Abstract
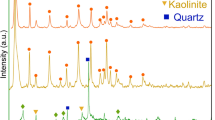
An innovative method for preparation of carbon-coated zeolites (black zeolites) has been developed. The method involves dehydration of sucrose on kaolin surfaces followed by hydrothermal treatment. Raw and dealuminated kaolins were used to prepare black zeolites (CZ1 and CZ2, respectively). Characteristics of the black zeolites were influenced by the criteria of the used kaolin. Analyses indicate the formation of Na-A and Na-P zeolites in CZ1. However, Na-A, Na-X, and Na-P zeolites were found in CZ2. Surface areas and pore volume values of CZ2 are lower than the corresponding bare zeolite (Z2). Meanwhile, these values remain approximately the same in case of CZ1 when compared with the corresponding bare zeolite (Z1). Carbon is probably trapped in the wide pores of CZ2 during the preparation process. Contrarily, carbon mainly coats the external surface of CZ1. The adsorption capacities of CZ1 and CZ2 using methylene blue (MB) were determined to be 48.33 and 57.64 mg/g, respectively. The higher estimated value of CZ2 is attributed to its higher surface area. Moreover, the adsorption capacities of black zeolites were much higher than the corresponding uncoated zeolites and the unsupported carbon. These indicate that the supported carbon on zeolites possesses higher exposed surfaces to MB.







Similar content being viewed by others
References
Dyer A, Morgan S, Wells P, Williams C (2000) The use of zeolites as slow release anthelmintic carriers. J Helminthol 74:137–141
Kyotani T, Nagai T, Inoue S, Tomita A (1997) Formation of new type of porous carbon by carbonization in zeolite nanochannels. Chem Mater 9:609–615
Sakintuna B, Aktaş Z, Yürüm Y (2003) Synthesis of porous carbon materials by carbonization in natural zeolite nanochannels. Prepr Pap -Am Chem Soc, Div Fuel Chem 48:614–615
Cordero T, Thrower PA, Radovic LR (1992) On the oxidation resistance of carbon-carbon composites obtained by chemical vapor infiltration of different carbon cloths. Carbon 30:365–374
Velo-Gala I, López-Peñalver JJ, Sánchez-Polo M, Rivera-Utrilla J (2017) Role of activated carbon surface chemistry in its photocatalytic activity and the generation of oxidant radicals under UV or solar radiation. Appl Catal B 207:412–423
Botomé ML, Poletto P, Junges J, Perondi D, Dettmer A, Godinho M (2017) Preparation and characterization of a metal-rich activated carbon from CCA-treated wood for CO2 capture. Chem Eng J 321:614–621
Suhas PJM, Carrott MML, Carrott R, Singh R, Singh LP, Chaudhary M (2017) An innovative approach to develop microporous activated carbons in oxidising atmosphere. J Clean Prod 156:549–555
Filipe-Ribeiro L, Milheiro J, Matos CC, Cosme F, Nunes FM (2017) Data on changes in red wine phenolic compounds, headspace aroma compounds and sensory profile after treatment of red wines with activated carbons with different physicochemical characteristics. Data Brief 12:188–202
Zhang X, Gu P, Li X, Zhang G (2017) Efficient adsorption of radioactive iodide ion from simulated wastewater by nano Cu2O/Cu modified activated carbon. Chem Eng J 322:129–139
Björklund K, Li LY (2017) Adsorption of organic stormwater pollutants onto activated carbon from sewage sludge. J Environ Manage 197:490–497
Lutz W, Zeolite Y (2014) Synthesis, modification, and properties—a case revisited. Adv Mater Sci Eng 2014:1–20
Petushkov A, Freeman J, Larsen SC (2010) Framework stability of nanocrystalline NaY in aqueous solution at varying pH. Langmuir 26(9):6695–6701
El-Mekkawi DM, Ibrahim FA, Selim MM (2016) Removal of methylene blue from water using zeolites prepared from Egyptian kaolins collected from different sources. J Environ Chem Eng 4(2):1417–1422
El-Mekkawi DM, Ibrahim FA, Selim MM (2015) Stability and thermal transformation studies of Zn2+- and Fe2+-loaded zeolite Y prepared from Egyptian kaolin. Solid State Sci 48:294–299
El-Mekkawi DM, Selim MM (2014) Removal of Pb2+ from water by using Na-Y zeolites prepared from Egyptian kaolins collected from different sources. J Environ Chem Eng 2(1):723–730
El-Mekkawi DM, Selim MM (2012) Effect of metal loading processes on the stability and thermal transformation of Co2+- and Cu2+-zeolite Y prepared from Egyptian kaolin. Mater Charact 69:37–44
Ibraheem HH, El-Mekkawi DM, Hassan SA, Selim MM (2010) Innovative method for the reduction of nitrophenols using nickel nanocatalysts in Zeolite-Y prepared from Egyptian kaolin. Egypt J Chem 53(4):565–579
Selim MM, EL-Mekkawi DM, Aboelenin RMM, Sayed Ahmed SA, Mohamed GM (2017) Preparation and characterization of Na-A zeolite from aluminum scrub and commercial sodium silicate for the removal of Cd2+ from water. doi: http://dx.doi.org/10.1016/j.jaubas.2017.05.002
Snellings R, Mertens G, Elsen J (2012) Supplementary cementitious materials. Rev Mineral Geochem 74:211–278
Patterson AL (1939) The Scherrer formula for X-ray particle size determination. Phys Rev 56:978–979
Adamson AW, Gast AP (1997) Physical chemistry of surfaces. Wiley, New York
Boccaleri E, Arrias A, Frache A, Gianelli W, Fino P, Camino G (2006) Comperehensive spectral and instrumental approaches for the easy monitoring of features and purity of different carbon nanostructures for nanocomposite applications. Mater Sci Engineer B 131:72–82
Arrais A, Diana E, Boccaleri E (2006) A study on the carbon soot derived from the wood combustion and on the relative alkali-extractable fraction. J Mater Sci 41:6035–6045. doi:10.1007/s10853-006-0511-z
Drag EB, Miecznikoski A, Abo-Lemon F, Rutkowski M (1985) Syhthesis of A, X and Y zeolites from clay minerals. In: Držaj B, Hocevar S, Pejovnik S (eds) Zeolites: synthesis, structure, technology and application. Elsevier Science publishers BV, Amestrdam, pp 147–154
Kotova OB (2016) Zeolite synthesis as potential application of coal fly ash. In: Litvinenko V (ed) XVIII international coal preparation congress: 28 June—01 July 2016 Saint-Petersburg, Russia. Springer International Publishing, Basel, pp 322–326
Barrett EP, Joyner LG, Halenda PH (1951) BJH method. J Am Chem Soc 73:373–380
Brunauer S, Emmett PH, Teller E (1938) Adsorption of gases in multimolecular layers. J Am Chem Soc 60:309–319
Lippens BC, de Boer JH (1965) Studies on pore system in catalysis: V. The t method. J Catal 4:319–323
Du X, Wu E (2007) Porosity of microporous zeolites A, X and ZSM-5 studied by small angle X-ray scattering and nitrogen adsorption. J Phys Chem Solids 68:1692–1699
Job R (2005) Zeolites and nanoclusters in zeolite host lattices. In: Fahrner WR (ed) Nanotechnology and nanoelectronics: materials, devices, measurement techniques. Springer, Berlin, pp 127–142
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Rights and permissions
About this article
Cite this article
Selim, M.M., EL-Mekkawi, D.M. & Ibrahim, F.A. Innovative synthesis of black zeolites-based kaolin and their adsorption behavior in the removal of methylene blue from water. J Mater Sci 53, 3323–3331 (2018). https://doi.org/10.1007/s10853-017-1744-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10853-017-1744-8