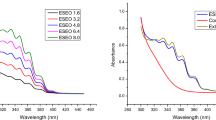
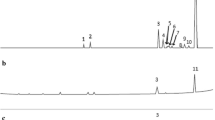
Abstract
Eucalyptus citriodora is one of the most widely used essential oils (EOs) because of its various antimicrobial, antioxidant and anti-inflammatory activities. However, its limited aqueous solubility restricts its use. The aim of this study was to develop supramolecular formulations able to retain E. citriodora EO in solution. For this purpose, new cyclodextrin (CD) polymers were synthesized and characterized using gel permeation chromatography, FTIR and NMR spectroscopies. Their retention ability toward E. citriodora in non-conventional green media (deep eutectic solvent (DES):water mixture), more precisely choline chloride:urea DES:water (70:30 wt%), was evaluated and compared to the corresponding native CD [β-cyclodextrin (β-CD)] or CD derivatives [hydroxypropylated-β-cyclodextrin (HP-β-CD) and low methylated-β-cyclodextrin (CRYSMEB)] using static headspace-gas chromatography (SH-GC). All the studied formulations showed a great capacity to retain and reduce the volatility of E. citriodora. The various polymers showed divergent retention efficiencies.






Similar content being viewed by others
References
Sebaaly, C., Jraij, A., Fessi, H., Charcosset, C., Greige-Gerges, H.: Preparation and characterization of clove essential oil-loaded liposomes. Food Chem. 178, 52–62 (2015). https://doi.org/10.1016/j.foodchem.2015.01.067
Kfoury, M., Hădărugă, N.G., Hădărugă, D.I., Fourmentin, S.: Cyclodextrins as encapsulation material for flavors and aroma. In: Encapsulations, pp. 127–192. Elsevier, Amsterdam (2016)
Nakhle, L., Kfoury, M., Greige-Gerges, H., Fourmentin, S.: Effect of dimethylsulfoxide, ethanol, α- and β-cyclodextrins and their association on the solubility of natural bioactive compounds. J. Mol. Liq. 310, 113156 (2020). https://doi.org/10.1016/j.molliq.2020.113156
Kfoury, M., Geagea, C., Ruellan, S., Greige-Gerges, H., Fourmentin, S.: Effect of cyclodextrin and cosolvent on the solubility and antioxidant activity of caffeic acid. Food Chem. 278, 163–169 (2019). https://doi.org/10.1016/j.foodchem.2018.11.055
Crini, G.: Review: a history of cyclodextrins. Chem. Rev. 114, 10940–10975 (2014). https://doi.org/10.1021/cr500081p
Kfoury, M., Auezova, L., Greige-Gerges, H., Fourmentin, S.: Promising applications of cyclodextrins in food: improvement of essential oils retention, controlled release and antiradical activity. Carbohydr. Polym. 131, 264–272 (2015). https://doi.org/10.1016/j.carbpol.2015.06.014
Kfoury, M., Landy, D., Fourmentin, S.: Characterization of cyclodextrin/volatile inclusion complexes: a review. Molecules 23, 1204 (2018). https://doi.org/10.3390/molecules23051204
Kfoury, M., Auezova, L., Fourmentin, S., Greige-Gerges, H.: Investigation of monoterpenes complexation with hydroxypropyl-β-cyclodextrin. J. Incl. Phenom. Macrocycl. Chem. 80, 51–60 (2014). https://doi.org/10.1007/s10847-014-0385-7
Kfoury, M., Auezova, L., Ruellan, S., Greige-Gerges, H., Fourmentin, S.: Complexation of estragole as pure compound and as main component of basil and tarragon essential oils with cyclodextrins. Carbohydr. Polym. 118, 156–164 (2015). https://doi.org/10.1016/j.carbpol.2014.10.073
Crini, G., Fourmentin, S., Fenyvesi, É., Torri, G., Fourmentin, M., Morin-Crini, N.: Cyclodextrins, from molecules to applications. Environ. Chem. Lett. 16, 1361–1375 (2018). https://doi.org/10.1007/s10311-018-0763-2
McCune, J.A., Kunz, S., Olesińska, M., Scherman, O.A.: DESolution of CD and CB macrocycles. Chem. Eur. J. 23, 8601–8604 (2017). https://doi.org/10.1002/chem.201701275
Rubin Pedrazzo, A., Trotta, F., Hoti, G., Cesano, F., Zanetti, M.: Sustainable mechanochemical synthesis of β-cyclodextrin polymers by twin screw extrusion. Environ. Sci. Pollut. Res. 29, 251–263 (2022). https://doi.org/10.1007/s11356-021-15187-5
Cecone, C., Hoti, G., Krabicová, I., Appleton, S.L., Caldera, F., Bracco, P., Zanetti, M., Trotta, F.: Sustainable synthesis of cyclodextrin-based polymers by exploiting natural deep eutectic solvents. Green Chem. 22, 5806–5814 (2020). https://doi.org/10.1039/D0GC02247K
Ciobanu, A., Mallard, I., Landy, D., Brabie, G., Nistor, D., Fourmentin, S.: Inclusion interactions of cyclodextrins and crosslinked cyclodextrin polymers with linalool and camphor in Lavandula angustifolia essential oil. Carbohydr. Polym. 87, 1963–1970 (2012). https://doi.org/10.1016/j.carbpol.2011.10.005
Ciobanu, A., Mallard, I., Landy, D., Brabie, G., Nistor, D., Fourmentin, S.: Retention of aroma compounds from Mentha piperita essential oil by cyclodextrins and crosslinked cyclodextrin polymers. Food Chem. 138, 291–297 (2013). https://doi.org/10.1016/j.foodchem.2012.10.106
Shishov, A., Bulatov, A., Locatelli, M., Carradori, S., Andruch, V.: Application of deep eutectic solvents in analytical chemistry. A review. Microchem. J. 135, 33–38 (2017). https://doi.org/10.1016/j.microc.2017.07.015
El Achkar, T., Greige-Gerges, H., Fourmentin, S.: Basics and properties of deep eutectic solvents: a review. Environ. Chem. Lett. (2021). https://doi.org/10.1007/s10311-021-01225-8
Martins, M.A.R., Pinho, S.P., Coutinho, J.A.P.: Insights into the nature of eutectic and deep eutectic mixtures. J. Solution Chem. 48, 962–982 (2019). https://doi.org/10.1007/s10953-018-0793-1
Abbott, A.P., Capper, G., Davies, D.L., Rasheed, R.K., Tambyrajah, V.: Novel solvent properties of choline chloride/urea mixturesElectronic supplementary information (ESI) available: spectroscopic data. See http://www.rsc.org/suppdata/cc/b2/b210714g/. Chemical Communications. 70–71 (2003). https://doi.org/10.1039/b210714g
Smith, E.L., Abbott, A.P., Ryder, K.S.: Deep eutectic solvents (DESs) and their applications. Chem. Rev. 114, 11060–11082 (2014). https://doi.org/10.1021/cr300162p
El Achkar, T., Moura, L., Moufawad, T., Ruellan, S., Panda, S., Longuemart, S., Legrand, F.-X., Costa Gomes, M., Landy, D., Greige-Gerges, H., Fourmentin, S.: New generation of supramolecular mixtures: characterization and solubilization studies. Int. J. Pharm. (2020). https://doi.org/10.1016/j.ijpharm.2020.119443
Martel, B., Ruffin, D., Weltrowski, M., Lekchiri, Y., Morcellet, M.: Water-soluble polymers and gels from the polycondensation between cyclodextrins and poly(carboxylic acid)s: a study of the preparation parameters. J. Appl. Polym. Sci. 97, 433–442 (2005). https://doi.org/10.1002/app.21391
Luppi, F., Cavaye, H., Dossi, E.: Nitrated cross-linked β-cyclodextrin binders exhibiting low glass transition temperatures. Propellants, Explos., Pyrotech. 43, 1023–1031 (2018). https://doi.org/10.1002/prep.201800137
Wintgens, V., Dalmas, F., Sébille, B., Amiel, C.: Novel phosphorus-containing cyclodextrin polymers and their affinity for calcium cations and hydroxyapatite. Carbohydr. Polym. 98, 896–904 (2013). https://doi.org/10.1016/j.carbpol.2013.06.073
Ohno, K., Wong, B., Haddleton, D.M.: Synthesis of well-defined cyclodextrin-core star polymers. J. Polym. Sci. A Polym. Chem. 39, 2206–2214 (2001). https://doi.org/10.1002/pola.1197
Anand, R., Malanga, M., Manet, I., Manoli, F., Tuza, K., Aykaç, A., Ladavière, C., Fenyvesi, E., Vargas-Berenguel, A., Gref, R., Monti, S.: Citric acid–γ-cyclodextrin crosslinked oligomers as carriers for doxorubicin delivery. Photochem. Photobiol. Sci. 12, 1841 (2013). https://doi.org/10.1039/c3pp50169h
Wintgens, V., Lorthioir, C., Dubot, P., Sébille, B., Amiel, C.: Cyclodextrin/dextran based hydrogels prepared by cross-linking with sodium trimetaphosphate. Carbohydr. Polym. 132, 80–88 (2015). https://doi.org/10.1016/j.carbpol.2015.06.038
Li, Y.-F., Ha, Y.-M., Guo, Q., Li, Q.-P.: Synthesis of two β-cyclodextrin derivatives containing a vinyl group. Carbohydr. Res. 404, 55–62 (2015). https://doi.org/10.1016/j.carres.2014.11.012
Nakhle, L., Kfoury, M., Mallard, I., Greige-Gerges, H., Landy, D.: Solubilization of Eucalyptus citriodora essential oil and citronellal in deep eutectic solvents:water:cyclodextrins mixtures. J. Mol. Liq. 359, 119371 (2022). https://doi.org/10.1016/j.molliq.2022.119371
Dugoni, G.C., Di Pietro, M.E., Ferro, M., Castiglione, F., Ruellan, S., Moufawad, T., Moura, L., Costa Gomes, M.F., Fourmentin, S., Mele, A.: Effect of water on deep eutectic solvent/β-cyclodextrin systems. ACS Sustain. Chem. Eng. 7, 7277–7285 (2019). https://doi.org/10.1021/acssuschemeng.9b00315
Cao, J., Cao, J., Wang, H., Chen, L., Cao, F., Su, E.: Solubility improvement of phytochemicals using (natural) deep eutectic solvents and their bioactivity evaluation. J. Mol. Liq. 318, 113997 (2020). https://doi.org/10.1016/j.molliq.2020.113997
Morin-Crini, N., Winterton, P., Fourmentin, S., Wilson, L.D., Fenyvesi, É., Crini, G.: Water-insoluble β-cyclodextrin–epichlorohydrin polymers for removal of pollutants from aqueous solutions by sorption processes using batch studies: a review of inclusion mechanisms. Prog. Polym. Sci. 78, 1–23 (2018). https://doi.org/10.1016/j.progpolymsci.2017.07.004
Saokham, P., Muankaew, C., Jansook, P., Loftsson, T.: Solubility of cyclodextrins and drug/cyclodextrin complexes. Molecules 23, 1161 (2018). https://doi.org/10.3390/molecules23051161
Landy, D., Fourmentin, S., Salome, M., Surpateanu, G.: Analytical improvement in measuring formation constants of inclusion complexes between β-cyclodextrin and phenolic compounds. J. Incl. Phenom. Macrocycl. Chem. 38, 187–198 (2000). https://doi.org/10.1023/A:1008156110999
Acknowledgements
The authors would like to acknowledge the National Council for Scientific Research of Lebanon (CNRS-L) and Université du Littoral Côte d'Opale (ULCO) for granting a doctoral fellowship to Lamia Nakhle. This work is a contribution to the CPER (Contrat de Plan Etat-Région) research project IRenE (Innovation et Recherche en Environnement) and is supported by the French Ministère de l’Enseignement Supérieur, the region Hauts-de-France and the European Regional Development Fund.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Nakhle, L., Kfoury, M., Mallard, I. et al. Cyclodextrin polymers in combination with water and deep eutectic solvent for the retention of Eucalyptus citriodora essential oil. J Incl Phenom Macrocycl Chem 102, 831–840 (2022). https://doi.org/10.1007/s10847-022-01161-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10847-022-01161-2