Abstract
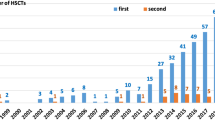
The results of hematopoietic stem cell transplant (HSCT) for primary immunodeficiency diseases (PID) have been improving over time. Unfortunately, develo** countries do not experience the same results. This first report of Brazilian experience of HSCT for PID describes the development and results in the field. We included data from transplants in 221 patients, performed at 11 centers which participated in the Brazilian collaborative group, from July 1990 to December 2015. The majority of transplants were concentrated in one center (n = 123). The median age at HSCT was 22 months, and the most common diseases were severe combined immunodeficiency (SCID) (n = 67) and Wiskott-Aldrich syndrome (WAS) (n = 67). Only 15 patients received unconditioned transplants. Cumulative incidence of GVHD grades II to IV was 23%, and GVHD grades III to IV was 10%. The 5-year overall survival was 71.6%. WAS patients had better survival compared to other diseases. Most deaths (n = 53) occurred in the first year after transplantation mainly due to infection (55%) and GVHD (13%). Although transplant for PID patients in Brazil has evolved since its beginning, we still face some challenges like delayed diagnosis and referral, severe infections before transplant, a limited number of transplant centers with expertise, and resources for more advanced techniques. Measures like newborn screening for SCID may hasten the diagnosis and ameliorate patients’ conditions at the moment of transplant.



Similar content being viewed by others
References
Picard C, Gaspar HB, Al-Herz W, Bousfiha A, Casanova JL, Chatila T, et al. International Union of Immunological Societies: 2017 primary Immunodeficiency Diseases Committee Report on Inborn Errors of Immunity. J Clin Immunol. 2018;38(1):96–128.
Gatti RA, Meuwissen HJ, Allen HD, et al. Immunological reconstitution of sex-linked lymphopenic immunological deficiency. Lancet. 1968;2:1366–9.
Bach FH, Albertini RJ, Joo P, et al. Bone-marrow transplantation in a patient with the Wiskott-Aldrich Syndrome. Lancet. 1968;2:1364–6.
Dvorak CD, Cowan MJ, Logan BR, Notarangelo LD, Griffith LM, Puck JM, et al. The Natural history of children with severe combined immunodeficiency: baseline features of the first fifty patients of the primary immune deficiency treatment consortium prospective study 6901. J Clin Immunol. 2013;33:1156–64.
Gennery AR, Slatter MA, Grandin L, Taupin P, Cant AJ, Veys P, et al. Transplantation of hematopoietic stem cells and long term survival for primary immunodeficiencies in Europe: Entering a new century, do we do better? J Allergy Clin Immunol. 2010;126:602–10.
Slack J, Albert MH, Balashov D, Belohradsky BH, Bertaina A, Bleesing J, et al. Outcome of hematopoietic cell transplantation for DNA double-strand break repair disorders. J Allergy Clin Immunol. 2017;S0091-6749(17):30567–5.
de la Morena MT, Leonard D, Torgerson TR, Cabral-Marques O, Slatter M, Aghamohammadi A, et al. Long-term outcomes of 176 patients with X-linked hyper-IgM syndrome treated with or without hematopoietic cell transplantation. J Allergy Clin Immunol. 2017;139(4):1282–92.
Griffith LM, Cowan MJ, Notarangelo LD, Kohn DB, Puck JM. Shearer et al. primary Immune Deficiency Treatment Consortium (PIDTC) update. J Allergy Clin Immunol. 2016;138(2):375–85.
Griffith LM, Cowan MJ, Notarangelo LD, Kohn DB, Puck JM, Pai SY, et al. Primary Immune Deficiency Treatment Consortium (PIDTC) report. J Allergy Clin Immunol. 2014;133(2):335–47.
Fernandes JF, Rocha V, Labopin M, Neven B, Moshous D, Gennery AR, et al. Transplantation in patients with SCID: mismatched related stem cells or unrelated cord blood? Blood. 2012;119(12):2949–55.
Leiva LE, Zelazco M, Oleastro M, Carneiro-Sampaio M, Condino-Neto A, Costa-Carvalho BT, et al. Primary immunodeficiency diseases in Latin America: the Second Report of the LAGID Registry. J Clin Immunol. 2007;27(1):101–8.
Condino-Neto A. The relevance of collaborative work: the Latin American Society for Immunodeficiencies (LASID) registry model. Clin Exp Immunol. 2014;178(Suppl 1):16–7.
Condino-Neto A, Sorensen RU, Gómez Raccio AC, King A, Espinosa-Rosales FJ, Franco JL. Current state and future perspectives of the Latin American Society for Immunodeficiencies (LASID). Allergol Immunopathol (Madr). 2015;43(5):493–7.
Espinosa-Rosales FJ, Condino-Neto A, Franco JL, Sorensen RU. Into action: improving access to optimum care for all primary immunodeficiency patients. J Clin Immunol. 2016;36(5):415–7.
Oliveira-Junior EB, Zurro NB, Prando C, Cabral-Marques O, Pereira PVS, Schimke LF, et al. Clinical and genotypic spectrum of chronic granulomatous disease in 71 Latin American patients: first report from the LASID Registry. Pediatr Blood Cancer. 2015;62:2101–7.
Costa-Carvalho B, González-Serrano M, Espinosa-Padilla S, Segundo G. Latin American challenges with the diagnosis and treatment of primary immunodeficiency diseases. Expert Rev Clin Immunol. 2017;13(5):483–9.
Fasth A. Osteopetrosis—more than only a disease of the bone. Am J Hematol. 2009;84(8):469–70.
Jaimovich G, Rolon JM, Baldomero H, Rivas M, Hanesman I, Bouzas L, et al. Latin America: the next region for haematopoietic transplant progress. Bone Marrow Transplant. 2017;52(5):798.
Gale RP, Seber A, Bonfim C, Pasquini M. Haematopoietic cell transplants in Latin America. Bone Marrow Transplant. 2016;51(7):898–905.
Carneiro-Sampaio M, Moraes-Vasconcelos D, Kokron C, Jacob CMA, Toledo-Barros M, Dorna MB, et al. Primary immunodeficiency diseases in different age groups: a report on 1,008 cases from a single Brazilian reference center. J Clin Immunol. 2013;33:716–24.
Güngör T, Teira P, Slatter M, Stussi G, Stepensky P, Moshous D, et al. Reduced-intensity conditioning and HLA-matched haematopoietic stem-cell transplantation in patients with chronic granulomatous disease: a prospective multicenter study. Lancet. 2014;383(9915):436–48.
Shekhovtsova Z, Bonfim C, Ruggeri A, Nichele S, Page K, Al Seraihy A, et al. A risk factor analysis of outcomes after unrelated cord blood transplantation for children with Wiskott-Aldrich syndrome. Haematologica. 2017;102(6):1112–9.
Eapen M, Ahn KW, Orchard PJ, Cowan MJ, Davies SM, Fasth A, et al. Long-term survival and late deaths after hematopoietic cell transplantation for primary immunodeficiency diseases and inborn errors of metabolism. Biol Blood Marrow Transplant. 2012;18(9):1438–45.
Moratto D, Giliani S, Bonfim C, Mazzolari E, Fischer A, Ochs HD, et al. Long-term outcome and lineage-specific chimerism in 194 patients with Wiskott-Aldrich syndrome treated by hematopoietic cell transplantation in the period 1980-2009: an international collaborative study. Blood. 2011;118(6):1675–84.
Glucksberg H, Storb R, Fefer A, Buckner CD, Neiman PE, Clift RA, et al. Clinical manifestations of graft-versus-host disease in human recipients of marrow from HL-A-matched sibling donors. Transplantation. 1974;18(4):295–304.
Storb R, Prentice RL, Sullivan KM, et al. Predictive factors in chronic graft-versus-host disease in patients with aplastic anemia treated by marrow transplantation from HLA-identical siblings. Ann Intern Med. 1983;98(4):461–6.
Gaspar HB, Qasim W, Davies EG, Rao K, Amrolia PJ, Veys P. How I treat severe combined immunodeficiency. Blood. 2013;122(23):3749–58.
Pai S-Y, Cowan MJ. Stem cell transplantation for primary immunodeficiency diseases: the north American experience. Curr Opin Allergy Clin Immunol. 2014;14(6):521–6.
Hassan A, Booth C, Brightwell A, Allwood Z, Veys P, Rao K, et al. Outcome of hematopoietic stem cell transplantation for adenosine deaminase-deficient severe combined immunodeficiency. Blood. 2012;120(17):3615–24.
Schuetz C, Neven B, Dvorak CC, Leroy S, Ege MJ, Pannicke U, et al. SCID patients with ARTEMIS vs RAG deficiencies following HCT: increased risk of late toxicity in ARTEMIS-deficient SCID. Blood. 2014;123(2):281–9.
Marciano BE, Huang CY, Joshi G, Rezaei N, Carvalho BC, Allwood Z, et al. BCG vaccination in patients with severe combined immunodeficiency: complications, risks, and vaccination policies. J Allergy Clin Immunol. 2014;133(4):1134–41.
Mazzucchelli JT, Bonfim C, Castro GG, Condino-Neto AA, Costa NM, Cunha L, et al. Severe combined immunodeficiency in Brazil: management, prognosis, and BCG-associated complications. J Investig Allergol Clin Immunol. 2014;24(3):184–91.
Marciano BE, Huang CY, Joshi G, Rezaei N, Carvalho BC, Allwood Z, et al. BCG vaccination in patients with severe combined immunodeficiency: complications, risks, and vaccination policies. J Allergy Clin Immunol. 2014;133(4):1134–41.
de Oliveira-Junior EB, Zurro NB, Prando C, Cabral-Marques O, Pereira PV, Schimke LF, et al. Clinical and genotypic spectrum of chronic granulomatous disease in 71 Latin American patients: first Report from the LASID Registry. Pediatr Blood Cancer. 2015;62(12):2101–7.
Buckley RH, Schiff SE, Schiff RI, Markert ML, Williams LW, Roberts JL, et al. Hematopoietic stem-cell transplantation for the treatment of severe combined immunodeficiency. N Engl J Med. 1999;340:508–16.
Antoine C, Müller S, Cant A, Cavazzana-Calvo M, Veys P, Vossen J, et al. Long-term survival and transplantation of haemopoietic stem cells for immunodeficiencies: report of the European experience 1968-99. Lancet. 2003;361(9357):553–60.
Pai S-Y, Logan BR, Griffith LM, Buckley RH, Parrott RE, Dvorak CC, et al. Transplantation outcomes for severe combined immunodeficiency, 2000-2009. N Engl J Med. 2014;371:434–46.
Luznik L, Jalla S, Engstrom LW, Iannone R, Fuchs EJ. Durable engraftment of major histocompatibility complex-incompatible cells after nonmyeloablative conditioning with fludarabine, low-dose total body irradiation, and posttransplantation cyclophosphamide. Blood. 2001;98(12):3456–64.
Luznik L, O'Donnell PV, Symons HJ, Chen AR, Leffell MS, Zahurak M, et al. HLA-haploidentical bone marrow transplantation for hematologic malignancies using nonmyeloablative conditioning and high-dose, posttransplantation cyclophosphamide. Biol Blood Marrow Transplant. 2008;14(6):641–50.
Brodsky RA, Luznik L, Bolaños-Meade J, Leffell MS, Jones RJ, Fuchs EJ. Reduced intensity HLA-haploidentical BMT with post transplantation cyclophosphamide in nonmalignant hematologic diseases. Bone Marrow Transplant. 2008;42(8):523–7.
Bolaños-Meade J, Fuchs EJ, Luznik L, Lanzkron SM, Gamper CJ, Jones RJ, et al. HLA-haploidentical bone marrow transplantation with posttransplant cyclophosphamide expands the donor pool for patients with sickle cell disease. Blood. 2012;120(22):4285–91.
Esteves I, Bonfim C, Pasquini R, Funke V, Pereira NF, Rocha V, et al. Haploidentical BMT and post-transplant Cy for severe aplastic anemia: a multicenter retrospective study. Bone Marrow Transplant. 2015;50(5):685–9.
Bonfim C, Ribeiro L, Nichele S, Loth G, Bitencourt M, Koliski A, et al. Haploidentical bone marrow transplantation with post-transplant cyclophosphamide for children and adolescents with Fanconi Anemia. Biol Blood Marrow Transplant. 2017;23(2):310–7.
Klein OR, Chen AR, Gamper C, Loeb D, Zambidis E, Llosa N, et al. Alternative-donor hematopoietic stem cell transplantation with post-transplantation cyclophosphamide for nonmalignant disorders. Biol Blood Marrow Transplant. 2016;22(5):895–901.
Shah NN, Freeman AF, Su H, Cole K, Parta M, Moutsopoulos NM, et al. Haploidentical related donor hematopoietic stem cell transplantation for dedicator-of-cytokinesis 8 deficiency using post-transplantation cyclophosphamide. Biol Blood Marrow Transplant. 2017;23(6):980–90.
Ouederni M, Mellouli F, Khaled BK, Kaabi H, Picard C, Bejaoui M. Successful haploidentical stem cell transplantation with post-transplant cyclophosphamide in a severe combined immune deficiency patient: a first report. J Clin Immunol. 2016;36(5):437–40.
Parta M, Hilligoss D, Kelly C, Kwatemaa N, Theobald N, Malech H, et al. Haploidentical hematopoietic cell transplantation with post-transplant cyclophosphamide in a patient with chronic granulomatous disease and active infection: a first report. J Clin Immunol. 2015;35:675–80.
Kwan A, Church JA, Cowan MJ, et al. Newborn screening for severe combined immunodeficiency and T-cell lymphopenia in California: results of the first two years. J Allergy Clin Immunol. 2013;132:140.
Baker MW, Grossman WJ, Laessig RH, Hoffman GL, Brokopp CD, Kurtycz DF, et al. Development of a routine newborn screening protocol for severe combined immunodeficiency. J Allergy Clin Immunol. 2009;124:522–7.
Kanegae MPP, Barreiros LA, Sousa JL, Brito MAS, Oliveira EB Jr, Soares LP, et al. Newborn screening for severe combined immunodeficiencies using trecs and krecs: second pilot study in Brazil. Rev Paul Pediatr. 2017;35(1):25–32.
Kanegae MPP, Barreiros LA, Mazzuchelli JTL, Hadachi SM, Guilhoto LMFF, Acquesta AL, et al. Neonatal screening for severe combined immunodeficiency in Brazil. J Pediatr. 2016;92(4):374–80.
Ozsahin H, Cavazzana-Calvo M, Notarangelo LD, Schulz A, Thrasher AJ, Mazzolari E, et al. Long-term outcome following hematopoietic stem-cell transplantation in Wiskott-Aldrich syndrome: collaborative study of the European Society for Immunodeficiencies and European Group for Blood and Marrow Transplantation. Blood. 2008;111(1):439–45.
Shin CR, Kim MO, Li D, Bleesing JJ, Harris R, Mehta P, et al. Outcomes following hematopoietic cell transplantation for Wiskott-Aldrich syndrome. Bone Marrow Transplant. 2012;47(11):1428–35.
Morillo-Gutierrez B, Beier R, Rao K, et al. Treosulfan based conditioning for allogeneic haematopoietic stem cell transplantation in children with Chronic Granulomatous Disease. Multicentre experience on behalf of the EBMT Inborn Errors Working Party. Blood. 2016;128:440–8.
Acknowledgments
The authors thank all the nurses, physicians, and multidisciplinary staff of the participating centers.
Authorship Contribution
JFF designed the study, enrolled and cared for patients, reviewed and analyzed data, and wrote the paper. CB and SN designed the study, enrolled and cared for patients, and reviewed the paper. All the other authors contributed to data collection, patient care, and reviewed the paper.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
This study was approved by the Institutional Review Boards of each center and performed in accordance with the 1964 Declaration of Helsinki.
Conflict of Interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Fernandes, J.F., Nichele, S., Daudt, L.E. et al. Transplantation of Hematopoietic Stem Cells for Primary Immunodeficiencies in Brazil: Challenges in Treating Rare Diseases in Develo** Countries. J Clin Immunol 38, 917–926 (2018). https://doi.org/10.1007/s10875-018-0564-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10875-018-0564-1