Abstract
Objectives
The aim of this study was to evaluate immunity in HIV-uninfected children with bacille Calmette–Guerin-induced disease (BCG-ID) over an 8-year period, with particular emphasis on underlying diseases.
Methods
Patient afflicted with BCG-ID proven by clinical courses, dermatologic features, pathology, specific polymerase chain reaction, and/or spoligoty** were enrolled between 2000 and 2007. Lymphocyte proliferation, polymorphonuclear function, interleukin (IL)-12/23–interferons (IFN)-γ circuit, and Toll-like receptor 2-associated signaling were investigated.
Results
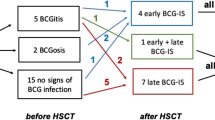
Of the 271,618 total live births who received the BCG vaccine, eight patients (seven males) with BCG-ID were enrolled during an 8-year period and presented as three disseminated, two distant, and three regional BCG-ID. Their age at onset ranged from 1 to 28 months. All had a vaccine-injection scar except for one with lower CD3 and natural killer cells, compatible with severe combined immunodeficiency (SCID) identified by IL-2 receptor common gamma chain (IL2RG) mutation (Arg226Lys). The other SCID patient with de novo IL2RG mutation (Trp74Gly) had more recurrent infections. The third patient with primary autoimmune neutropenia had disseminated BCG-ID extending to abdominal wall. The fourth patient with chronic mucocutaneous candidiasis had regional BCG-ID and impaired lymphocyte proliferation to Candida and BCG antigens. No defective evidence of polymorphonuclear functions, IL-12/23–IFN-γ circuit, and Toll-like receptor 2-associated signaling was detected in the remaining four patients.
Conclusion
Immunologic analysis in HIV-uninfected patients with BCG-ID reveals primary immunodeficiency diseases, especially in those with deficiencies in T-cell and neutrophil functions observed in our cohort, including primary autoimmune neutropenia and chronic mucocutaneous candidiasis.



Similar content being viewed by others
Abbreviations
- BCG:
-
bacille Calmette–Guerin
- CMC:
-
chronic mucocutaneous candidiasis
- CGD:
-
chronic granulomatous disease
- IL2RG:
-
IL-2 receptor common gamma chain
- PBMCs:
-
peripheral blood mononuclear cells
- PIDD:
-
primary immunodeficiency diseases
- ROS:
-
reactive oxygen species
- SCID:
-
severe combined immunodeficiency
References
Colditz GA, Brewer TF, Berkey CS, Wilson ME, Burdick E, Fineberg HV, Mosteller F. Efficacy of BCG vaccine in the prevention of tuberculosis. Meta-analysis of the published literature. JAMA 1994;271:698–702. doi:10.1001/jama.271.9.698.
World Health Organization. BCG vaccine: WHO position paper. Wkly Epidemiol Rec 2004;79:27–38. Available from http://www.who.int/wer. Accessed 14 September 2005.
Kroger L, Korppi M, Brander E, Kroger H, Wasz-Hockert O, Backman A, Rapola J, Launiala K, Katila ML. Osteitis caused by bacille Calmette–Guerin vaccination: a retrospective analysis of 222 cases. J Infect Dis 1995;172:574–6.
Casanova JL, Jouanguy E, Lamhamedi S, Blanche S, Fischer A. Immunological conditions of children with BCG disseminated infection. Lancet 1995;346:581. doi:10.1016/S0140-6736(95)91421-8.
Filipe-Santos O, Bustamante J, Chapgier A, Vogt G, de Beaucoudrey L, Feinberg J, Jouanguy E, Boisson-Dupuis S, Fieschi C, Picard C, Casanova JL. Inborn errors of IL-12/23- and IFN-gamma-mediated immunity: molecular, cellular, and clinical features. Semin Immunol 2006;18:347–61. doi:10.1016/j.smim.2006.07.010.
Hornung V, Rothenfusser S, Britsch S, Britsch S, Towarowski A, Krug A, Sarris A, Lubenow N, Speiser D, Endres S, Hartmann G. Quantitative expression of Toll-like receptor 1–10 mRNA in cellular subsets of human peripheral blood mononuclear cells and sensitivity to CpG oligodeoxynucleotides. J Immunol 2002;168:4531–7.
Stenger S, Modlin RL. Control of Mycobacterium tuberculosis through mammalian Toll-like receptors. Curr Opin Immunol 2002;14:452–7. doi:10.1016/S0952-7915(02)00355-2.
Brightbill HD, Libraty DH, Krutzik SR, Yang RB, Belisle JT, Bleharski JR, Maitland M, Norgard MV, Plevy SE, Smale ST, Brennan PJ, Bloom BR, Godowski PJ, Modlin RL. Host defense mechanisms triggered by microbial lipoproteins through toll-like receptors. Science 1999;285:732–6. doi:10.1126/science.285.5428.732.
Means TK, Jones BW, Schromm AB, Shurtleff BA, Smith JA, Keane J, Golenbock DT, Vogel SN, Fenton MJ. Differential effects of a Toll-like receptor antagonist on Mycobacterium tuberculosis-induced macrophage responses. J Immunol 2001;166:4074–82.
Hesseling AC, Rabie H, Marais BJ, Manders M, Lips M, Schaaf HS, Gie RP, Cotton MF, van Helden PD, Warren RM, Beyers N. Bacille Calmette–Guerin vaccine-induced disease in HIV-infected and HIV-uninfected children. Clin Infect Dis 2006;42:548–58. doi:10.1086/499953.
Lee WI, Kuo ML, Huang JL, Lin SJ, Wu CJ. Distribution and clinical aspects of primary immunodeficiencies in a Taiwan pediatric tertiary hospital during a 20-year period. J Clin Immunol 2005;25:162–73. doi:10.1007/s10875-005-2822-2.
Serour F, Mizrahi A, Somekh E, Feinberg J, Picard C, Casanova JL, Dalal I. Analysis of the interleukin-12/interferon-γ pathway in children with non-tuberculous mycobacterial cervical lymphadenitis. Eur J Pediatr 2007;166:835–41. doi:10.1007/s00431-006-0338-2.
Cleary AM, Tu W, Enright A, Giffon T, Dewaal-Malefyt R, Gutierrez K, Lewis DB. Impaired accumulation and function of memory CD4 T cells in human IL-12 receptor β1 deficiency. J Immunol 2003;170:597–603.
Dorman SE, Picard C, Lammas D, Heyne K, van Dissel JT, Baretto R, Rosenzweig SD, Newport M, Levin M, Roesler J, Kumararatne D, Casanova JL, Holland SM. Clinical features of dominant and recessive interferon gamma receptor 1 deficiencies. Lancet 2004;364:2113–21. doi:10.1016/S0140-6736(04)17552-1.
Jouanguy E, Lamhamedi-Cherradi S, Altare F, Fondanèche MC, Tuerlinckx D, Blanche S, Emile JF, Gaillard JL, Schreiber R, Levin M, Fischer A, Hivroz C, Casanova JL. Partial interferon-gamma receptor 1 deficiency in a child with tuberculoid bacillus Calmette–Guerin infection and a sibling with clinical tuberculosis. J Clin Invest 1997;100:2658–64. doi:10.1172/JCI119810.
Picard C, Fieschi C, Altare F, Al-Jumaah S, Al-Hajjar S, Feinberg J, Dupuis S, Soudais C, Al-Mohsen IZ, Génin E, Lammas D, Kumararatne DS, Leclerc T, Rafii A, Frayha H, Murugasu B, Wah LB, Sinniah R, Loubser M, Okamoto E, Al-Ghonaium A, Tufenkeji H, Abel L, Casanova JL. Inherited interleukin-12 deficiency: IL12B genotype and clinical phenotype of 13 patients from six kindreds. Am J Hum Genet 2002;70:336–48. doi:10.1086/338625.
Fieschi C, Dupuis S, Catherinot E, Feinberg J, Bustamante J, Breiman A, Altare F, Baretto R, Le Deist F, Kayal S, Koch H, Richter D, Brezina M, Aksu G, Wood P, Al-Jumaah S, Raspall M, Da Silva Duarte AJ, Tuerlinckx D, Virelizier JL, Fischer A, Enright A, Bernhöft J, Cleary AM, Vermylen C, Rodriguez-Gallego C, Davies G, Blütters-Sawatzki R, Siegrist CA, Ehlayel MS, Novelli V, Haas WH, Levy J, Freihorst J, Al-Hajjar S, Nadal D, De Moraes Vasconcelos D, Jeppsson O, Kutukculer N, Frecerova K, Caragol I, Lammas D, Kumararatne DS, Abel L, Casanova JL. Low penetrance, broad resistance, and favorable outcome of interleukin 12 receptor beta1 deficiency: medical and immunological implications. J Exp Med 2003;197:527–35. doi:10.1084/jem.20021769.
Dupuis S, Dargemont C, Fieschi C, Thomassin N, Rosenzweig S, Harris J, Holland SM, Schreiber RD, Casanova JL. Impairment of mycobacterial but not viral immunity by a germline human STAT1 mutation. Science 2001;293:300–3. doi:10.1126/science.1061154.
Dupuis S, Jouanguy E, Al-Hajjar S, Fieschi C, Al-Mohsen IZ, Al-Jumaah S, Yang K, Chapgier A, Eidenschenk C, Eid P, Al Ghonaium A, Tufenkeji H, Frayha H, Al-Gazlan S, Al-Rayes H, Schreiber RD, Gresser I, Casanova JL. Impaired response to interferonalpha/beta and lethal viral disease in human STAT1 deficiency. Nat Genet 2003;33:388–91. doi:10.1038/ng1097.
Report on Registration of Births and Deaths, 2000–2007. Registry of Births and Deaths, National Registeration Department, Taiwan
Lee WI, Jaing TH, Hsieh MY, Kuo ML, Lin SJ, Huang JL. Distribution, infections, treatments and molecular analysis in a large cohort of patients with primary immunodeficiency diseases (PIDs) in Taiwan. J Clin Immunol 2006;26:274–83. doi:10.1007/s10875-006-9013-7.
Bustamante J, Aksu G, Vogt G, de Beaucoudrey L, Genel F, Chapgier A, Filipe-Santos O, Feinberg J, Emile JF, Kutukculer N, Casanova JL. BCG-osis and tuberculosis in a child with chronic granulomatous disease. J Allergy Clin Immunol 2007;120:32–8. doi:10.1016/j.jaci.2007.04.034.
FitzGerald JM. Management of adverse reactions to bacille Calmette–Guérin vaccine. Clin Infect Dis 2000;31(Suppl 3):S75–6. doi:10.1086/314074.
Casanova JL, Blanche S, Emile JF, Jouanguy E, Lamhamedi S, Altare F, Stéphan JL, Bernaudin F, Bordigoni P, Turck D, Lachaux A, Albertini M, Bourrillon A, Dommergues JP, Pocidalo MA, Le Deist F, Gaillard JL, Griscelli C, Fischer A. Idiopathic disseminated bacillus Calmette–Guérin infection: a French national retrospective study. Pediatrics 1996;98:774–8.
Talbot EA, Perkins MD, Silva SF, Frothingham R. Disseminated bacilli Calmette–Guerin disease after vaccination: case report and review. Clin Infect Dis 1997;24:1139–46. doi:10.1086/513642.
Flynn JL, Ernst J. Immune responses in tuberculosis. Curr Opin Immunol 2000;12:432–6. doi:10.1016/S0952-7915(00)00116-3.
Abbas AK, Lichtman AH, editors. Effector mechanisms of cell-mediated immunity. In: Cellular and molecular immunology, 5th ed. Philadelphia: Saunders; 2003. pp. 298–317.
Canaday DH, Ziebold C, Noss EH, Chervenak KA, Harding CV, Boom WH. Activation of human CD8+ alpha beta TCR+ cells by Mycobacterium tuberculosis via an alternate class I MHC antigen-processing pathway. J Immunol 1999;162:372–9.
Smith SM, Klein MR, Malin AS, Sillah J, Huygen K, Andersen P, McAdam KP, Dockrell HM. Human CD8(+) T cells specific for Mycobacterium tuberculosis secreted antigens in tuberculosis patients and healthy BCG-vaccinated controls in The Gambia. Infect Immun 2000;68:7144–8. doi:10.1128/IAI.68.12.7144-7148.2000.
Babior BM. NADPH oxidase. Curr Opin Immunol 2004;16:42–7. doi:10.1016/j.coi.2003.12.001.
Lee WI, Torgerson TR, Schumacher MJ, Yel L, Zhu Q, Ochs HD. Molecular analysis of a large cohort of patients with the hyper immunoglobulin M (IgM) syndrome. Blood 2005;105:1881–90. doi:10.1182/blood-2003-12-4420.
Reichenbach J, Rosenzweig S, Doffinger R, Dupuis S, Holland SM, Casanova JL. Mycobacterial diseases in primary immunodeficiencies. Curr Opin Allergy Clin Immunol 2001;1:503–11. doi:10.1097/00130832-200112000-00003.
Jain A, Ma CA, Liu S, Brown M, Cohen J, Strober W. Specific missense mutations in NEMO result in hyper-IgM syndrome with hypohydrotic ectodermal dysplasia. Nat Immunol 2001;2:223–8. doi:10.1038/85277.
Janssen R, van Wengen A, Hoeve MA, ten Dam M, van der Burg M, van Dongen J, van de Vosse E, van Tol M, Bredius R, Ottenhoff TH, Weemaes C, van Dissel JT, Lankester A. The same IkappaBalpha mutation in two related individuals leads to completely different clinical syndromes. J Exp Med 2004;200:559–68. doi:10.1084/jem.20040773.
Hodsagi M, Uhereczky G, Kiraly L, Pinter E. BCG dissemination in chronic granulomatous disease (CGD). Dev Biol Stand 1986;58:339–46.
Vieira AP, Vasconcelos J, Fernandes JC, Antunes H, Basto AS, Macedo C, Zaman A, Santos E, Melo JC, Roos D. Lymphadenopathy after BCG vaccination in a child with chronic granulomatous disease. Pediatr Dermatol 2004;21:646–51. doi:10.1111/j.0736-8046.2004.21606.x.
Lee PP, Chan KW, Jiang L, Chen T, Li C, Lee TL, Mak PH, Fok SF, Yang X, Lau YL. Susceptibility to mycobacterial infections in children with X-linked chronic granulomatous disease: a review of 17 patients living a region endemic for tuberculosis. Pediatr Infect Dis J 2008;27:224–30.
Kamerbeek J, Schouls A, Kolk M, van Agterveld D, van Soolingen S, Kuijper A, Bunschoten A, Molhuizen H, Shaw R, Goyal M, van Embden J. Simultaneous detection and strain differentiation of Mycobacterium tuberculosis for diagnosis and epidemiology. J Clin Microbiol 1997;35:907–14.
Acknowledgments
The authors wish to thank all of the patients and their families for their kind cooperation, as well as their physicians for the referrals. This study was supported by Chang-Gung Medical Research Progress Grant CMRPG 450061 and CMRPG 340222 and National Science Council Grants C93-2314-B-182A-053-MY2 and NMRPG3131.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Lee, WI., Liang, FC., Huang, JL. et al. Immunologic Analysis of HIV-Uninfected Taiwanese Children with BCG-Induced Disease. J Clin Immunol 29, 319–329 (2009). https://doi.org/10.1007/s10875-008-9265-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10875-008-9265-5