Abstract
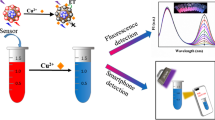
A naked eye and fluorescence turn-on 1,8-naphtahlimide based chemosensor,1, possessing Schiff base linkage was utilized for the rapid detection of highly toxic triphosgene. The proposed sensor selectively detected triphosgene over various other competitive analytes including phosgene with the detection limit of 6.15 and 1.15 µM measured using UV-vis and fluorescence spectrophotometric techniques, respectively. Colorimetric changes observed in solution phase were processed by image analysis using smartphone leading to on-site and inexpensive determination of triphosgene. Further, solid phase sensing of triphosgene was carried out by 1 loaded PEG membranes and silica gel.








Similar content being viewed by others
Data Availability
The data provided in the manuscript is original and will be made available at any time on request basis.
Code Availability
Not Applicable.
References
Zhang H, Rudkevich DM (2007) A FRET approach to phosgene detection. Chem Commun 1238–1239:1238. https://doi.org/10.1039/B614725A
Joy A, Anim-Danso E, Kohn J (2009) Simple, rapid, and highly sensitive detection of diphosgene and triphosgene by spectrophotometric methods. Talanta 80:231–235. https://doi.org/10.1016/j.talanta.2009.06.059
Chu H, **e Q, Zhao X, Xu Z, Geng S, Liu C, Zhou X, Yang L, Han W, Zhou J (2022) Selective and visual detection of triphosgene by a red turn-on fluorescent probe. Sens Actuators B Chem 371:132557. https://doi.org/10.1016/j.snb.2022.132557
Cotarca L, Geller T, Répási J (2017) Bis(trichloromethyl)carbonate (BTC, Triphosgene): A Safer Alternative to Phosgene? Org Process Res Dev 21:1439–1446. https://doi.org/10.1016/j.talanta.2009.06.059
Fu YL, Chong YY, Li H, Feng W, Song QH (2021) Sensitive and visual detection of phosgene by a TICT-based BODIPY dye with 8-(o-hydroxy)aniline as the active site. Chem Eur J 27:4977–4984. https://doi.org/10.1002/chem.202005169
Li ZJ, Zhang WJ, Bi WZ, Ma QJ, Feng SX, Chen XL, Qu LB (2021) An amino-substituted 2-(2′ -hydroxyphenyl)benzimidazole for the fluorescent detection of phosgene based on an ESIPT mechanism. RSC Adv 11:10836–10841. https://doi.org/10.1039/D1RA00811K
Wang SL, Li C, Song QH (2019) Fluorescent Chemosensor for Dual-Channel discrimination between Phosgene and Triphosgene. Anal Chem 91:5690–5697. https://doi.org/10.1021/acs.analchem.8b05777
Sun Y, Zhou X, Sun L, Zhao X, He Y, Gao G, Han W, Zhou J (2022) Lysosome targeting red fluorescent probe for broad carboxylesterases detection in breast cancer cells. Chin Chem Lett 33:4229–4232. https://doi.org/10.1016/j.cclet.2022.01.087
Zhu L, Lin W (2022) Constructing a NIR fluorescent probe for ratiometric imaging viscosity in mice and detecting blood viscosity in folliculitis mice and peritonitis mice. Sens Actuators B Chem 352:131042. https://doi.org/10.1016/j.snb.2021.131042
Li Z, Gao J, Guo Z, Zhao H, Liu L, Wang M, Zhang P, Chen G, Li X, Wei C (2022) Monitoring the fluctuation of H2S in insulin-resistant HepG2 cells and diabetic mice with a dual-locked NIR fluorescent probe. Sens Actuators B Chem 353:131141. https://doi.org/10.1016/j.snb.2021.131141
Zhang C, **e H, Zhan T, Zhang J, Chen B, Qian Z, Zhang G, Zhang W, Zhou J (2019) A new mitochondrion targetable fluorescent probe for carbon monoxide-specific detection and live cell imaging. Chem Commun 55:9444–9447. https://doi.org/10.1039/C9CC03909K
Wang SL, Zhong L, Prof Song QH (2018) sensitive and selective detection of Phosgene, Diphosgene, and Triphosgene by a 3,4-Diaminonaphthalimide in solutions and the gas phase. Chem Eur J 24:5652–5658. https://doi.org/10.1002/chem.201800051
Zhou S, Rong Y, Wang H, Liu X, Wei L, Song X (2018) A naphthalimide-indole fused chromophore-based fluorescent probe for instantaneous detection of thiophenol with a red emission and a large Stokes shift. Sens Actuators B Chem 276:136–141. https://doi.org/10.1016/j.snb.2018.08.096
Chung J, Li H, Lim CS, Kim HM, Yoon J (2020) Two-photon imaging of hydrogen polysulfides in living cells and hippocampal tissues. Sens Actuators B Chem 322:128564. https://doi.org/10.1016/j.snb.2020.128564
Du B, Zhang FH, Cao W, Wang PH, Li ZJ, Ding ZJ (2018) A naphthalimide-based fluorescent turn-on sensor for the selective detection of diethyl chlorophosphate. Chem Select 3:13470–13473. https://doi.org/10.1002/slct.201800544
Oshchepkov AS, Oshchepkov MS, Oshchepkova MV, Al-Hamry A, Kanoun O, Kataev EA (2021) Naphthalimide-based fluorescent polymers for molecular detection. Adv Opt Mater 9:2001913. https://doi.org/10.1002/adom.202001913
Qin JC, Yan J, Wang BD, Yang ZY (2016) Rhodamine–naphthalene conjugate as a novel ratiometric fluorescent probe for recognition of Al3+. Tetrahedron Lett 57:1935–1939. https://doi.org/10.1016/j.tetlet.2016.03.074
Li X, Zhou Y, Zhang Y, Son YA (2015) Investigation of a Naphthalimide Based OH– Sensor with Quinoline attached. Mol CrystLiqCryst 622:84–93. https://doi.org/10.1080/15421406.2015.1097004
Meng Q, Jia H, Gao X, Wang Y, Zhang R, Wang R, Zhang Z (2015) Reversible and selective fluorescence detection of histidine using a Naphthalimide-Based Chemosensing Ensemble. Chem Asian J 10:2411–2418. https://doi.org/10.1002/asia.201500690
Gu P, Xu X, Zhou F, Zhao T, Ye G, Liu G, Xu Q, Ge J, Xu Q, Lu J (2014) Study of Linear and Nonlinear Optical Properties of four derivatives of substituted Aryl Hydrazones of 1,8-Naphthalimide. Chin J Chem 32:205–211. https://doi.org/10.1002/cjoc.201300842
Zeng L, Zeng H, Jiang L, Wang S, Hou JT, Yoon J (2019) A single fluorescent chemosensor for simultaneous discriminative detection of Gaseous Phosgene and a nerve Agent Mimic. Anal Chem 91:12070–12076. https://doi.org/10.1021/acs.analchem.9b03230
Xu H, Zhang H, Zhao L, Peng C, Liu G, Cheng T (2020) Naphthalimide-based fluorescent probe for highly sensitive and selective detection of nerve agent mimic DCP in solution and vapor phase. New J Chem 44:10713–10718. https://doi.org/10.1039/D0NJ00416B
Benesi HA, Hildebrand JH (1949) A spectrophotometric investigation of the interaction of iodine with aromatic hydrocarbons. J Am Chem Soc 71:2703–2707. https://doi.org/10.1021/ja01176a030
Long GL (1983) Limit of detection. A closer look at the IUPAC definition. AnalChem 55:712A. https://doi.org/10.1021/ac00258a724
Kaur B, Gupta A, Kaur N (2020) A simple Schiff base as a multi responsive and sequential sensor towards Al3+, F– and Cu2+ ions. J PhotochemPhotobiol A 389:112140. https://doi.org/10.1016/j.jphotochem.2019.112140
Liu C, Xu J, Yang F, Zhou W, Li Z, Wei L, Yu M (2015) Nanomolar Cu2+ and F– naked-eye detection with a 1,8-naphthalimide-based colorimetric probe. Sens Actuators B Chem 212:364–370. https://doi.org/10.1016/j.snb.2015.02.010
Yadav P, Laddha H, Agarwal M, Gupta R (2021) Colorimetric assay of fluoride goes digital: on the spot testing of F– ions in water using smartphone’s digital imaging and test strip assay by a novel chromofluorogenic receptor based on 1,8-naphthalimide. J Mol Liq 324:114690. https://doi.org/10.1016/j.molliq.2020.114690
Kaur B, Gupta A, Kaur N (2020) A novel, anthracene-based naked eye probe for detecting Hg2+ ions in aqueous as well as solid state media. Microchem J 153:104508. https://doi.org/10.1016/j.microc.2019.104508
Acknowledgements
The authors are grateful to SAIF, Panjab University, Chandigarh for recording the NMR spectra and mass spectral studies.
Funding
The authors are greatly obliged to DST INSPIRE(IF200020)for the financial assistance.
Author information
Authors and Affiliations
Contributions
Nisha Jain: Conceptualization, Data curation, Formal analysis, Visualization, Writing-original draft. Navneet Kaur: Methodology, Writing-review & editing, Supervision, Project administration.
Corresponding author
Ethics declarations
Ethics Approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Consent to Participate
The authors declare that they consent to participate.
Consent for Publication
The authors provide consent for the publication.
Conflicts of Interest
The authors have no conflicts to declare.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Jain, N., Kaur, N. A Naked Eye and Fluorescence Turn-on Sensor for Smartphone Assisted Solid and Solution Phase Sensing of Highly Toxic Triphosgene. J Fluoresc 34, 935–943 (2024). https://doi.org/10.1007/s10895-023-03328-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10895-023-03328-7