Abstract
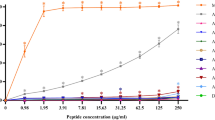
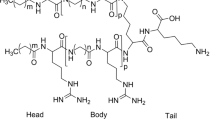
Pyrrhocoricin and drosocin, representatives of the short, proline-rich antimicrobial peptide family kill bacteria by inactivating the bacterial heat shock protein DnaK and inhibiting chaperone-assisted protein folding. The molecular architecture of these peptides features an N-terminal DnaK-binding half and a C-terminal delivery unit, capable of crossing bacterial membranes. Cell penetration is enhanced if multiple copies of pyrrhocoricin are conjugated. To obtain drug leads with improved antimicrobial properties, and possible utility as therapeutic agents, we synthesized chimeric dimers, in which pyrrhocoricin’s potent DnaK-binding domain was connected to drosocin’s superior cell penetrating module. Indeed, the new constructs not only exhibited enhanced in vitro antibacterial properties against the originally sensitive strains Escherichia coli, Klebsiella pneumoniae and Salmonella typhimurium, but also showed activity against Staphylococcus aureus, a bacterial strain resistant to native pyrrhocoricin and drosocin. The improved antimicrobial profile could be demonstrated with assays designed to distinguish intracellular or membrane activities. While a novel mixed pyrrhocoricin–drosocin dimer and the purely pyrrhocoricin-based old dimer bound E. coli DnaK with an identical 4 μM K d, the mixed dimers penetrated a significantly larger number of E. coli and S. aureus cells than the previous analogs and destroyed a larger percentage of bacterial membrane structures. Toxicity to human red blood cells could not be observed up to the highest peptide concentration tested, 640 μM. In addition, repetitive reculturing of E. coli or S. aureus cells with sublethal concentrations of the mixed dimer did not result in resistance induction to the novel peptide antibiotic. The new concept of pyrrhocoricin–drosocin mixed dimers yields antibacterial peptide derivatives acting with a multiple mode of action, and can serve as a useful addition to the current antimicrobial therapy repertoire.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Abbreviations
- Chex:
-
1-amino-1-cyclohexane carboxylic acid
- Dab:
-
2,4-diamino-butyric acid
- DIC:
-
differential interference contrast
- Hsp:
-
heat shock protein
- LPS:
-
lipopolysaccharide
- MALDI:
-
matrix-assisted laser desorption/ionization
- MHB:
-
Muller–Hinton broth
- MIC:
-
minimal inhibitory concentration
- PBS:
-
phosphate buffered saline
- Pip:
-
4-amino-piperidine-4-carboxylic acid
- RP-HPLC:
-
reversed-phase high performance liquid chromatography
- TNF:
-
tumor necrosis factor
References
D. Andreu L. Rivas (1998) Biopolymers 47 415–433
A.-M. Bencivengo M. Cudic R. Hoffmann L. Otvos SuffixJr. (2001) Lett. Pept. Sci 8 201–209
H. G. Boman (1995) Annu. Rev. Immunol 13 61–92
D. M. Bowdish D. J. Davidson D. P. Speert R. E. Hancock (2004) J. Immunol 172 3758–3765
M.A. Bower M. Cudic W. Campbell J.D. Wade L. Otvos SuffixJr. (2003) Lett. Pept. Sci 10 466–473
P. Bulet J.-L. Dimarcq C. Hetru M. Lagueux M. Charlet G. Hegy A. Dorsselaer Particlevan J. A. Hoffmann (1993) J.Biol. Chem 268 14893–14897
P. Bulet L. Urge S. Ohresser C. Hetru L. Otvos SuffixJr. (1996) Eur. J. Biochem 238 64–69
P. Casteels P. Tempst (1994) Biochem. Biophys. Res. Commun 199 339–345
P. Casteels C. Ampe F. Jacobs M. Vaeck P. Tempst (1989) EMBO J 8 2387–2391
P. Casteels J. Romagnolo M. Castle K. Casteels-Josson H. Erdjument-Bromage P. Tempst (1994) J. Biol. Chem 269 26107–26115
M. Castle A. Nazarian S.-S. Yi P. Tempst (1999) J. Biol. Chem 274 32555–32564
L. S. Chesnokova S. V. Slepenkov S. N. Witt (2004) FEBS Lett 565 65–69 Occurrence Handle10.1016/j.febslet.2004.03.075
S. Cociancich A. Ghazi C. Hetru J. A. Hoffmann L. Letellier (1993) J. Biol. Chem 268 19239–19245
S. Cociancich A. Dupont G. Hegy R. Lanot F. Holder C. Hetru J. A. Hoffmann P. Bulet (1994) Biochem. J 300 567–575
M. Cudic L. Otvos SuffixJr. (2002) Curr. Drug Targets 3 101–106
M. Cudic P. Bulet R. Hoffmann D. J. Craik L. Otvos SuffixJr. (1999) Eur. J. Biochem 266 549–558
M. Cudic B. A. Condie D. J. Weiner E. S. Lysenko Z. Q. O. I. **ang P. Bulet L. Otvos SuffixJr. (2002) Peptides 23 2071–2083
M. Cudic C. V. Lockatell D. E. Johnson L. Otvos SuffixJr. (2003) Peptides 24 807–820
D. A. Devine R. E. Hancock (2002) Curr. Pharm. Des 8 703–714
A. DiNardo A. Vitiello R. L. Gallo (2003) J. Immunol 170 2274–2278
G. B. Fields R. L. Noble (1990) Int. J. Pept. Protein Res 35 161–214
B. B. Finlay R. E. W. Hancock (2004) Nat. Rev. Microbiol 2 497–504
C. L. Friedrich D. Moyles T. J. Beveridge R. E. Hancock (2000) Antimicrob. Agents Chemother 44 2086–2092
R. L. Gallo K. M. Huttner (1998) J. Invest. Dermatol 111 739–743
R. Gennaro M. Zanetti M. Benincasa E. Podda M. Miani (2002) Curr. Pharm. Des 8 763–778
R.E.W. Hancock M.G. Scott (2000) Proc. Natl. Acad. Sci. USA 97 8856–8861
R. Hoffmann P. Bulet L. Urge L. Otvos SuffixJr. (1999) Biochim. Biophys. Acta 1426 459–467
G. Kragol S. Lovas G. Varadi B. A. Condie R. Hoffmann L. Otvos SuffixJr. (2001) Biochemistry 40 3016–3026
G. Kragol R. Hoffmann M. A. Chattergoon S. Lovas M. Cudic P. Bulet B. A. Condie K. J. Rosengren L. J. Montaner L. Otvos SuffixJr. (2002) Eur. J. Biochem 269 4226–4237
S. Ludtke K. He H. Huang (1995) Biochemistry 34 16764–16769
J. A. Mackintosh D. A. Veal A. J. Beattie A. A. Gooley (1998) J. Biol. Chem 273 6139–6143
A. McManus L. Otvos SuffixJr. R. Hoffmann D. J. Craik (1999) Biochemistry 38 705–714
M. Meister B. Lemaitre J. A. Hoffmann (1997) BioEssays 19 1019–1026
L. Otvos SuffixJr. (2000) J. Pept. Sci 6 497–511
L. Otvos SuffixJr. K. Bokonyi I. Varga B. I. Otvos R. Hoffmann H. C. J. Ertl J. D. Wade A. M. McManus D. J. Craik P. Bulet (2000a) Protein Sci 9 742–749
L. O. I. Otvos SuffixJr. M. E. Rogers P. J. Consolvo B. A. Condie S. Lovas P. O. I. Bulet M. Blaszczyk-Thurin (2000b) Biochemistry 39 14150–14159
L. Otvos SuffixJr. M. Cudic B. Y. Chua G. Deliyannis D. C. Jackson (2004) Mol. Pharmaceut 1 220–232
M. G. Scott D. J. Davidson M. R. Gold D. Bowdish R. E. W. Hancock (2002) J. Immunol 169 3883–3891
P. Srivastava (2002) Annu. Rev. Immunol 20 395–425
H. Steiner D. Andreu R. B. Merrifield (1988) Biochim. Biophys. Acta 939 260–266
L. Urge L. Otvos SuffixJr. (1995) Lett. Pept. Sci 1 207–212
S. Uttenweiler-Joseph M. Moniatte M. Lagueux A. Van Dorsselaer J. A. Hoffmann P. Bulet (1998) Proc. Natl. Acad. Sci. USA 95 11342–11347
T. Wieprecht O. Apostolov M. Beyermann J. Seelig (2000) Biochemistry 39 442–452
W. C. Wimley M. E. Selsted S. H. White (1994) Protein Sci 3 1361–1373
M. Wu E. Maier R. Benz R. E. W. Hancock (1999) Biochemistry 38 7235–7242
D. Yang Q. Chen A. P. Schmidt G. M. Anderson J. M. Wang J. Wooters J. J. Oppenheim O. Chertov (2000) J. Exp. Med 192 1069–1074
M. Zasloff (2002) Nature 415 389–395 Occurrence Handle10.1038/415389a Occurrence Handle1:CAS:528:DC%2BD38XhtVCgsLs%3D Occurrence Handle11807545
L. Zhang R. Benz R. E. W. Hancock (1999) Biochemistry 38 8102–8111
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
This article is published under an open access license. Please check the 'Copyright Information' section either on this page or in the PDF for details of this license and what re-use is permitted. If your intended use exceeds what is permitted by the license or if you are unable to locate the licence and re-use information, please contact the Rights and Permissions team.
About this article
Cite this article
Otvos, L., Snyder, C., Condie, B. et al. Chimeric Antimicrobial Peptides Exhibit Multiple Modes of Action. Int J Pept Res Ther 11, 29–42 (2005). https://doi.org/10.1007/s10989-004-1719-x
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/s10989-004-1719-x