes Dev 21:2861–2873. https://doi.org/10.1101/gad.1599207
Klionsky DJ (2007) Autophagy: from phenomenology to molecular understanding in less than a decade. Nat Rev Mol Cell Biol 8:931–937. https://doi.org/10.1038/nrm2245
Mizushima N, Yoshimori T, Levine B (2010) Methods in mammalian autophagy research. Cell 140:313–326. https://doi.org/10.1016/j.cell.2010.01.028
Tian L, Ma L, Guo E, Deng X, Ma S, **a Q, Cao Y, Li S (2013) 20-Hydroxyecdysone upregulates Atg genes to induce autophagy in the Bombyx fat body. Autophagy 9:1172–1187. https://doi.org/10.4161/auto.24731
Franzetti E, Huang ZJ, Shi YX, **e K, Deng XJ, Li JP, Li QR, Yang WY, Zeng WN, Casartelli M, Deng HM, Cappellozza S, Grimaldi A, **a Q, Feng Q, Cao Y, Tettamanti G (2012) Autophagy precedes apoptosis during the remodeling of silkworm larval midgut. Apoptosis 17:305–324. https://doi.org/10.1007/s10495-011-0675-0
Goncu E, Parlak O (2008) Some autophagic and apoptotic features of programmed cell death in the anterior silk glands of the silkworm, Bombyx mori. Autophagy 4:1069–1072. https://doi.org/10.4161/auto.6953
Ichimura Y, Kirisako T, Takao T, Satomi Y, Shimonishi Y, Ishihara N, Mizushima N, Tanida I, Kominami E, Ohsumi M, Noda T, Ohsumi Y (2000) A ubiquitin-like system mediates protein lipidation. Nature 408:488–492. https://doi.org/10.1038/35044114
Nakatogawa H, Ichimura Y, Ohsumi Y (2007) Atg8, a ubiquitin-like protein required for autophagosome formation, mediates membrane tethering and hemifusion. Cell 130:165–178. https://doi.org/10.1016/j.cell.2007.05.021
Noda NN, Ohsumi Y, Inagaki F (2010) Atg8-family interacting motif crucial for selective autophagy. FEBS Lett 584:1379–1385. https://doi.org/10.1016/j.febslet.2010.01.018
Pankiv S, Alemu EA, Brech A, Bruun JA, Lamark T, Overvatn A, Bjørkøy G, Johansen T (2010) FYCO1 is a Rab7 effector that binds to LC3 and PI3P to mediate microtubule plus end-directed vesicle transport. J Cell Biol 188:253–269. https://doi.org/10.1083/jcb.200907015
Joachim J, Jefferies HB, Razi M, Frith D, Snijders AP, Chakravarty P, Judith D, Tooze SA (2015) Activation of ULK kinase and autophagy by GABARAP trafficking from the centrosome is regulated by WAC and GM130. Mol Cell 60:899–913. https://doi.org/10.1016/j.molcel.2015.11.018
Maruyama Y, Sou YS, Kageyama S, Takahashi T, Ueno T, Tanaka K, Komatsu M, Ichimura Y (2014) LC3B is indispensable for selective autophagy of p62 but not basal autophagy. Biochem Biophys Res Commun 446:309–315. https://doi.org/10.1016/j.bbrc.2014.02.093
Scott RC, Juhász G, Neufeld TP (2007) Direct induction of autophagy by Atg1 inhibits cell growth and induces apoptotic cell death. Curr Biol 17:1–11. https://doi.org/10.1016/j.cub.2006.10.053
Hu C, Zhang X, Teng YB, Hu HX, Li WF (2010) Structure of autophagy-related protein Atg8 from the silkworm Bombyx mori. Acta Crystallogr F 66:787–790. https://doi.org/10.1107/S1744309110018464
Ji MM, Lee JM, Mon H, Iiyama K, Tatsuke T, Morokuma D, Hino M, Yamashita M, Hirata K, Kusakabe T (2017) Lipidation of BmAtg8 is required for autophagic degradation of p62 bodies containing ubiquitinated proteins in the silkworm, Bombyx mori. Insect Biochem Mol Biol 89:86–96. https://doi.org/10.1016/j.ibmb.2017.08.006
Ji MM, Lee JM, Mon H, Xu J, Tatsuke T, Kusakabe T (2016) Proteasome inhibitor MG132 impairs autophagic flux through compromising formation of autophagosomes in Bombyx cells. Biochem Biophys Res Commun 479:690–696. https://doi.org/10.1016/j.bbrc.2016.09.151
Hu D, Xue S, Zhao C, Wei M, Yan H, Quan Y, Yu W (2018) Comprehensive profiling of lysine acetylome in baculovirus infected silkworm (Bombyx mori) cells. Proteomics 18:1700133. https://doi.org/10.1002/pmic.201700133
Lee IH, Cao L, Mostoslavsky R, Lombard DB, Liu J, Bruns NE, Tsokos M, Alt FW, Finkel T (2008) A role for the NAD-dependent deacetylase Sirt1 in the regulation of autophagy. Proc Natl Acad Sci USA 105:3374–3379. https://doi.org/10.1073/pnas.0712145105
Füllgrabe J, Lynch-Day MA, Heldring N, Li W, Struijk RB, Ma Q, Hermanson O, Rosenfeld MG, Klionsky DJ, Joseph B (2013) The histone H4 lysine 16 acetyltransferase hMOF regulates the outcome of autophagy. Nature 500:468–471. https://doi.org/10.1038/nature12313
Yi C, Ma M, Ran L, Zhang J, Tong J, Zhu J, Ma C, Sun Y, Zhang S, Feng W, Zhu L, Le Y, Gong X, Yan X, Hong B, Jiang FJ, **e Z, Miao D, Deng H, Yu L (2012) Function and molecular mechanism of acetylation in autophagy regulation. Science 336:474–477. https://doi.org/10.1126/science.1216990
Lee IH, Finkel T (2009) Regulation of autophagy by the p300 acetyltransferase. J Biol Chem 284:6322–6328. https://doi.org/10.1074/jbc.M807135200
Lin SY, Li TY, Liu Q, Zhang C, Li X, Chen Y, Zhang SM, Lian G, Liu Q, Ruan K, Wang Z, Zhang CS, Chien KY, Wu J, Li Q, Han J, Lin SC (2012) GSK3-TIP60-ULK1 signaling pathway links growth factor deprivation to autophagy. Science 336:477–481. https://doi.org/10.1126/science.1217032
Morselli E, Maiuri MC, Markaki M, Megalou E, Pasparaki A, Palikaras K, Criollo A, Galluzzi L, Malik SA, Vitale I, Michaud M, Madeo F, Tavernarakis N, Kromer G (2010) Caloric restriction and resveratrol promote longevity through the Sirtuin-1-dependent induction of autophagy. Cell Death Dis 1:e10. https://doi.org/10.1038/cddis.2009.8
Huang R, Xu Y, Wan W, Shou X, Qian J, You Z, Liu B, Chang C, Zhou T, Lippincott-Schwartz J, Liu W (2015) Deacetylation of nuclear LC3 drives autophagy initiation under starvation. Mol Cell 57:456–466. https://doi.org/10.1016/j.molcel.2014.12.013
Wei Y, Zou Z, Becker N, Anderson M, Sumpter R, **ao G, Kinch L, Koduru P, Christudass CS, Veltri RW, Grishin NV, Peyton M, Minna J, Bhagat G, Levine B (2013) EGFR-mediated Beclin 1 phosphorylation in autophagy suppression, tumor progression, and tumor chemoresistance. Cell 154:1269–1284. https://doi.org/10.1016/j.cell.2013.08.015
Kim J, Kundu M, Viollet B, Guan KL (2011) AMPK and mTOR regulate autophagy through direct phosphorylation of Ulk1. Nat Cell Biol 13:132–141. https://doi.org/10.1038/ncb2152
Kamada Y, Funakoshi T, Shintani T, Nagano K, Ohsumi M, Ohsumi Y (2000) Tor-mediated induction of autophagy via an Apg1 protein kinase complex. J Cell Biol 150:1507–1513. https://doi.org/10.1083/jcb.150.6.1507
Jordan TX, Randall G (2012) Manipulation or capitulation: virus infections with autophagy. Microbes Infect 14:126–139. https://doi.org/10.1016/j.micinf.2011.09.007
Lv S, Xu Q, Sun E, Yang T, Li J, Feng Y, Zhang Q, Wang H, Zhang J, Wu D (2015) Autophagy activated by bluetongue virus infection plays a positive role in its replication. Viruses 7:4657–4675. https://doi.org/10.3390/v7082838
Shelly S, Lukinova N, Bambina S, Berman A, Cherry S (2009) Autophagy is an essential component of Drosophila immunity against vesicular stomatitis virus. Immunity 30:588–598. https://doi.org/10.1016/j.immuni.2009.02.009
Lee HK, Lund JM, Ramanathan B, Mizushima N, Iwasaki A (2007) Autophagy-dependent viral recognition by plasmacytoid dendritic cells. Science 315:1398–1401. https://doi.org/10.1126/science.1136880
Wang L, **ao Q, Zhou XL, Zhu Y, Dong ZQ, Chen P, Pan MH, Lu C (2017) Bombyx mori nuclear polyhedrosis virus (BmNPV) induces host cell autophagy to benefit infection. Viruses 10:E14. https://doi.org/10.3390/v10010014
Fitzwalter BE, Thorburn A (2015) Recent insights into cell death and autophagy. FEBS J 282:4279–4288. https://doi.org/10.1111/febs.13515
Ho SN, Hunt HD, Horton RM, Pullen JK, Pease LR (1989) Site-directed mutagenesis by overlap extension using the polymerase chain reaction. Gene 77:51–59. https://doi.org/10.1016/0378-1119(89)90358-2
Reggiori F, Klionsky DJ (2002) Autophagy in the eukaryotic cell. Eukaryot Cell 1:11–21. https://doi.org/10.1128/EC.01.1.11-21.2002
Drake KR, Kang M, Kenworthy AK (2010) Nucleocytoplasmic distribution and dynamics of the autophagosome marker EGFP-LC3. PLoS ONE 5:e9806. https://doi.org/10.1371/journal.pone.0009806
Klionsky DJ, Abdelmohsen K, Abe A et al (2016) Guidelines for the use and interpretation of assays for monitoring autophagy. Autophagy 12:1–222. https://doi.org/10.1080/15548627.2015.1100356
Noda NN, Satoo K, Fujioka Y, Kumeta H, Ogura K, Nakatogawa H, Ohsumi Y, Inagaki F (2011) Structural basis of Atg8 activation by a homodimeric E1, Atg7. Mol Cell 44:462–475. https://doi.org/10.1016/j.molcel.2011.08.035
Tait SW, Ichim G, Green DR (2014) Die another way—non-apoptotic mechanisms of cell death. J Cell Sci 127:2135–2144. https://doi.org/10.1242/jcs.093575
Reggiori F, Monastyrska I, Verheije MH, Calì T, Ulasli M, Bianchi S, Bernasconi R, de Haan CA, Molinari M (2010) Coronaviruses hijack the LC3-I-positive EDEMosomes, ER-derived vesicles exporting short-lived ERAD regulators, for replication. Cell Host Microbe 7:500–508. https://doi.org/10.1016/j.chom.2010.05.013
Acknowledgements
This work was supported by the Science foundation of Zhejiang province (No. LY17C170006) and National High-tech R&D program (863 Program) (No. 2011AA100603).
Author information
Authors and Affiliations
Contributions
W.Y conceived and designed the experiments. S.X, F.M and D.H performed the experiments. H.Y, J.L, E.O, Y.Z and Y.Q participated in data analysis and provided technical assistance. W.Y and F.M drafted the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Xue, S., Mao, F., Hu, D. et al. Acetylation of BmAtg8 inhibits starvation-induced autophagy initiation. Mol Cell Biochem 457, 73–81 (2019). https://doi.org/10.1007/s11010-019-03513-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11010-019-03513-y