Abstract
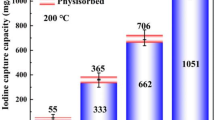
Novel Cu2O/Cu@C/SiO2 materials with low cost were conveniently synthesized using C/SiO2 derived from rice-husk as support and used for iodine gas adsorption. The iodine adsorption behaviors affected by Cu content, contact time and temperature were investigated. The adsorption capacity as high as 820 mg/g was achieved benefiting from the synergistic effect between Cu2O/Cu and C/SiO2. The results demonstrated that both Cu2O and Cu changed to CuI after iodine adsorption, indicating that the dominant adsorption mechanism was chemical reaction through formation of CuI. This work revealed that the obtained Cu2O/Cu@C/SiO2 could be included in the list of candidate iodine gas adsorbents.












Similar content being viewed by others
References
Muhire C, Reda AT, Zhang DX, Xu XY, Cui C (2022) An overview on metal Oxide based materials for iodine capture and storage. Chem Eng J 431:133816
Tian ZJ, Chee TS, Meng RX, Hao YX, Zhou XY, Ma B, Zhu L, Duan T, **ao CL (2022) Incipient wetness impregnation to prepare bismuth-modified all-silica beta zeolite for efficient radioactive iodine capture. Environ Funct Mater 1:92–104
He LW, Chen L, Dong XL, Zhang SH, Zhang MX, Dai X, Liu XJ, Lin P, Li KF, Chen CL, Pan TT, Ma FY, Chen JC, Yuan MJ, Zhang YG, Chen L, Zhou RH, Han Y, Chai ZF, Wang SA (2021) A nitrogen-rich covalent organic framework for simultaneous dynamic capture of iodine and methyl iodide. Chem 7:699–714
Sun HX, La PQ, Zhu ZQ, Liang WD, Yang BP, Li A (2015) Capture and reversible storage of volatile iodine by porous carbon with high capacity. J Mater Sci 50:7326–7332
Muhammad R, Attia NF, Cho S, Park J, Jung M (2020) Exploitation of surface heterogeneity and textural properties in nanoporous carbon fabrics for efficient iodine capture. Thin Solid Films 706:138049
Bo A, Sarina S, Zheng ZF, Yang DJ, Liu HW, Zhu HY (2013) Removal of radioactive iodine from water using Ag2O grafted titanate nanolamina as efficient adsorbent. J Hazard Mater 246–247:199–205
Chen YY, Yu SH, Yao QZ, Fu SQ, Zhou GT (2018) One-step synthesis of Ag2O@Mg (OH)2 nanocomposite as an efficient scavenger for iodine and uranium. J Colloid Interface Sci 510:280–291
Yang DJ, Liu HW, Liu L, Sarina S, Zheng ZF, Zhu HY (2013) Silver oxide nanocrystals anchored on titanate nanotubes and nanofbers: promising candidates for entrapment of radioactive iodine anions. Nanoscale 5:11011–11018
Wang W, Chu TW (2018) Formation of AgI/Ag3PO4 solid solution on alumina for enhancing radioactive iodine adsorption at high temperatures. Nucl Sci Tech 29:1–11
Azambre B, Chebbi M, Hijazi A (2020) Effects of the cation and Si/Al ratio on CH3I adsorption by faujasite zeolites. Chem Eng J 379:122308
Nan Y, Tavlarides LL, Depaoli DW (2016) Adsorption of Iodine on hydrogen-reduced silver-exchanged mordenite: experiments and modeling. Am Inst Chem Eng 63:1024–1035
Hwan J, Cho Y, Myeong J, Yim M (2015) Bismuth-embedded SBA-15 mesoporous silica for radioactive iodine capture and stable storage. J Nucl Mater 465:556–564
**an Q, Chen L, Fan WJ, Liu Y, He XM, Dan H, Zhu L, Ding Y, Duan T (2022) Facile synthesis of novel Bi0-SBA-15 adsorbents by an improved impregnation reduction method for highly efficient capture of iodine gas. J Hazard Mater 424:127678
Reda AT, Zhang DX, Xu XY, Xu SY (2022) Highly stable iodine capture by pillared montmorillonite functionalized Bi2O3@g-C3N4 nano sheets. Sep Purif Technol 292:120994
Tian ZJ, Chee TS, Zhang XW, Lei LC, **ao CL (2021) Novel bismuth-based electrospinning materials for highly efficient capture of radioiodine. Chem Eng J 412:128687
Tian ZJ, Chee TS, Zhu L, Dua T, Zhang XW, Lei LH, **ao CL (2021) Comprehensive comparison of bismuth and silver functionalized nickel foam composites in capturing radioactive gaseous iodine. J Hazard Mater 417:125978
Zou H, Yi FC, Song MX, Wang XQ, Bian L, Li W, Pan N, Jiang XQ (2019) Novel synthesis of Bi-Bi2O3-TiO2-C composite for capturing iodine-129 in off-gas. J Hazard Mater 365:81–87
Li M, Yuan GY, Zeng Y, Yang YY, Liao JL, Yang JJ, Liu N (2020) Flexible surface-supported MOF membrane via a convenient approach for efficient iodine adsorption. J Radioanal Nucl Ch 324:1167–1177
Chen P, He XH, Pang MB, Dong XT, Zhao S, Zhang W (2020) Iodine capture using Zr-based metal-organic frameworks (Zr-MOFs): adsorption performance and mechanism. Appl Mater Interfaces 12:20429–20439
Banerjee D, Chen X, Lobanov S, Plonka AM, Daly J, Kim T, Thallapally PK, Parise J, Banerjee D, Chen X, Lobanov SS, Plonka AM, Chan X, John A, Kim T, Thallapally PK, Parise JB (2018) Iodine adsorption in metal organic frameworks in presence of humidity. Nat Commun 10:10622–10626
Zahid M, Zhang D, Xu X, Pan M, Ul Haq MH, Reda AT, Xu W (2021) Barbituric andthiobarbituric acid-based UiO-66-NH2 adsorbents for iodine gas capture: haracterization, efficiency and mechanisms. J Hazard Mater 416:125835
Liu T, Zhao Y, Song M, Pang XH, Shi XF, Jia JJ, Chi LF, Lu G (2023) Ordered macro-microporous single crystals of covalent organic frameworks with efficient sorption of iodine. J Am Chem Soc 145:2544–2552
**e YQ, Pan TT, Lei Q, Chen CL, Dong XL, Yuan YY, Maksoud WA, Zhao L, Cavallo L, Pinnau I, Han Y (2022) Efficient and simultaneous capture of iodine and methyl iodide achieved by a covalent organic framework. Nat Commun 13:2878
Liu XW, Zhang AR, Ma R, Wu B, Wen T, Ai YJ, Sun MT, ** J, Wang SH, Wang XK (2022) Experimental and theoretical insights into copper phthalocyanine-based covalent organic frameworks for highly efficient radioactive iodine capture. Chin Chem Lett 33:3549–3555
**e YQ, Pan TT, Lei Q, Chen CL, Dong XL, Yuan YY, Shen J, Cai YC, Zhou CH, Pinnau I, Han Y (2021) Ionic functionalization of multivariate covalent organic frameworks to achieve an exceptionally high iodine-capture capacity. Angew Chem Int Ed 60:22432–22440
Jiang B, Qi Y, Li XF, Guo XH, Jia ZM, Zhang J, Li Y, Ma LJ (2022) Efficient gaseous iodine capture enhanced by charge-induced effect of covalent organic frameworks with dense tertiary-amine nodes. Chin Chem Lett 33:3556–3560
Moore RC, Pearce CI, Morad JW, Chatterjee S, Levitskaia TG, Asmussen RM, Lawter AR, Neeway JJ, Qafoku NP, Rigali MJ, Saslow SA, Szecsody JE, Thallapally PK, Wang G, Freedman VL (2020) Iodine immobilization by materials through sorption and redox-driven processes: A literature review. Sci Total Environ 716:132820
Haq Z, Bancroft GM, Fyfe WS, Bird G, Lopata VJ (1980) Sorption of iodide on copper. Environ Sci Technol 14:1106–1110
Lefèvre G, Bessie J, Ehrhardt JJ, Walcarius A (2003) Immobilization of iodide on copper (I) sulfide minerals. J Environ Radioact 70:73–83
Lefèvre G, Walcarius A, Ehrhardt JJ, Bessie J (2000) Sorption of iodide on cuprite (Cu2O). Langmuir 16:4519–4527
Mao P, Jiang JL, Pan YH, Duanmu CS, Chen SW, Yang Y, Zhang SL, Chen YH (2018) Enhanced uptake of iodide from solutions by hollow Cu-Based adsorbents. Materials (Basel) 769:1–11
He XM, Li C, **ao X, Gan Y, Yu JP, Luo JY, Dan H, Wang YJ, Ding Y, Duan T (2023) Improved utilization of Cu0 for efficient adsorption of iodine in gas and solution by mesoporous Cu0-SBA-15 via solvothermal reduction method. Chem Eng J 462:142175
Zhang XY, Gu P, Li XY, Zhang GH (2017) Efficient adsorption of radioactive iodide ion from simulated wastewater by nano Cu2O/Cu modified activated carbon. Chem Eng J 322:129–139
Zhao XY, Gu P, Zhou SS, Li XY, Zhang GH, Dong LH (2018) Enhanced removal of iodide ions by nano Cu2O/Cu modified activated carbon from simulated wastewater with improved countercurrent two-stage adsorption. Sci Total Environ 626:612–620
Tang HM, Zhang MZ, Ran FY, Sun SY, Golubev YA, Zhao Q, Lin S, Duan T, Kotova OB (2023) Efficient immobilization of iodide from aqueous solution by Cu2O/Cu modifed shungite. J Radioanal Nucl Ch 332:3059–3068
Wang EC, Chen L, He XM, **ang YC, Jia SY, Chen YS, Guo ZX, Luo JY, Dan H, Ding Y, Duan T (2023) Capture of iodine gas by Bi-based composites derived from rice husk: Influence of the type of support on the iodine adsorption and retention. Chem Eng J 465:143069
**ong YW, Ai YR, Wang HX, Wang EC, Dan H, Ding Y (2023) High capacity adsorption of iodine gas by Ag0@C/SiO2 derived from rice husk: synergistic effect between C/SiO2 supports and Ag0 sites. J Radioanal Nucl Ch 332:3059–3068
Azambre B, Chebbi M, Leroy O, Cantrel L (2018) Effects of zeolitic parameters and irradiation on the retention properties of silver zeolites exposed to molecular iodine. Ind Eng Chem Res 57:1468–1479
Jiang M, Zhu L, Zhao Q, Chen GY, Wang ZR, Zhang JJ, Zhang L, Lei JH, Duan T (2022) Novel synthesis of NaY-NH4F-Bi2S3 composite for enhancing iodine capture. Chem Eng J 443:136477
Chee T-S, Tian Z, Zhang Q, Lei L, **ao C (2020) Efficient capture of radioactive iodine by a new bismuth-decorated electrospinning carbon nanofiber. J Nucl Mater 542:152526
Zhu H, Wang B, Zhu WK, Duan T, He GQ, Wei Y, Sun D, Zhou J (2022) Interface assembly of specific recognition gripper wrap** on activated collagen fiber for synergistic capture effect of iodine. Colloid Surface B 210:112216
Geng T, Ye S, Zhu Z, Zhang W (2018) Triazine-based conjugated microporous polymers with N,N,N′,N′-tetraphenyl-1, 4-phenylenediamine, 1,3,5-tris (diphenylamino) benzene and 1,3,5-tris [(3-methylphenyl)-phenylamino] benzene as the core for high iodine capture and fluorescence sensing of o-nitrophenol. J Mater Chem A 6:2808–2816
Li P, Zhao TJ, Zhou JH, Sui ZJ, Dai YC, Yuan WK (2005) Characterization of carbon nanofiber composites synthesized by sha** process. Carbon 43:2701–2710
Kolay S, Sahu AK, Jha P, Sharma RK, Jagannath MR (2023) Capture of volatile I2 by dithioglycol functionalized HKUST-1 and its polymeric composite beads. J Solid State Chem 324:124080
Mao P, Liu Y, Liu XD, Wang YC, Liang J, Zhou QH, Dai YX, Jiao Y, Chen SW, Yang Y (2017) Bimetallic AgCu/Cu2O hybrid for the synergetic adsorption of iodide from solution. Chemosphere 180:317–325
Cheng M, Geng JX, Wang YJ, Sun LX, Dou Q, Fu HY (2023) Cuprous oxide-loaded AlPO4-5 for highly efficient iodide ions adsorption in chloride molten salt. Micropor Mesopor Mater 359:112664
Zhang LQ, Zhang MJ, Guo HL, Tian ZH, Ge LF, He GJ, Huang JJ, Wang JT, Liu TX, Parkin IP, Lai FL (2022) A Universal polyiodide regulation using quaternization engineering toward high value-added and ultra-stable zinc-iodine batteries. Adv Sci 11:11
Tian SH, Yi ZD, Chen JQ, Fu SY (2023) In situ growth of UiO-66-NH2 in wood-derived cellulose for iodine adsorption. J Hazard Mater 443:130236
Cao DW, Nasori N, Wang ZJ, Wen LY, Xu R, Mi Y, Lei Y (2016) Facile surface treatment on Cu2O photocathodes for enhancing the photoelectrochemical response. Appl Catal B: Environ 198:398–403
Zhou J, Qi C, Li T, Lan T, Bai P, Liu F, Yuan Z, Zheng W, Yan W, Yan T (2022) Porous copper-loaded zeolites for high-effciency capture of iodine from spent fuel reprocessing off-Gas. Inorg Chem 61:7746–7753
Sharma B, Rabinal MK (2013) Ambient synthesis and optoelectronic properties of copper iodide semiconductor nanoparticles. J Alloy Compd 556:198–202
Lin L, Guan HD, Zou DL, Dong ZJ, Liu Z, Xu FF, **e ZG, Li YX (2017) A pharmaceutical hydrogen-bonded covalent organic polymer for enrichment of volatile iodine. Rsc Adv 7:54407
Dan H, Ding Y, Wang EC, Yang WP, He XM, Li C, **an Q, Yi FC, Zhu WK (2020) Manganese dioxide-loaded mesoporous SBA-15 silica composites for effective removal of strontium from aqueous solution. Environ Res 191:110040
Yu J, Song L, Wang Y (2023) Three novel indole-bearing porous organic polymers for efficient iodine capture from both vapor and organic phases. New J Chem 47:18070–18085
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare that they have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Wang, Y., Zhang, X. Convenient synthesis of novel Cu2O/Cu@C/SiO2 composite from rice-husk for efficient iodine gas capture. J Radioanal Nucl Chem (2024). https://doi.org/10.1007/s10967-024-09561-z
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10967-024-09561-z