Abstract
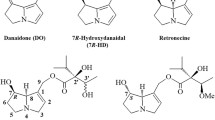
Ithomiine butterflies use pyrrolizidine alkaloids (PAs) as precursors for male pheromones, such as dihydropyrrolizines or lactones. In contrast to most other ithomiine genera, none of these compounds have ever been detected in Oleria species. The absence of these compounds is thought to be the result of limited access to PA-containing plants. Here we investigate the contents of the androconia of Oleria onega caught in the wild when PA containing plants were abundant. Although the PA lycopsamine was detected in the hairpencils, none of the other known PA-derived compounds were present. Instead, the unsubstituted core of the PA necine base, 1-methylene-1H-pyrrolizine (13), a very unstable compound, was found. The identity of this compound was proven by synthesis. Although its formation in nature appears very likely, 13 is also formed during GC analysis of PAs, making its natural occurrence uncertain. Nevertheless, its reactivity makes it a good candidate for a signaling compound, because its rapid degradation can be used to convey spatial and temporal information. In addition, several other compounds, likely used in intraspecific communication, were identified. All of these compounds are reported for the first time as natural products or from insects. These include 9-hydroxynonanoic acid (21) and (Z)-9-hydroxy-6-enoic acid (18), as well as their condensation products with 11-hexadecenoic- and octadecenoic acids. Furthermore, self-condensation products, such as (Z)-9-[(9-hydroxynon-6-enoyl)oxy]- and 9-[(9-hydroxynonanoyl)oxy]nonanoic acid and non-6-enoic acids (35, 36, 38, 40) were identified, together with the known compounds 2-heptadecanol (39) and 6,10,14-trimethylpentadecan-2-ol (37). In summary, O. onega appears to lack enzymes to produce dihydropyrrolizines. In stark contrast to other ithomiine genera, a unique blend of oxidized fatty acids seems to be used instead.










Similar content being viewed by others
References
Ayasse M, Schiestl FP, Paulus HF, Ibarra F, Francke W (2003) Pollinator attraction in a sexually deceptive orchid by means of unconventional chemicals. Proc R Soc B Biol Sci 270:517–522
Bogner F, Boppré M (1989) Single cell recordings reveal hydroxydanaidal as the volatile compound attracting insects to pyrrolizidine alkaloids. Entomol Exp Appl 50:171–184
Boppré M (1986) Insects Pharmacophagously utilizing defensive plant chemicals (pyrrolizidine alkaloids). Naturwissenschaften 73:17–26
Brandänge S, Lundin C (1971) A synthesis of endo- and exo-1-ethoxycarbonyl-pyrrolizidine. Acta Chem Scand 25:2447–2450
Chazot N, Willmott KR, Lamas G, Freitas AVL, Piron-Prunier F, Arias CF, Mallet J, De-Silva DL, Elias M (2019) Renewed diversification following Miocene landscape turnover in a Neotropical butterfly radiation. Early online, Global Ecol Biogeogr 8:1118–1132
Cossy J, Pete JP (1986) A one step synthesis of ω-hydroxyacetylenic carboxylic acids. Tetrahedron Lett 27:573–574
Dasmahapatra KK, Lamas G, Simpson F, Mallet J (2010) The anatomy of a ‘suture zone’ in Amazonian butterflies: a coalescent-based test for vicariant geographic divergence and speciation. Mol Ecol 19:4283–4301
De-Silva DL, Vásquez AS, Mallet J (2011) Selection for enemy-free space - eggs placed away from the host plant increase survival of a neotropical ithomiine butterfly. Ecol Entomol 36:667–672
Edgar JA, Culvenor CCJ, Pliske TE (1976) Isolation of a lactone, structurally related to the esterifying acids of pyrrolizidine alkaloids, from the coastal fringes of male Ithomiinae. J Chem Ecol 2:263–270
Eisner T, Meinwald J (1995) The chemistry of sexual selection. Proc Natl Acad Sci U S A 92:50–55
Fashe MM, Juvonen RO, Petsalo A, Rahnasto-Rilla M, Auriola S, Soininen P, Vepsäläinen J, Pasanen M (2014) Identification of a new reactive metabolite of pyrrolizidine alkaloid Retrorsine: (3H-Pyrrolizin-7-yl)methanol. Chem Res Toxicol 27:1950–1957
Hedenström E, Wallin EA, Andersson J, Bång J, Wang H-L, Löfstedt C, Brattström O, Baquet P (2014) Stereoisomeric analysis of 6,10,14-Trimethylpentadecan-2-ol and the corresponding ketone in wing extracts from African Bicyclus butterfly species. J Chem Ecol 41:44–51
Honda Y, Honda K, Omura H (2006) Major components in the hairpencil secretion of a butterfly, Euploea mulciber (Lepidoptera, Danaidae): their origins and male behavioral responses to pyrrolizidine alkaloids. J Insect Physiol 52:1043–1053
Jefford CW, Thornton SR, Sienkiewicz K (1994) An enantiospecific entry to indolizidines by intramolecular acylation of N-pyrrole esters. Tetrahedron Lett 35:3905–3908
Johnson F, Paul KG, Favara D (1982) An efficient synthesis of methyl dl-cis-jasmonate. J Org Chem 47:4254–4255
Jurenka R (2004) Insect pheromone biosynthesis. The chemistry of pheromones and other Semiochemicals I, Top Curr Chem 239. Springer, Berlin
Kim S-U, Kim K-R, Kim J-W, Kim S, Kwon Y-U, Oh D-K, Park J-B (2015) Microbial synthesis of plant oxylipins from γ-linolenic acid through designed biotransformation pathways. J Agric Food Chem 63:2773–2781
Kim S, Kang S, Kim G, Lee Y (2016) Copper-catalyzed aza-michael addition of aromatic amines or aromatic aza-heterocycles to α,β-unsaturated Olefins. J Org Chem 81:4048–4057
Kindl J, Jiros P, Kalinova B, Zacek P, Valterova I (2012) Females of the bumblebee parasite, Aphomia sociella, excite males using a courtship pheromone. J Chem Ecol 38:400–407
Lercker G, Capella P, Conte LS, Ruini F, Giordani G (1981) Components of royal jelly I. Identification of the organic acids. Lipids 16:912–919
Liénard MA, Wang H-L, Lassance J-M, Löfstedt C (2014) Sex pheromone biosynthetic pathways are conserved between moths and the butterfly Bicyclus anynana. Nat Commun 5:3957
Mann F (2016) Die Diversität von Pheromonen in Schmetterlingen der Familie Nymphalidae. PhD thesis. TU Braunschweig, Braunschweig
Mattocks AR, Jukes R, Brown J (1989) Simple procedures for preparing putative toxic metabolites of pyrrolizidine alkaloids. Toxicon 27:561–567
Nesbitt BF, Beevor PS, Cork A, Hall DR, David H, Nandagopal V (1986) The female sex pheromone of sugarcane stalk borer, Chilo auricilius Identification of four components and field tests. J Chem Ecol 12:1377–1388
Neuenschwander M (2015) Low-temperature olefin syntheses in view of parent Fulvenes and Fulvalenes. Helv Chim Acta 98:731–762
Nieberding CM, de Vos H, Schneider MV, Lassance J-M, Estramil N, Andersson J, Bång J, Hedenström E, Löfstedt C, Brakefield PM, Somers M (2008) The male sex pheromone of the butterfly Bicyclus anynana: towards an evolutionary analysis. PLoS One 3:e2751
Pankiw T, Huang ZY, Winston ML, Robinson GE (1998) Queen mandibular gland pheromone influences worker honey bee (Apis mellifera L.) foraging ontogeny and juvenile hormone titers. J Insect Physiol 44:685–692
Schulz S (1992) Absolute configuration and synthesis of 2-hydroxy-2-(1-hydroxyethyl)-3-methyl-g-butyrolactone, a presumed pheromone of Ithomiine Butterflies. Liebigs Ann Chem :829–834
Schulz S (1998) Insect-plant interactions --- metabolism of plant compounds to pheromones and allomones by Lepidoptera and leaf beetles. Eur J Org Chem 1998:13–20
Schulz S (2009) Alkaloid-derived male courtship pheromones. In: Conner WE (ed) Tiger moths and woolly bears: behavior, ecology, and evolution of the Arctiidae. Univ. Press, Oxford, pp 145–153
Schulz S, Francke W, Edgar JA, Schneider D (1988) Volatile compounds from Androconial organs of Danaine and Ithomiine butterflies. Z Naturforsch Sect C J Biosci 43c:99–104
Schulz S, Francke W, Boppré M, Eisner T, Meinwald J (1993) Insect pheromone biosynthesis: Stereochemical pathway of hydroxydanaidal production from alkaloidal precursors in Creatonotos transiens (Lepidoptera, Arctiidae). Proc Natl Acad Sci U S A 90:6834–6838
Schulz S, Beccaloni G, Brown KS Jr, Boppré M, AVL F, Ockenfels P, Trigo JR (2004) Semiochemicals derived from pyrrolizidine alkaloids in male ithomiine butterflies (Lepidoptera: Nymphalidae Ithomiinae). Biochem Syst Ecol 32:699–713
Trigo JR, Barata LES, Brown KS Jr (1994) Stereochemical inversion of pyrrolizidine alkaloids by Mechanitis polymnia (Lepidoptera: Nymphalidae: Ithomiinae): specificity and evolutionary significance. J Chem Ecol 20:2883–2899
Trigo JR, Brown KS Jr, Witte L, Hartmann T, Ernst L, LES B (1996) Pyrrolizidine alkaloids: different acquisition and use patterns in Apocynaceae and Solanaceae feeding ithomiine butterflies (Lepidoptera: Nymphalidae). Biol J Linn Soc 58:99–123
van den Dool H, Kratz P (1963) A generalization of the retention index system including linear temperature programmed gas-liquid partition chromatography. J Chromatogr 11:463–471
Wegener R (2002) Identifizierung und Synthese von Inhaltsstoffen aus Blattkäfern und Pflanzen mit biologischer Aktivität in tritrophischen Systemen. PhD thesis. TU Braunschweig, Braunschweig
Acknowledgements
We thank Stephanie Gallusser for the live Oleria onega janarilla photo and Céline Houssin for the Oleria onega photos in the SM. We thank the Peruvian authorities and Dr. Gerardo Lamas (Museo de Historia Natural, Universidad Mayor de San Marcos) for research permits (002-2015-SERFOR-DGGSPFFS) and PEHCBM-Area de Conservacion Regional Cordillera Escalera (023-2016/GRSM/PEHCBM/DMA/ACR-CE). We thank the Deutsche Forschungsgemeinschaft (DFG) (Schu 984/12-1) and the French National Agency for Research (ANR) through the project SPECREP (grant number ANR-14-CE02-0011 awarded to ME and employing MM as a postdoc) for financial support.
Author information
Authors and Affiliations
Corresponding author
Additional information
Patrick Stamm and Florian Mann contributed equally to this work.
Electronic supplementary material
ESM 1
(PDF 2948 kb)
Rights and permissions
About this article
Cite this article
Stamm, P., Mann, F., McClure, M. et al. Chemistry of the Androconial Secretion of the Ithomiine Butterfly Oleria onega. J Chem Ecol 45, 768–778 (2019). https://doi.org/10.1007/s10886-019-01100-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10886-019-01100-5