Abstract
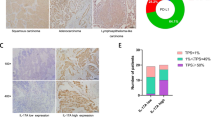
The present study is to investigate the effect and mechanism of action of interleukin (IL)-17A and its receptor IL-17RA on non-small cell lung cancer (NSCLC). A total of 139 NSCLC patients were included in the study. NSCLC tissues and tumor-adjacent tissues were collected from the patients. Human NSCLC cell lines H157, H1975, and A549 were used for in vitro studies. MTT assay was performed to determine cell proliferation. Wound healing assay was used to determine cell motility. Transwell assay was carried out to detect migration and invasion. Quantitative real-time polymerase chain reaction was conducted to measure mRNA expression, while Western blotting was used for determine protein expression. Immunohistochemistry was employed to evaluate IL-17RA expression in 139 primary human NSCLC tissues. Levels of IL-17RA in NSCLC tissues were higher than tumor-adjacent normal tissues, and associated with clinical outcomes. Kaplan–Meier survival analysis indicated that NSCLC patients with positive IL-17RA expression had a poor survival. In addition, IL-17A/IL-17RA affected NSCLC cell migration and invasion in vitro. Treatment with IL-17A/IL-17RA increased the expression of MMP-2 and MMP-9 in NSCLC cells. Furthermore, phosphorylation of p38 was enhanced in IL-17RA-overexpressing NSCLC cells. P38 MAPK-specific inhibitor SB203580 suppressed the migration and invasion of NSCLC cells. MMP-2 and MMP-9 were downstream effectors of IL-17RA and p38 signaling pathways. The present study demonstrates that P38 MAPK activity is crucial for IL-17A/IL-17RA to promote NSCLC metastasis. In addition, IL-17A/IL-17RA signaling may be a novel and promising cancer therapeutic target for the treatment of NSCLC.







Similar content being viewed by others
References
Miller KD, Siegel RL, Lin CC, Mariotto AB, Kramer JL, Rowland JH, Stein KD, Alteri R, Jemal A (2016) Cancer treatment and survivorship statistics, 2016. CA: Cancer J Clin 66(4):271–289. https://doi.org/10.3322/caac.21349
Siegel RL, Miller KD, Jemal A (2017) Cancer statistics, 2017. CA: Cancer J Clin 67(1):7–30. https://doi.org/10.3322/caac.21387
Vineis P, Wild CP (2014) Global cancer patterns: causes and prevention. Lancet 383(9916):549–557. https://doi.org/10.1016/s0140-6736(13)62224-2
Korn T, Bettelli E, Oukka M, Kuchroo VK (2009) IL-17 and Th17 Cells. Annu Rev Immunol 27:485–517. https://doi.org/10.1146/annurev.immunol.021908.132710
Weaver CT, Hatton RD, Mangan PR, Harrington LE (2007) IL-17 family cytokines and the expanding diversity of effector T cell lineages. Annu Rev Immunol 25:821–852. https://doi.org/10.1146/annurev.immunol.25.022106.141557
Ji Y, Zhang W (2010) Th17 cells: positive or negative role in tumor? Cancer Immunol Immunother 59(7):979–987. https://doi.org/10.1007/s00262-010-0849-6
Wang L, Yi T, Kortylewski M, Pardoll DM, Zeng D, Yu H (2009) IL-17 can promote tumor growth through an IL-6-Stat3 signaling pathway. J Exp Med 206(7):1457–1464. https://doi.org/10.1084/jem.20090207
Gu FM, Li QL, Gao Q, Jiang JH, Zhu K, Huang XY, Pan JF, Yan J, Hu JH, Wang Z, Dai Z, Fan J, Zhou J (2011) IL-17 induces AKT-dependent IL-6/JAK2/STAT3 activation and tumor progression in hepatocellular carcinoma. Mol Cancer 10:150. https://doi.org/10.1186/1476-4598-10-150
Hus I, Bojarska-Junak A, Chocholska S, Tomczak W, Wos J, Dmoszynska A, Rolinski J (2013) Th17/IL-17A might play a protective role in chronic lymphocytic leukemia immunity. PLoS ONE 8(11):e78091. https://doi.org/10.1371/journal.pone.0078091
Wright JF, Bennett F, Li B, Brooks J, Luxenberg DP, Whitters MJ, Tomkinson KN, Fitz LJ, Wolfman NM, Collins M, Dunussi-Joannopoulos K, Chatterjee-Kishore M, Carreno BM (2008) The human IL-17F/IL-17A heterodimeric cytokine signals through the IL-17RA/IL-17RC receptor complex. J Immunol 181(4):2799–2805
Huang Q, Du J, Fan J, Lv Z, Qian X, Zhang X, Han J, Chen C, Wu F, ** Y (2014) The effect of proinflammatory cytokines on IL-17RA expression in NSCLC. Med Oncol 31(9):144. https://doi.org/10.1007/s12032-014-0144-z
Jiang YX, Li PA, Yang SW, Hao YX, Yu PW (2015) Increased chemokine receptor IL-17RA expression is associated with poor survival in gastric cancer patients. Int J Clin Exp Pathol 8(6):7002–7008
Liu Y, Zhao X, Sun X, Li Y, Wang Z, Jiang J, Han H, Shen W, Corrigan CJ, Sun Y (2015) Expression of IL-17A, E, and F and their receptors in human prostatic cancer: comparison with benign prostatic hyperplasia. Prostate 75(16):1844–1856. https://doi.org/10.1002/pros.23058
Wang K, Kim MK, Di Caro G, Wong J, Shalapour S, Wan J, Zhang W, Zhong Z, Sanchez-Lopez E, Wu LW, Taniguchi K, Feng Y, Fearon E, Grivennikov SI, Karin M (2014) Interleukin-17 receptor a signaling in transformed enterocytes promotes early colorectal tumorigenesis. Immunity 41(6):1052–1063. https://doi.org/10.1016/j.immuni.2014.11.009
Wang M, Wang L, Ren T, Xu L, Wen Z (2013) IL-17A/IL-17RA interaction promoted metastasis of osteosarcoma cells. Cancer Biol Ther 14(2):155–163. https://doi.org/10.4161/cbt.22955
Asukai K, Kawamoto K, Eguchi H, Konno M, Nishida N, Koseki J, Noguchi K, Hasegawa S, Ogawa H, Yamada D, Tomimaru Y, Tomokuni A, Asaoka T, Noda T, Wada H, Gotoh K, Marubashi S, Nagano H, Doki Y, Mori M, Ishii H (2015) Prognostic impact of peritumoral IL-17-positive cells and IL-17 Axis in patients with intrahepatic cholangiocarcinoma. Ann Surg Oncol 22(Suppl 3):S1524–S1531. https://doi.org/10.1245/s10434-015-4782-y
Wang ZD, Huang C, Li ZF, Yang J, Li BH, Liang RR, Dai ZJ, Liu ZW (2010) Chrysanthemum indicum ethanolic extract inhibits invasion of hepatocellular carcinoma via regulation of MMP/TIMP balance as therapeutic target. Oncol Rep 23(2):413–421
Kessenbrock K, Plaks V, Werb Z (2010) Matrix metalloproteinases: regulators of the tumor microenvironment. Cell 141(1):52–67. https://doi.org/10.1016/j.cell.2010.03.015
Hadler-Olsen E, Winberg JO, Uhlin-Hansen L (2013) Matrix metalloproteinases in cancer: their value as diagnostic and prognostic markers and therapeutic targets. Tumour Biol 34(4):2041–2051. https://doi.org/10.1007/s13277-013-0842-8
Liu J, ** W, Zu Y, Sun W (2014) Correlations of lysyl oxidase with MMP2/MMP9 expression and its prognostic value in non-small cell lung cancer. Int J Clin Exp Pathol 7(9):6040–6047
Greenberg AK, Basu S, Hu J, Yie TA, Tchou-Wong KM, Rom WN, Lee TC (2002) Selective p38 activation in human non-small cell lung cancer. Am J Respir Cell Mol Biol 26(5):558–564. https://doi.org/10.1165/ajrcmb.26.5.4689
Kato S, Yokoyama S, Hayakawa Y, Li L, Iwakami Y, Sakurai H, Saiki I (2016) P38 pathway as a key downstream signal of connective tissue growth factor to regulate metastatic potential in non-small-cell lung cancer. Cancer Sci 107(10):1416–1421. https://doi.org/10.1111/cas.13009
Zhang Q, Liu S, Parajuli KR, Zhang W, Zhang K, Mo Z, Liu J, Chen Z, Yang S, Wang AR, Myers L, You Z (2017) Interleukin-17 promotes prostate cancer via MMP7-induced epithelial-to-mesenchymal transition. Oncogene 36(5):687–699. https://doi.org/10.1038/onc.2016.240
Li J, Lau GK, Chen L, Dong SS, Lan HY, Huang XR, Li Y, Luk JM, Yuan YF, Guan XY (2011) Interleukin 17A promotes hepatocellular carcinoma metastasis via NF-kB induced matrix metalloproteinases 2 and 9 expression. PLoS One 6(7):e21816. https://doi.org/10.1371/journal.pone.0021816
Maeda S, Omata M (2008) Inflammation and cancer: role of nuclear factor-kappaB activation. Cancer Sci 99(5):836–842. https://doi.org/10.1111/j.1349-7006.2008.00763.x
Kwon HC, Kim SH, Oh SY, Lee S, Lee JH, Jang JS, Kim MC, Kim KH, Kim SJ, Kim SG, Kim HJ (2012) Clinicopathologic significance of expression of nuclear factor-kappaB RelA and its target gene products in gastric cancer patients. World J Gastroenterol 18(34):4744–4750. https://doi.org/10.3748/wjg.v18.i34.4744
Zhang Y, Liu J, Kou J, Yu J, Yu B (2012) DT-13 suppresses MDA-MB-435 cell adhesion and invasion by inhibiting MMP-2/9 via the p38 MAPK pathway. Mol Med Rep 6(5):1121–1125. https://doi.org/10.3892/mmr.2012.1047
Kim ES, Kim MS, Moon A (2004) TGF-beta-induced upregulation of MMP-2 and MMP-9 depends on p38 MAPK, but not ERK signaling in MCF10A human breast epithelial cells. Int J Oncol 25(5):1375–1382
Acknowledgements
This work was supported by the National Natural Science Foundation of China (No. 81460354) and **njiang Uygur Autonomous Region Natural Science Foundation (No. 2018D01C266).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
All authors declare no financial competing interests. All authors declare no non-financial competing interests.
Rights and permissions
About this article
Cite this article
Wu, Z., He, D., Zhao, S. et al. IL-17A/IL-17RA promotes invasion and activates MMP-2 and MMP-9 expression via p38 MAPK signaling pathway in non-small cell lung cancer. Mol Cell Biochem 455, 195–206 (2019). https://doi.org/10.1007/s11010-018-3483-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11010-018-3483-9