Abstract
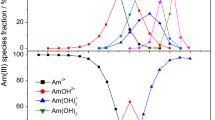
The predominant mineral, montmorillonite, in smectite-rich clays, the most promising buffer & backfill, controls their sorption characteristics towards radionuclides. The role of sulphate on Am(III) sorption by Na-montmorillonite has been investigated. The influence of pH, ionic strength & \({\text{[SO}}_{{4}}^{{2 - }} {]}\) on Am(III) sorption by Na-montmorillonite has been studied. The contribution of Am(III) hydroxo complexes in aqueous speciation is ~ 50% at pH 7.5 while that of Eu(III) hydroxo complexes is ~ 10%. In contrast to Eu(III), Am(III) sorption could be modeled by either ≡SOAmOH+ or ≡SOAmCO3. Log K for Am(III) ion exchange & ≡SOAmCO3 is less than that for similar Eu(III) species.









Similar content being viewed by others
References
Schwyn B, Wersin P, Ruedi J, Schneider J, Altmann S, Missana T, Noseck U (2012) FUNMIG integrated project results and conclusions from a safety case perspective. Appl Geochem 27:501–515
Missana T, Alonso U, Garcia-Gutierrez M, Mingarro M (2008) Role of bentonite colloids on europium and plutonium migration in a granite fracture. Appl Geochem 23:1484–1497
Lutzenkirchen J (2012) Summary of studies on (ad)sorption as a ‘“well-established”’ process within FUNMIG activities. Appl Geochem 27:427–443
Bradbury MH, Baeyens B (2005) Modelling the sorption of Mn(II), Co(II), Ni(II), Zn(II), Cd(II), Eu(III), Am(III), Sn(IV), Th(IV), Np(V) and U(VI) on montmorillonite: linear free energy relationships and estimates of surface binding constants for some selected heavy metals and actinides. Geochim Cosmochim Acta 69:875–892
Rabung Th, Pierret MC, Bauer A, Geckeis H, Bradbury MH, Baeyens B (2005) Sorption of Eu(III)/Cm(III) on Ca-montmorillonite and Na-illite. Part 1: batch sorption and time-resolved laser fluorescence spectroscopy experiments. Geochim Cosmochim Acta 69:5393–5402
Wang X, Sun Y, Alsaedi A, Hayat T, Wang X (2015) Interaction mechanism of Eu(III) with MX-80 bentonite studied by batch, TRLFS and kinetic desorption techniques. Chem Eng J 264:570–576
Adebowale KO, Unuabonah IE, Olu-Owolabi BI (2005) Adsorption of some heavy metal ions on sulfate- and phosphate-modified kaolin. Appl Clay Sci 29:145–148
Zhu J, Cozzolino V, Fernandez M, Torres Sánchez RM, Pigna M, Huang Q, Violante A (2011) Sorption of Cu on a Fe-deformed montmorillonite complex: effect of pH, ionic strength, competitor heavy metal, and inorganic and organic ligands. Appl Clay Sci 52:339–344
Olu-Owolabi BI, Unuabonah EI (2010) Kinetics and thermodynamics of the removal of Zn2+ and Cu2+ from aqueous solution by sulphate and phosphate modified bentonite clay. J Hazad Mat 184:731–738
Fernandes MM, Baeyens B, Dahn R, Scheinost AC, Bradbury MH (2012) U(VI) sorption on montmorillonite in the absence and presence of carbonate: a macroscopic and microscopic study. Geochim Cosmochim Acta 93:262–277
Fernandes MM, Scheinost AC, Baeyens B (2016) Sorption of trivalent lanthanides and actinides onto montmorillonite: macroscopic, thermodynamic and structural evidence for ternary hydroxo and carbonato surface complexes on multiple sorption sites. Water Res 99:74–82
Troyera LD, Maillot F, Wang Z, Mehta VS, Giammar DE, Catalano JG (2016) Effect of phosphate on U (VI) sorption to montmorillonite: ternary complexation and precipitation barrier. Geochim Cosmochim Acta 175:86–99
Bachmaf S, Planer-Friedrich B, Merkel BJ (2008) Effect of sulphate, carbonate, and phosphate on the uranium(VI) sorption behavior onto bentonite. Radiochim Acta 96:359–366
Fernandes MM, Baeyens B, Bradbury MH (2008) The influence of carbonate complexation on lanthanide/actinide sorption on montmorillonite. Radiochim Acta 96:691–697
Chen Z, ** Q, Guo Z, Montavon G, Wu W (2014) Surface complexation modeling of Eu(III) and phosphate on Na-bentonite: binary and ternary adsorption systems. Chem Eng J 256:61–68
Patel MA, Kar AS, Kumar S, Tomar BS (2017) Effect of phosphate on sorption of Eu(III) by montmorillonite. J Radioanal Nucl Chem 313:537–545
Liu X, Simunek J, Li L, He J (2013) Identification of sulfate sources in groundwater using isotope analysis and modeling of flood irrigation with waters of different quality in the **ghuiqu district of China. Environ Earth Sci 69:1589–1600
Samborska K, Halas S, Bottrell SH (2013) Sources and impact of sulphate on groundwaters of Triassic carbonate aquifers, Upper Silesia. Pol J Hydrol 486:136–150
Ding M, Kelkar S, Meijer A (2014) Surface complexation modeling of americium sorption onto volcanic tuff. J Env Radioactiv 136:181–187
Kumar S, Pente AS, Bajpai RK, Kaushik CP, Tomar BS (2013) Americium sorption on smectite-rich natural clay from granitic groundwater. Appl Geochem 35:28–34
Patel MA, Kar AS, Kumar S, Das MK, Raut VV, Tomar BS (2019) Effect of sulfate on sorption of Eu(III) by Na-montmorillonite. Radiochim Acta 107(2):115–128
Bradbury MH, Baeyens B (2002) Sorption of Eu on Na- and Ca-montmorillonite: experimental investigations and modelling with cation exchange and surface complexation. Geochim Cosmochim Acta 66:2325–2334
Herbelin AL, Westall JC (1999) FITEQL—a computer program for determination of chemical equilibrium constant from experimental data. Department of chemistry. Oregon State University, Oregon, pp. 97331
Gustafsson JP (2014) Visual MINTEQ ver. 3.1.
Bradbury MH, Baeyens B (2006) Modelling sorption data for the actinides Am(III), Np(V) and Pa(V) on montmorillonite. Radiochim Acta 94:619–625
Coppin F, Berger G, Bauer A, Castet S, Loubet M (2002) Sorption of lanthanides on smectite and kaolinite. Chem Geol 182:57–68
Bradbury MH, Baeyens B (2009) Sorption modelling on illite Part II: actinide sorption and linear free energy relationships. Geochim Cosmochim Acta 73:1004–1013
Dzombak DA, Morel FM (1990) Surface complexation modeling: hydrous ferric hydroxide. Wiley-Interscience, New York
Choppin GR, Jensen MP (2006) Actinides in solution: complexation and kinetics In: Morss LR, Edelstein NK and Fuger J (eds) In: The chemistry of actinide and transactinide elements. Springer, Dordrecht
Kielland J (1937) Individual activity coefficients of ions in aqueous solutions. J Am Chem Soc 59:1675–1678
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors have no competing interests to declare that are relevant to the content of this article.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Kar, A.S., Patel, M.A. & Tomar, B.S. Retention of Am(III) by montmorillonite: effect of sulphate. J Radioanal Nucl Chem 332, 3069–3077 (2023). https://doi.org/10.1007/s10967-023-09014-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10967-023-09014-z