Abstract
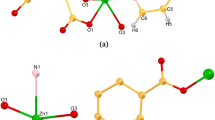
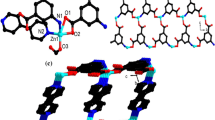
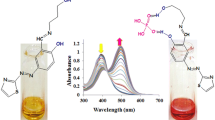
In this research, Zn- or Cd-based metal–organic frameworks (coded ZnMOF-1 and CdMOF-1) containing benzene-1,4-dicarboxylic acid (H2bdc) and pyridyl-based Schiff base (4-pyridylcarboxaldehydeisonicotinoylhydrazone (L)) dual ligands were successfully assembled via a conventional solvothermal method. The photoluminescence quenching response of ZnMOF-1 and CdMOF-1 and their sensing sensitivity and selectivity towards various inorganic anions were evaluated in aqueous media. Crystallographic and thermogravimetric studies confirm the formation of both MOFs with good crystallinity and thermal stability. Photoluminescence studies also verify the selectivity of ZnMOF-1 and CdMOF-1 for efficient sensing of inorganic oxyanions (like chromate/dichromate: CrO42− and Cr2O72−). Further, it was noted that only chromate/dichromate (CrO42−/Cr2O72−) anions showed a significant turn-off quenching effect while other anions (like F−, Br−, I−, Cl−, ClO4−, SCN−, SO42−, NO3−, and NO2−) have a low/negligible effect on the photoluminescence intensity of both MOFs. The limit of detection (LOD) of chromate/dichromate by ZnMOF-1 and CdMOF-1 was 9.79/10.94 µM and 2.68/1.48 µM, respectively. A probable mechanism for turn-off quenching response towards chromate and dichromate anions could be attributed to the spectral overlap of both excitation and emission spectra of ZnMOF-1/CdMOF-1 with the absorption spectra of chromate/dichromate anions. As a result, the energy transfer from ZnMOF-1 or CdMOF-1 to the target chromate and dichromate anions decreased fluorescence intensity (i.e., fluorescence quenching effect).
Graphical Abstract
















Similar content being viewed by others
Availability of Data and Material/ Data Availability (Data Transparency, If Link Please Provide the Link to Access)
Data and material information is provided and will be shared on request.
Code Availability (Software Application or Custom Code)
This is not applicable.
References
Vikrant K, Tsang DC, Raza N, Giri BS, Kukkar D, Kim K-H (2018) Potential utility of metal–organic framework-based platform for sensing pesticides. ACS Appl Mater Interfaces 10(10):8797–8817
Zhang Y, Yuan S, Day G, Wang X, Yang X, Zhou H-C (2018) Luminescent sensors based on metal-organic frameworks. Coord Chem Rev 354:28–45
Farahani YD, Safarifard V (2019) A luminescent metal-organic framework with pre-designed functionalized ligands as an efficient fluorescence sensing for Fe3+ ions. J Solid State Chem 270:428–435
Liu C, Yan B (2016) A novel photofunctional hybrid material of pyrene functionalized metal-organic framework with conformation change for fluorescence sensing of Cu2+. Sens Actuators B Chem 235:541–546
Zhang W, Tang Y, Shi A, Bao L, Shen Y, Shen R, Ye Y (2018) Recent developments in spectroscopic techniques for the detection of explosives. Materials 11(8):1364
Farahani YD, Safarifard V (2019) Highly selective detection of Fe3+, Cd2+ and CH2Cl2 based on a fluorescent Zn-MOF with azine-decorated pores. J Solid State Chem 275:131–140
Parmar B, Rachuri Y, Bisht KK, Suresh E (2017) Mixed-ligand LMOF fluorosensors for detection of Cr (VI) oxyanions and Fe3+/Pd2+ cations in aqueous media. Inorg Chem 56(18):10939–10949
Kaur M, Kumar S, Younis SA, Yusuf M, Lee J, Weon S, Kim K-H, Malik AK (2021) Post-synthesis modification of metal-organic frameworks using schiff base complexes for various catalytic applications. Chem Eng J 423:130230
Pamei M, Puzari A (2019) Luminescent transition metal–organic frameworks: An emerging sensor for detecting biologically essential metal ions. Nano-Struct Nano-Objects 19:100364
Razavi SAA, Morsali A (2020) Metal ion detection using luminescent-MOFs: Principles, strategies and roadmap. Coord Chem Rev 415:213299
Parmar B, Bisht KK, Rachuri Y, Suresh E (2020) Zn (II)/Cd (II) based mixed ligand coordination polymers as fluorosensors for aqueous phase detection of hazardous pollutants. Inorg Chem Front 7(5):1082–1107
Kaur M, Kumar S, Yusuf M, Lee J, Brown RJ, Kim K-H, Malik AK (2021) Post-synthetic modification of luminescent metal-organic frameworks using schiff base complexes for biological and chemical sensing. Coord Chem Rev 449:214214
Fan L, Zhao D, Li B, Wang F, Deng Y, Peng Y, Wang X, Zhang X (2022) Luminescent binuclear zinc (II) organic framework as bifunctional water-stable chemosensor for efficient detection of antibiotics and Cr (VI) anions in water. Spectrochim Acta A: Mol Biomol Spectrosc 264:120232
Parmar B, Rachuri Y, Bisht KK, Suresh E (2016) Syntheses and structural analyses of new 3D isostructural Zn (II) and Cd (II) luminescent MOFs and their application towards detection of nitroaromatics in aqueous media. ChemistrySelect 1(19):6308–6315
Parmar B, Rachuri Y, Bisht KK, Laiya R, Suresh E (2017) Mechanochemical and conventional synthesis of Zn (II)/Cd (II) luminescent coordination polymers: dual sensing probe for selective detection of chromate anions and TNP in aqueous phase. Inorg Chem 56(5):2627–2638
Acknowledgements
The authors are thankful to the Chemistry Department, Punjabi University, Patiala, for providing lab and instrument facilities. One of the author Manpreet kaur is highly obliged to the UGC, New Delhi, India, for UGC-SRF fellowship.
Funding
One of the authors, Manpreet kaur is grateful to University Grants Commission (UGC), New Delhi, India, for providing senior research fellowship.
Author information
Authors and Affiliations
Contributions
Manpreet Kaur: Performed the experimentation. Ashok Kumar Malik and Mohamad Yusuf: Writing and checking the paper.
Corresponding author
Ethics declarations
Ethics Approval/Declarations (Include Appropriate Approvals or Waivers)
There are no ethic approvals required for this research work.
Consent to Participate (Include Appropriate Statements)
All authors have participated in the revision of the manuscript.
Consent for Publication (include Appropriate Statements)
All authors agree for the publication.
Conflicts of Interest/Competing Interests (include Appropriate Disclosures)
All the authors declare that there is no conflict of interest.
Competing Interests
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Highlights
• Two metal–organic frameworks (ZnMOF-1 and CdMOF-1) associated with pyridyl Schiff-base ligand were successfully synthesized.
• Both MOFs displayed a good photoluminescent recognition ability for oxyanions (like chromate and dichromate) in the aqueous phase.
• Competetive energy absorption and FRET mechanisms explained the fluorescence quenching effect of both MOFs.
• The prepared Zn or Cd-MOFs exhibited excellent sensing recyclability for at least three cycles.
Supplementary Information
Below is the link to the electronic supplementary material.
10895_2022_3036_MOESM1_ESM.docx
Supplementary file1 (DOCX 906 KB) FT-IR, PL spectra, SV plots, quenching percentage graph, UV−vis spectra, LOD calculations, comparison table for chromate and dichromate anions.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Kaur, M., Yusuf, M. & Malik, A.K. Schiff Base-functionalized Metal–organic Frameworks for Selective Sensing of Chromate and Dichromate in Water. J Fluoresc 33, 61–75 (2023). https://doi.org/10.1007/s10895-022-03036-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10895-022-03036-8