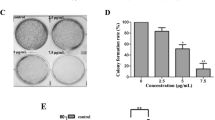
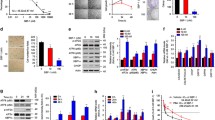
Abstract
Human manganese superoxide dismutase (hMnSOD) is a new type of cancer suppressor. Nonamer of arginine (R9) is an efficient protein transduction domain (PTD). The aim of the study was to improve the transduction efficiency of hMnSOD and investigate its activity in vitro. In this study, we designed, constructed, expressed, and purified a novel fusion protein containing the hMnSOD domain and R9 PTD (hMnSOD–R9). The DNA damaged by Fenton’s reagent was found to be significantly reduced when treated with hMnSOD–R9. hMnSOD–R9 fusion protein was successfully delivered into HeLa cells. The MTT assay showed that proliferation of various cancer cell lines were inhibited by hMnSOD–R9 in a dose-dependent manner. In addition, the cell cycle of HeLa cells was arrested at the sub-G0 phase by hMnSOD–R9. hMnSOD–R9 induced apoptosis of HeLa cells in a dose-dependent manner. With hMnSOD–R9 treatment, Bax, JNK, TBK1 gene expression was increased and STAT3 gene expression was gradually down-regulated in HeLa cells. We also found that apoptosis was induced by hMnSOD–R9 in HeLa cells via up-regulation of cleaved caspase-3 and down-regulation phospho-STAT3 pathway. These results indicated that hMnSOD–R9 may provide benefits to cervical cancer treatment.




Similar content being viewed by others
References
Michiels C, Raes M, Toussaint O, Remacle J (1994) Importance of Se-glutathione peroxidase, catalase, and Cu/Zn-SOD for cell survival against oxidative stress. Free Radic Biol Med 17:235–248
Pani G, Colavitti R, Bedogni B, Fusco S, Ferraro D, Borrello S, Galeotti T (2004) Mitochondrial superoxide dismutase: a promising target for new anticancer therapies. Curr Med Chem 11:1299–1308
Bravard A, Sabatier L, Hoffschir F, Ricoul M, Luccioni C, Dutrillaux B (1992) SOD2: a new type of tumor-suppressor gene? Int J Cancer 51:476–480
Zhong W, Oberley LW, Oberley TD, Yan T, Domann FE, St Clair DK (1996) Inhibition of cell growth and sensitization to oxidative damage by overexpression of manganese superoxide dismutase in rat glioma cells. Cell Growth Differ 7:1175–1186
Oberley TD (2004) Mitochondria, manganese superoxide dismutase, and cancer. Antioxid Redox Signal 6:483–487
Li Z, Khaletskiy A, Wang J, Wong JY, Oberley LW, Li JJ (2001) Genes regulated in human breast cancer cells overexpressing manganese-containing superoxide dismutase. Free Radic Biol Med 30:260–267
van den Berg A, Dowdy SF (2011) Protein transduction domain delivery of therapeutic macromolecules. Curr Opin Biotechnol 22:888–893
Mitchell DJ, Kim DT, Steinman L, Fathman CG, Rothbard JB (2000) Polyarginine enters cells more efficiently than other polycationic homopolymers. J Pept Res 56:318–325
Zhou HH, Zhang AX, Zhang Y, Zhu DY (2012) Cloning, expression, and purification of a recombinant Tat-HA-NR2B9c peptide. Protein Expr Purif 85:239–245
Feng W, Mei S, Wenjie Y, Luyuan H (2011) High-level soluble expression of recombinant human manganese superoxide dismutase in Escherichia coli, and its effects on proliferation of the leukemia cell. Protein Expr Purif 77:46–52
Li CL, Xu TT, Chen RB, Huang XX, Zhao YC, Bao YY, Zhao WD, Zheng ZY (2013) Cloning, expression and characterization of antimicrobial porcine beta defensin 1 in Escherichia coli. Protein Expr Purif 88:47–53
Liu D, Zhang R, Yang X, Xu Y, Tang Z, Tian W, Shen Q (2011) Expression, purification and characterization of two thermostable endoglucanases cloned from a lignocellulosic decomposing fungi Aspergillus fumigatus Z5 isolated from compost. Protein Expr Purif 79:176–186
Yamada S, Kanno H, Kawahara N (2012) Trans-membrane peptide therapy for malignant glioma by use of a peptide derived from the MDM2 binding site of p53. J Neurooncol 109:7–14
Verma AR, Vijayakumar M, Rao CV, MatHeLa CS (2010) In vitro and in vivo antioxidant properties and DNA damage protective activity of green fruit of Ficus glomerata. Food Chem Toxicol 48:704–709
Kim YG, Park HY (2004) The effects of Pycnogenol on DNA damage in vitro and expression of superoxide dismutase and HP1 in Escherichia coli SOD and catalase deficient mutant cells. Phytother Res 18:900–905
Chen W, Shen X, **a X, Xu G, Ma T, Bai X, Liang T (2012) NSC 74859-mediated inhibition of STAT3 enhances the anti-proliferative activity of cetuximab in hepatocellular carcinoma. Liver Int 32:70–77
He Y, Huang C, Sun X, Long XR, Lv XW, Li J (2012) MicroRNA-146a modulates TGF-beta1-induced hepatic stellate cell proliferation by targeting SMAD4. Cell Signal 24:1923–1930
De Preter K, Speleman F, Combaret V, Lunec J, Laureys G, Eussen BHJ, Francotte N, Board J, Pearson ADJ, De Paepe A, Van Roy N, Vandesompele J (2002) Quantification of MYCN, DDX1, and NAG gene copy number in neuroblastoma using a real-time quantitative PCR assay. Mod Pathol 15:159–166
Yao Y, Zhang YW, Sun LG, Liu B, Bao YL, Lin H, Zhang Y, Zheng LH, Sun Y, Yu CL, Wu Y, Wang GN, Li YX (2012) Juglanthraquinone C, a novel natural compound derived from Juglans mandshurica Maxim, induces S phase arrest and apoptosis in HepG2 cells. Apoptosis 17:832–841
Chumanov RS, Burgess RR (2010) Expression, purification, and refolding of active Nrf2 transcription factor fused to protein transduction TAT tag. Protein Expr Purif 74:280–288
Choi HS, An JJ, Kim SY, Lee SH, Kim DW, Yoo KY, Won MH, Kang TC, Kwon HJ, Kang JH, Cho SW, Kwon OS, Park J, Eum WS, Choi SY (2006) PEP-1-SOD fusion protein efficiently protects against paraquat-induced dopaminergic neuron damage in a Parkinson disease mouse model. Free Radic Biol Med 41:1058–1068
Clarke RG, Lund EK, Johnson IT, Pinder AC (2000) Apoptosis can be detected in attached colonic adenocarcinoma HT29 cells using annexin V binding, but not by TUNEL assay or sub-G0 DNA content. Cytometry 39:141–150
Liu QL, Hang XM, Liu XL, Tan J, Li DT, Yang H (2012) Cloning and heterologous expression of the manganese superoxide dismutase gene from Lactobacillus casei Lc18. Ann Microbiol 62:129–137
Li AH, Kong Y, Cho SH, Lee HW, Na BK, Pak JK, Kim TS (2005) Molecular cloning and characterization of the copper/zinc and manganese superoxide dismutase genes from the human parasite Clonorchis sinensis. Parasitology 130:687–697
Noorbakhsh R, Mortazavi SA, Sankian M, Shahidi F, Assarehzadegan MA, Varasteh A (2010) Cloning, expression, characterization, and computational approach for cross-reactivity prediction of manganese superoxide dismutase allergen from pistachio nut. Allergol Int 59:295–304
Li JJ, Oberley LW, St Clair DK, Ridnour LA, Oberley TD (1995) Phenotypic changes induced in human breast cancer cells by overexpression of manganese-containing superoxide dismutase. Oncogene 10:1989–2000
Zhong W, Oberley LW, Oberley TD, St Clair DK (1997) Suppression of the malignant phenotype of human glioma cells by overexpression of manganese superoxide dismutase. Oncogene 14:481–490
Liu R, Oberley TD, Oberley LW (1997) Transfection and expression of MnSOD cDNA decreases tumor malignancy of human oral squamous carcinoma SCC-25 cells. Hum Gene Ther 8:585–595
Li N, Oberley TD, Oberley LW, Zhong W (1998) Overexpression of manganese superoxide dismutase in DU145 human prostate carcinoma cells has multiple effects on cell phenotype. Prostate 35:221–233
Mansouri A, Tarhuni A, Larosche I, Reyl-Desmars F, Demeilliers C, Degoul F, Nahon P, Sutton A, Moreau R, Fromenty B, Pessayre D (2010) MnSOD overexpression prevents liver mitochondrial DNA depletion after an alcohol binge but worsens this effect after prolonged alcohol consumption in mice. Dig Dis 28:756–775
H-l Guo, Zhao H-w Xu, Z-f Ma H, Song X, Guan J, Z-j Li, J-m Yu (2005) Manganese superoxide dismutase gene transfection of mouse small intestinal epithelial cells protects them from radiation injury. Zhonghua Zhongliu Zazhi 27:672–675
Bogoyevitch MA, Ngoei KRW, Zhao TT, Yeap YYC, Ng DCH (2010) c-Jun N-terminal kinase (JNK) signaling: recent advances and challenges. Biochim Biophys Acta 1804:463–475
Coffey ET, Smiciene G, Hongisto V, Cao J, Brecht S, Herdegen T, Courtney MJ (2002) c-Jun N-terminal protein kinase (JNK) 2/3 is specifically activated by stress, mediating c-Jun activation, in the presence of constitutive JNK1 activity in cerebellar neurons. J Neurosci 22:4335–4345
Westphal D, Dewson G, Czabotar PE, Kluck RM (2011) Molecular biology of Bax and Bak activation and action. Biochim Biophys Acta 1813:521–531
** J, **ao YC, Chang JH, Yu JY, Hu HB, Starr R, Brittain GC, Chang M, Cheng XH, Sun SC (2012) The kinase TBK1 controls IgA class switching by negatively regulating noncanonical NF-kappa B signaling. Nat Immunol 13:1101–1109
Siddiquee KAZ, Turkson J (2008) STAT3 as a target for inducing apoptosis in solid and hematological tumors. Cell Res 18:254–267
Mora LB, Buettner R, Seigne J, Diaz J, Ahmad N, Garcia R, Bowman T, Falcone R, Fairclough R, Cantor A, Muro-Cacho C, Livingston S, Karras J, Pow-Sang J, Jove R (2002) Constitutive activation of Stat3 in human prostate tumors and cell lines: direct inhibition of Stat3 signaling induces apoptosis of prostate cancer cells. Cancer Res 62:6659–6666
Garcia R, Bowman TL, Niu GL, Yu H, Minton S, Muro-Cacho CA, Cox CE, Falcone R, Fairclough R, Parsons S, Laudano A, Gazit A, Levitzki A, Kraker A, Jove R (2001) Constitutive activation of Stat3 by the Src and JAK tyrosine kinases participates in growth regulation of human breast carcinoma cells. Oncogene 20:2499–2513
Chen CL, Hsieh FC, Lieblein JC, Brown J, Chan C, Wallace JA, Cheng G, Hall BM, Lin J (2007) Stat3 activation in human endometrial and cervical cancers. Br J Cancer 96:591–599
Acknowledgments
This work was supported by grants from the National Natural Science Foundation of China (30700704), Science and Technology Innovation Project from Department of Education of Guangdong Province (2012KJCX0017) and the Key Laboratory of Regenerative Biology, Guangzhou Institute of Biomedicine and Health, Chinese Academy of Sciences (KLRB201105).
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Zhang, Z., Huang, L., Wu, Q. et al. A recombinant trans-membrane protein hMnSOD–R9 inhibits the proliferation of cervical cancer cells in vitro. Mol Cell Biochem 385, 79–86 (2014). https://doi.org/10.1007/s11010-013-1816-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11010-013-1816-2