Abstract
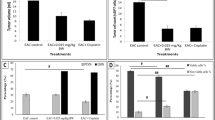
Lung cancer is the prime cause of death after breast cancer among non-infectious diseases and is highly prevalent across the world. Several chemotherapies, immunotherapies, radiation therapies, and combination therapies have been employed, but their side effects, low efficiency, and off-target effects necessitate the exploration of other sources of therapeutic agents. The A549 lung cancer cell line was procured from NCCS, Pune, and treated for 48 h with increasing concentrations from 2 to 20 µg/mL of NN-32, a snake venom peptide obtained from the Naja naja cobra species. The MTT assay was done to assess the working concentration of NN-32. Apoptosis, Reactive Oxygen Species (ROS) production, mitochondrial membrane potential (MMP), and cell cycle arrest analysis were carried out and analysed using flow cytometry. The results were analysed using GraphPad Prism 8. The MTT assay revealed that the cytotoxic effect of NN-32 is limited to cancer cells at concentrations ranging from 2 to 20 µg/mL, and for subsequent experiments, 5 µg/mL and 10 µg/mL \(\left( {\frac{1}{ 2 }{\text{IC}}_{{{50}}} \;{\text{and IC}}_{{{5}0}} \;{\text{respectively}}} \right)\) were used. Early apoptosis was significantly observed at 5 μg/mL and 10 μg/mL when compared with the control (P < 0.05). Phase contrast microscopy and Scanning Electronic Microscopy (SEM) were used to detect apoptotic bodies within the cells. NN-32 showed a higher percentage of cells in the S phase of the cell cycle when compared with the control. It also induced ROS production and disrupted MMP. NN-32, a snake venom peptide, has a cytotoxic effect on lung cancer cells, making it a promising anti-cancer agent.







Similar content being viewed by others
Data Availability
Data are all contained within the paper.
References
Al-Asmari AK, Riyasdeen A, Al-Shahrani MH, Islam M (2016) Snake venom causes apoptosis by increasing the reactive oxygen species in colorectal and breast cancer cell lines. Onco Targets Ther 9:6485–6498
Alberg AJ, Brock MV, Ford JG, Samet JM, Spivack SD (2013) Epidemiology of lung cancer. Chest 143(5 Suppl):e1S-e29S
Attarde SS, Pandit SV (2017) Cytotoxic activity of NN-32 toxin from Indian spectacled cobra venom on human breast cancer cell lines. BMC Complement Altern Med 17(1):503. https://doi.org/10.1186/s12906-017-2018-3
Attarde SS, Pandit SV (2020) In vivo toxicity profile of NN-32 and nanogold conjugated GNP-NN-32 from Indian spectacled cobra venom. Curr Pharm Biotechnol 21(14):1479–1488. https://doi.org/10.2174/1389201021666200519101221
Baskar R, Lee KA, Yeo R, Yeoh KW (2012) Cancer and radiation therapy: current advances and future directions. Int J Med Sci 9(3):193–199
Bordon KCF, Cologna CT, Fornari-Baldo EC et al (2020) From animal poisons and venoms to medicines: achievements, challenges and perspectives in drug discovery. Front Pharmacol 11:1132
Cabral C, Efferth T, Pires IM, Severino P, Lemos MFL (2018) Natural products as a source for new leads in cancer research and treatment. Evid-Based Complement Altern Med 2018:e8243680
Calmette A, Saenz A, Costil L (1933) Effects du venin de cobra sur les greffes cancereuses et sur le cancer spontane (adeno-carcinome) de la souris. CR Acad Sci 197:205–210
Chatterjee K, Zhang J, Honbo N, Karliner JS (2010) Doxorubicin cardiomyopathy. Cardiology 115(2):155–162
Cragg GM, Pezzuto JM (2016) Natural products as a vital source for the discovery of cancer chemotherapeutic and chemopreventive agents. MPP 25(Suppl. 2):41–59
Cura JE, Blanzaco DP, Brisson C, Cura MA, Cabrol R, Larrateguy L, Mendez C, Sechi JC, Silveira JS, Theiller E, de Roodt AR, Vidal JC (2002) Phase I and pharmacokinetics study of crotoxin (cytotoxic PLA(2), NSC-624244) in patients with advanced cancer. Clin Cancer Res 8(4):1033–1041
Das T, Bhattacharya S, Halder B et al (2011) Cytotoxic and antioxidant property of a purified fraction (NN-32) of Indian Naja naja venom on Ehrlich ascites carcinoma in BALB/c mice. Toxicon 57(7–8):1065–1072
Das T, Bhattacharya S, Biswas A, Gupta SD, Gomes A, Gomes A (2013) Inhibition of leukemic U937 cell growth by induction of apoptosis, cell cycle arrest and suppression of VEGF, MMP-2 and MMP-9 activities by cytotoxin protein NN-32 purified from Indian spectacled cobra (Naja naja) venom. Toxicon 65:1–4
Debnath A, Saha A, Gomes A et al (2010) A lethal cardiotoxic-cytotoxic protein from the Indian monocellate cobra (Naja kaouthia) venom. Toxicon 56(4):569–579
Ebrahim K, Shirazi FH, Vatanpour H, Zare A, Kobarfard F, Rabiei H (2014) Anticancer activity of cobra venom polypeptide, cytotoxin-II, against human breast adenocarcinoma cell line (MCF-7) via the induction of apoptosis. J Breast Cancer 17(4):314–322
Ebrahim K, Vatanpour H, Zare A, Shirazi FH, Nakhjavani M (2016) Anticancer activity a of Caspian Cobra (Naja naja oxiana) snake venom in human cancer cell lines via induction of apoptosis. Iran J Pharm Res 15(Suppl):101–112
Frangieh J, Rima M, Fajloun Z et al (2021) Snake venom components: tools and cures to target cardiovascular diseases. Molecules 26(8):2223
Fu Y, An N, Li K, Zheng Y, Liang A (2012) Chlorotoxin-conjugated nanoparticles as potential glioma-targeted drugs. J Neurooncol 107(3):457–462. https://doi.org/10.1007/s11060-011-0763-6
Gasanov SE, Dagda RK, Rael ED (2014) Snake venom cytotoxins, phospholipase A2s, and Zn2+-dependent metalloproteinases: mechanisms of action and pharmacological relevance. J Clin Toxicol 4(1):1000181. https://doi.org/10.4172/2161-0495.1000181
Gomes A, Bhattacharya S, Chakraborty M, Bhattacharjee P, Mishra R, Gomes A (2010) Anti-arthritic activity of Indian monocellate cobra (Naja kaouthia) venom on adjuvant induced arthritis. Toxicon 55(2):670–673
Harris JB, Goonetilleke A (2004) Animal poisons and the nervous system: what the neurologist needs to know. J Neurol Neurosurg Psychiatry 75(suppl 3):iii40–iii46
Hiu JJ, Yap MKK (2020) Cytotoxicity of snake venom enzymatic toxins: phospholipase A2 and l-amino acid oxidase. Biochem Soc Trans 48(2):719–731
Huang M, Lu JJ, Ding J (2021) Natural products in cancer therapy: past present and future. Nat Prod Bioprospect 11(1):5–13
Johnson DH, Schiller JH, Bunn PA (2014) Recent clinical advances in lung cancer management. J Clin Oncol 32(10):973–982
Koh DCI, Armugam A, Jeyaseelan K (2006) Snake venom components and their applications in biomedicine. Cell Mol Life Sci 63(24):3030–3041
Li Y, Fan J, Ju D (2019) 15 - Neurotoxicity concern about the brain targeting delivery systems. In: Gao H, Gao X (eds) Brain targeted drug delivery system. Academic Press, New York, pp 377–408
Lin S, Chang C, Hsu C et al (2020) Natural compounds as potential adjuvants to cancer therapy: preclinical evidence. Br J Pharmacol 177(6):1409–1423
Mathur P, Sathishkumar K, Chaturvedi M, Das P, Sudarshan KL, Santhappan S, Nallasamy V, John A, Narasimhan S, Roselind FS, ICMR-NCDIR-NCRP Investigator Group (2020) Cancer statistics, 2020: report from National Cancer Registry Programme, India. JCO Glob Oncol 6:1063–1075. https://doi.org/10.1200/GO.20.00122
Nath A, Sathishkumar K, Das P, Sudarshan KL, Mathur P (2022) A clinicoepidemiological profile of lung cancers in India—results from the National Cancer Registry Programme. Indian J Med Res 155(2):264–272
Pal SK, Gomes A, Dasgupta SC, Gomes A (2002) Snake venom as therapeutic agents: from toxin to drug development. Indian J Exp Biol 40(12):1353–1358
Panigrahi MK, Saka VK, Jaganathan V, Sinnassamy M, Narahari NK (2018) Changing trend of clinical and epidemiological profile of lung cancer—experience from a tertiary care centre in Southern India. J Clin Diagn Res 12:LC01–LC03. https://doi.org/10.7860/JCDR/2018/36768.12148
Park MH, Jo M, Won D et al (2012) Snake venom toxin from vipera lebetina turanicainduces apoptosis of colon cancer cells via upregulation of ROS- and JNK-mediated death receptor expression. BMC Cancer 12(1):228. https://doi.org/10.1186/1471-2407-12-228
Rodriguez-Canales J, Parra-Cuentas E, Wistuba II (2016) Diagnosis and molecular classification of lung cancer. Cancer Treat Res 170:25–46
Ruiz-Cordero R, Devine WP (2020) Targeted therapy and checkpoint immunotherapy in lung cancer. Surg Pathol Clin 13(1):17–33
Shanbhag VKL (2015) Applications of snake venoms in treatment of cancer. Asian Pac J Trop Biomed 5(4):275–276. https://doi.org/10.1016/S2221-1691(15)30344-0
Sinaei N, Mirakabadi AZ, Jafari E et al (2023) The cytotoxic effects of partially purified cytotoxic peptides of Naja naja Oxiana venom on human glioblastoma multiforme: an in vitro study. Int J Pept Res Ther 29:14. https://doi.org/10.1007/s10989-022-10479-x
Singh N, Agrawal S, Jiwnani S et al (2021) Lung cancer in India. J Thorac Oncol 16(8):1250–1266
Snakes PR (2005). In: Wexler P (ed) Encyclopedia of toxicology, 2nd edn. Elsevier, Amsterdam, pp 57–60
Son DJ, Park MH, Chae SJ et al (2007) Inhibitory effect of snake venom toxin from Vipera lebetina turanica on hormone-refractory human prostate cancer cell growth: induction of apoptosis through inactivation of nuclear factor kappaB. Mol Cancer Ther 6(2):675–683
Stone SF, Isbister GK, Shahmy S et al (2013) Immune response to snake envenoming and treatment with antivenom; complement activation, cytokine production and mast cell degranulation. PLoS Negl Trop Dis 7(7):e2326
Torre LA, Siegel RL, Jemal A (2016) Lung cancer statistics. Adv Exp Med Biol 893:1–19. https://doi.org/10.1007/978-3-319-24223-1_1
UrraVivas-Ruiz FADE, Sanchez EF (2022) An emergent role for mitochondrial bioenergetics in the action of snake venom toxins on cancer cells. Front Oncol. https://doi.org/10.3389/fonc.2022.938749
Vyas VK, Brahmbhatt K, Bhatt H, Parmar U (2013) Therapeutic potential of snake venom in cancer therapy: current perspectives. Asian Pac J Trop Biomed 3(2):156–162
Zappa C, Mousa SA (2016) Non-small cell lung cancer: current treatment and future advances. Transl Lung Cancer Res 5(3):288
Acknowledgements
We are grateful to Dr. Kishori Apte for valuable suggestions, Mr. Saurabh Attarde for providing the NN-32 and for time-to-time suggestions and thankful to the Head, Department of Zoology for providing laboratory facilities.
Funding
NA.
Author information
Authors and Affiliations
Contributions
Conception, experiment design, overall monitoring and final approval of the article was done by Dr. SVP corresponding author. Conducting Experiments in Laboratory were contributed jointly by AJ and PK, Data collection, Data analysis, graphical representation and interpretation were handled by PK. Article was written by PK and edited by Dr. SVP. All Authors read and approved the final manuscripts
Corresponding author
Ethics declarations
Competing interests
We declare that there is no conflict of interest.
Ethical Approval/Ethics Approval and Consent to Participate
The study protocol was approved by the Institutional Biosafety Committee (IBSC) of Savitribai Phule Pune University, Pune, India.
Consent for Publication
We have the consent for this publication.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Kurkute, P., Jadhav, A. & Pandit, S.V. Anticancer Potential and Cytotoxic Activity of NN-32, a Snake Venom Peptide, Against A549, Lung Cancer Cell Line. Int J Pept Res Ther 29, 67 (2023). https://doi.org/10.1007/s10989-023-10527-0
Accepted:
Published:
DOI: https://doi.org/10.1007/s10989-023-10527-0