Abstract
Purpose
This meta-analysis aimed to review the safety and efficacy of topical cyclosporine A (CsA) and topical tacrolimus in allergic eye disease.
Methods
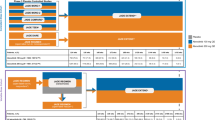
A systematic search identified thirteen studies and a total of 445 patients for inclusion, making this the largest meta-analysis published on the subject. The current review was performed in accordance with Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA).
Results
Thirteen randomized control trials were included in the meta-analysis. Eleven studies used CsA as the treatment, and two used Tacrolimus. In total, 445 participants were included, of whom 76.6% were male. The mean age of participants across the included studies was 14 years. All studies reported clinical signs as evaluated by an examining clinician. Signs were usually assessed by anatomical region, with the most common regions being the conjunctiva and the cornea, and the most common signs assessed were hyperemia and papillae. Three studies accounted for more than 50% of the meta-analysis's weight. Effect size (d) ranged from − 2.37 to − 0.03, negative values favoring immunomodulators. Fixed Effect Meta-Analysis returned an SMD of − 0.81 (95% CI [− 0.98, − 0.65]). However, there was significant heterogeneity (I2 = 61%, Qw = 30.76) in the outcome measure (P = 0.0021); therefore, a random-effect meta-analysis was also completed in which the pooled SMD was − 0.98 (95% CI [− 1.26, − 0.69], τ2 = 0.16).
Conclusions
This study affirms the current scientific community's stance that immunomodulators effectively treat clinical signs, including blepharitis, conjunctival hyperemia, edema, papillae, and corneal damage in severe ocular allergic disease.





Similar content being viewed by others
References
Csorba A, Soproni A, Maneschg O, Nagy Z, Szamosi A (2019) Application of corticosteroid eye drops for allergic eye diseases in children. Orv Hetil 160(9):329–337
Baranwal V, Mishra A, Sharma V, Gupta S, Sunder S, Verma S (2019) The prevalence of various eye diseases among patients of different nationalities attending the ophthalmology clinic at a tertiary care united nations hospital: a 5 year retrospective analysis. J Contemp Med Res 6(9):I7–I10
Graif Y, Garty B-Z, Livne I, Green MS, Shohat T (2004) Prevalence and risk factors for allergic rhinitis and atopic eczema among schoolchildren in Israel: results from a national study. Ann Allergy Asthma Immunol 92(2):245–249
Zhong J-Y, Lee Y-C, Hsieh C-J, Tseng C-C, Yiin L-M (2018) Association between dry eye disease, air pollution and weather changes in Taiwan. Int J Environ Res Public Health 15(10):2269
Shaker M, Salcone E (2016) An update on ocular allergy. Curr Opin Allergy Clin Immunol 16(5):505–510. https://doi.org/10.1097/ACI.0000000000000299
Patel DS, Arunakirinathan M, Stuart A, Angunawela R (2017) Allergic eye disease. BMJ 359:j4706. https://doi.org/10.1136/bmj.j4706
Bielory L (2011) Ocular allergy. Mt Sinai J Med 78(5):740–758. https://doi.org/10.1002/msj.20291
Bielory L (2008) Ocular allergy treatment. Immunol Allergy Clin North Am 28(1):189–224. https://doi.org/10.1016/j.iac.2007.12.001.
Das AV, Donthineni PR, Prashanthi GS, Basu S (2019) Allergic eye disease in children and adolescents seeking eye care in India: electronic medical records driven big data analytics report II. Ocul Surf 17(4):683–689
Akpek EK, Dart JK, Watson S, Christen W, Dursun D, Yoo S, et al (2004) A randomized trial of topical cyclosporin 0.05% in topical steroid–resistant atopic keratoconjunctivitis. Ophthalmology 111(3):476–482.
Keklikci U, Dursun B, Cingu AK (2014) Topical cyclosporine a 0.05% eyedrops in the treatment of vernal keratoconjunctivitis-randomized placebo-controlled trial. Adv Clin Exp Med Official Organ Wroclaw Med Univ 23(3):455–461.
Kiliç A, Gürler B (2006) Topical 2% cyclosporine A in preservative-free artificial tears for the treatment of vernal keratoconjunctivitis. Can J Ophthalmol 41(6):693–698
Ricciardi L, Furci F, Stefania I (2019) H1-antihistamines for allergic diseases: old aged but not old-fashioned drugs. Int J Aller Medications 5:037
Leonardi A, Capobianco D, Benedetti N, Capobianco A, Cavarzeran F, Scalora T et al (2019) Efficacy and tolerability of ketotifen in the treatment of seasonal allergic conjunctivitis: comparison between ketotifen 0.025% and 0.05% eye drops. Ocular immunol Inflammation 27(8):1352–1356.
Mounsey AL, Gray RE (2016) Topical antihistamines and mast cell stabilizers for treating allergic conjunctivitis. Am Fam Physician 93(11):915–916
Church MK (2016) Allergy, histamine and antihistamines. Histamine and histamine receptors in health and disease. Springer, Cham, pp 321–331.
Butrus S, Portela R (2005) Ocular allergy: diagnosis and treatment. Ophthalmol Clin North Am 18(4):485–492. https://doi.org/10.1016/j.ohc.2005.07.007.
Leonardi A, Silva D, Perez FD, Bozkurt B, Sharma V, Allegri P et al (2019) Management of ocular allergy. Allergy. https://doi.org/10.1111/all.13786
Wu B, Tong J, Ran Z (2019) Tacrolimus therapy in steroid-refractory ulcerative colitis: a review. Inflamm Bowel Dis. https://doi.org/10.1093/ibd/izz068
Schneeweiss S, Doherty M, Zhu S, Funch D, Schlienger RG, Fernandez-Vidaurre C et al (2009) Topical treatments with pimecrolimus, tacrolimus and medium- to high-potency corticosteroids, and risk of lymphoma. Dermatology 219(1):7–21. https://doi.org/10.1159/000209289
Daniell M, Constantinou M, Vu HT, Taylor HR (2006) Randomised controlled trial of topical ciclosporin A in steroid dependent** allergic conjunctivitis. Br J Ophthalmol 90(4):461–464. https://doi.org/10.1136/bjo.2005.082461
Urban RC Jr, Cotlier E (1986) Corticosteroid-induced cataracts. Surv Ophthalmol 31(2):102–110
Bonini S, Bonini S, Lambiase A, Marchi S, Pasqualetti P, Zuccaro O et al (2000) Vernal keratoconjunctivitis revisited: a case series of 195 patients with long-term followup. Ophthalmology 107(6):1157–1163
Chan HH, Salmon JF (2019) Glaucoma caused by topical corticosteroid application to the eyelids. Med J Aust 210(4):142–153
Raj A, Salvador-Culla B, Anwar H, Sykakis E, Figueiredo MS, Figueiredo FC (2020) Long-term outcomes on de novo ocular hypertensive response to topical corticosteroids after corneal transplantation. Cornea 39(1):45–51
Wu YT, Truong TN, Tam C, Mendoza MN, Zhu L, Evans DJ et al (2019) Impact of topical corticosteroid pretreatment on susceptibility of the injured murine cornea to Pseudomonas aeruginosa colonization and infection. Exp Eye Res 179:1–7
**agal J, Gupta PC, Pilania RK, Ram J (2019) Systemic toxicity of topical corticosteroids. Indian J Ophthalmol 67(4):559
Cutolo CA, Barabino S, Bonzano C, Traverso CE (2019) The use of topical corticosteroids for treatment of dry eye syndrome. Ocul Immunol Inflamm 27(2):266–275
Bilgin AB, Dogan ME, Ayaz Y, Apaydin KC (2019) Comparison of efficacy and safety of loteprednol etabonate 0.5% and topical dexamethasone 1% in post-vitrectomy inflammation. Ocular Immunol Inflammation 27(2):312–318.
Ilyas H, Slonim CB, Braswell GR, Favetta JR, Schulman M (2004) Long-term safety of loteprednol etabonate 0.2% in the treatment of seasonal and perennial allergic conjunctivitis. Eye Contact Lens 30(1):10–13.
Öner V, Türkcü FM, Taş M, Alakuş MF, İşcan Y (2012) Topical loteprednol etabonate 0.5% for treatment of vernal keratoconjunctivitis: efficacy and safety. Japan J Ophthalmol 56(4):312–318.
Kraft M, Soost S, Worm M (2020) Topical and systemic corticosteroids. Kanerva’s Occup Dermatol, pp 1433–1444.
Simpson EL, Flohr C, Eichenfield LF, Bieber T, Sofen H, Taïeb A et al (2018) Efficacy and safety of lebrikizumab (an anti-IL-13 monoclonal antibody) in adults with moderate-to-severe atopic dermatitis inadequately controlled by topical corticosteroids: a randomized, placebo-controlled phase II trial (TREBLE). J Am Acad Dermatol 78(5):863–871.e11.
Armstrong S, Green P (2018) “Apply sparingly”? A review of topical corticosteroids. Dalhousie Med J 45(1).
Corpuz M, Johnstone T, Kazarian A, Ostrom R (2018) Inhaled corticosteroids stimulate cAMP production and enhance βAR signaling in a non-genomic fashion in human airway smooth muscle cells. FASEB J 32(1_supplement):686.
Chalmers JR, Axon E, Harvey J, Santer M, Ridd MJ, Lawton S et al (2019) Different strategies for using topical corticosteroids in people with eczema. Cochrane Database Syst Rev 2019(6).
Yang Q, Oranje AP, Panda S, van der Wouden JC, Moed H (2018) Different strategies for using topical corticosteroids for established eczema. Cochrane Database System Rev 2018(1).
Zhang Y, Xu J, Fang F (2019) Future directions for corticosteroids in treatment of sepsis—reply. JAMA Internal Med 179(6):845.
Lim SY, Bolster MB (2019) Corticosteroids 28. Neurorheumatology: A Comprehenisve Guide to Immune Mediated Disorders of the Nervous System, 261.
Erdinest N, Ben-Eli H, Solomon A (2019) Topical tacrolimus for allergic eye diseases. Curr Opin Allergy Clin Immunol 19(5):535–543
Erdinest N, Solomon A (2014) Topical immunomodulators in the management of allergic eye diseases. Curr Opin Allergy Clin Immunol 14(5):457–463. https://doi.org/10.1097/ACI.0000000000000089
Singalavanija S, Noppakun N, Limpongsanuruk W, Wisuthsarewong W, Aunhachoke K, Chunharas A et al (2006) Efficacy and safety of tacrolimus ointment in pediatric Patients with moderate to severe atopic dermatitis. J Med Assoc Thai 89(11):1915–1922
Shoughy SS, Jaroudi MO, Tabbara KF (2016) Efficacy and safety of low-dose topical tacrolimus in vernal keratoconjunctivitis. Clin Ophthalmol 10:643–647. https://doi.org/10.2147/OPTH.S99157
Miyazaki D, Fukushima A, Ohashi Y, Ebihara N, Uchio E, Okamoto S et al (2017) Steroid-sparing effect of 0.1% Tacrolimus eye drop for treatment of shield ulcer and corneal epitheliopathy in refractory allergic ocular diseases. Ophthalmology 124(3):287–294. https://doi.org/10.1016/j.ophtha.2016.11.002.
Liendo VL, Vola ME, Barreiro TP, Wakamatsu TH, Gomes JP, Santos MSod (2017). Topical tacrolimus for the treatment of severe allergic keratoconjunctivitis in children. Arquivos Brasileiros de Oftalmologia 80(4):211–214.
Kheirkhah A, Zavareh MK, Farzbod F, Mahbod M, Behrouz MJ (2011) Topical 0.005% tacrolimus eye drop for refractory vernal keratoconjunctivitis. Eye (Lond) 25(7):872–880. https://doi.org/10.1038/eye.2011.75.
Chatterjee S, Agrawal D (2016) Tacrolimus in corticosteroid-refractory vernal keratoconjunctivitis. Cornea 35(11):1444–1448. https://doi.org/10.1097/ICO.0000000000000918
Benaim D, Ttart F, Bauvin O, Delcampe A, Joly P, Muraine M et al (2019) Tacrolimus ointment in the management of atopic keratoconjunctivitis. J Francais d'Ophtalmologie 42(4):e147–e151
Barot RK, Shitole SC, Bhagat N, Patil D, Sawant P, Patil K (2016) Therapeutic effect of 0.1% tacrolimus eye ointment in allergic ocular diseases. J Clin Diagn Res JCDR 10(6):NC05.
BenEzra D, Pe’er J, Brodsky M, Cohen E (1986) Cyclosporine eyedrops for the treatment of severe vernal keratoconjunctivitis. Am J Ophthalmol 101(3):278–282
Secchi AG, Tognon MS, Leonardi A (1990) Topical use of cyclosporine in the treatment of vernal keratoconjunctivitis. Am J Ophthalmol 110(6):641–645
Bleik JH, Tabbara KF (1991) Topical cyclosporine in vernal keratoconjunctivitis. Ophthalmology 98(11):1679–1684
Matsuda S, Koyasu S (2000) Mechanisms of action of cyclosporine. Immunopharmacology 47(2–3):119–125. https://doi.org/10.1016/s0162-3109(00)00192-2
Kosrirukvongs P, Luengchaichawange C (2004) Topical cyclosporine 0.5 per cent and preservative-free ketorolac tromethamine 0.5 per cent in vernal keratoconjunctivitis. J Med Assoc Thai 87(2):190–197.
Spadavecchia L, Fanelli P, Tesse R, Brunetti L, Cardinale F, Bellizzi M et al (2006) Efficacy of 1.25% and 1% topical cyclosporine in the treatment of severe vernal keratoconjunctivitis in childhood. Pediatr Allergy Immunol 17(7):527–532. https://doi.org/10.1111/j.1399-3038.2006.00427.x
Peters DH, Fitton A, Plosker GL, Faulds DT (1993) A review of its pharmacology, and therapeutic potential in hepatic and renal transplantation. Drugs 46(4):746–794.
Hooks MA (1994) Tacrolimus, a new immunosuppressant—a review of the literature. Ann Pharmacother 28(4):501–511
Laskow DA, Neylan JF III, Shapiro RS, Pirsch JD, Vergne-Marini PJ, Tomlanovich SJ (1998) The role of tacrolimus in adult kidney transplantation: a review. Clin Transplant 12(6):489–503
Shapiro R (1998) Tacrolimus in pediatric renal transplantation: a review. Pediatr Transplant 2(4):270–276
Sengoku T, Morita K, Sakuma S, Motoyama Y, Goto T (1999) Possible inhibitory mechanism of FK506 (tacrolimus hydrate) ointment for atopic dermatitis based on animal models. Eur J Pharmacol 379(2–3):183–189. https://doi.org/10.1016/s0014-2999(99)00500-2
Sakuma S, Higashi Y, Sato N, Sasakawa T, Sengoku T, Ohkubo Y et al (2001) Tacrolimus suppressed the production of cytokines involved in atopic dermatitis by direct stimulation of human PBMC system.(Comparison with steroids). Int Immunopharmacol 1(6):1219–1226.
Soter NA, Fleischer AB Jr, Webster GF, Monroe E, Lawrence I (2001) Tacrolimus ointment for the treatment of atopic dermatitis in adult patients: part II, safety. J Am Acad Dermatol 44(1 Suppl):S39–S46. https://doi.org/10.1067/mjd.2001.109817
Kang S, Paller A, Soter N, Satoi Y, Rico MJ, Hanifin JM (2003) Safe treatment of head/neck AD with tacrolimus ointment. J Dermatol Treat 14(2):86–94
Lan CC, Lin CT, Chen GS, Huang CC, Chen YT, Wang LF (2003) Tacrolimus ointment for the treatment of atopic dermatitis: report of first clinical experience in Taiwan. Kaohsiung J Med Sci 19(6):296–303
Rikkers SM, Holland GN, Drayton GE, Michel FK, Torres MF, Takahashi S (2003) Topical tacrolimus treatment of atopic eyelid disease. Am J Ophthalmol 135(3):297–302. https://doi.org/10.1016/s0002-9394(02)01982-7
Fung JJ (2004) Tacrolimus and transplantation: a decade in review. Transplantation 77(9 Suppl):S41–S43. https://doi.org/10.1097/01.tp.000126926.61434.a5
Moher D, Liberati A, Tetzlaff J, Altman DG (2010) Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. Int J Surg 8(5):336–341
Higgins JP, Green S (2008) Cochrane handbook for systematic reviews of interventions. The Cochrane Collaboration. http://www.cochrane-handbook.org
Cumpston MS, McKenzie JE, Welch VA, Brennan SE (2022) Strengthening systematic reviews in public health: guidance in the Cochrane Handbook for Systematic Reviews of Interventions. J Public Health 44(4):e588–e592
Gupta V, Sahu PK (2001) Topical cyclosporin A in the management of vernal keratoconjunctivitis. Eye (Lond) 15(Pt 1):39–41. https://doi.org/10.1038/eye.2001.10
Hingorani M, Moodaley L, Calder VL, Buckley RJ, Lightman S (1998) A randomized, placebo-controlled trial of topical cyclosporin A in steroid-dependent atopic keratoconjunctivitis. Ophthalmology 105(9):1715–1720
Pucci N, Novembre E, Cianferoni A, Lombardi E, Bernardini R, Caputo R et al (2002) Efficacy and safety of cyclosporine eyedrops in vernal keratoconjunctivitis. Ann Allergy Asthma Immunol 89(3):298–303. https://doi.org/10.1016/S1081-1206(10)61958-8
Zhang X, Tang J, Wang Q (2014) Analysis on tacrolimus efficacy in the treatment of vernal keratoconjunctivitis. Int Eye Sci 14:666–669
Leonardi A, Doan S, Amrane M, Ismail D, Montero J, Németh J et al (2019) A randomized, controlled trial of cyclosporine a cationic emulsion in pediatric vernal keratoconjunctivitis: the VEKTIS study. Ophthalmology 126(5):671–681
Ohashi Y, Ebihara N, Fujishima H, Fukushima A, Kumagai N, Nakagawa Y et al (2010) A randomized, placebo-controlled clinical trial of tacrolimus ophthalmic suspension 0.1% in severe allergic conjunctivitis. J Ocul Pharmacol Ther 26(2):165–174. https://doi.org/10.1089/jop.2009.0087.
Deeks JJ, Higgins JP, Altman DG, Group CSM (2019) Analysing data and undertaking meta‐analyses. Cochrane handbook for systematic reviews of interventions, pp 241–284.
Ebihara N, Ohashi Y, Uchio E, Okamoto S, Kumagai N, Shoji J et al (2009) A large prospective observational study of novel cyclosporine 0.1% aqueous ophthalmic solution in the treatment of severe allergic conjunctivitis. J Ocul Pharmacol Ther 25(4):365–372. https://doi.org/10.1089/jop.2008.0103.
Pucci N, Caputo R, Mori F, De LC, Di GL, Massai C et al (2010) Long-term safety and efficacy of topical cyclosporine in 156 children with vernal keratoconjunctivitis. Int J Immunopathol Pharmacol 23(3):865–871
Labcharoenwongs P, Jirapongsananuruk O, Visitsunthorn N, Kosrirukvongs P, Saengin P, Vichyanond P (2012) A double-masked comparison of 0.1% tacrolimus ointment and 2% cyclosporine eye drops in the treatment of vernal keratoconjunctivitis in children. Asian Pac J Allergy Immunol 30(3):177–184.
Vichyanond P, Kosrirukvongs P (2013) Use of cyclosporine A and tacrolimus in treatment of vernal keratoconjunctivitis. Curr Allergy Asthma Rep 13(3):308–314. https://doi.org/10.1007/s11882-013-0345-0
Utine CA, Stern M, Akpek EK (2010) Clinical review: topical ophthalmic use of cyclosporin A. Ocul Immunol Inflamm 18(5):352–361. https://doi.org/10.3109/09273948.2010.498657
Survase SA, Kagliwal LD, Annapure US, Singhal RS (2011) Cyclosporin A—a review on fermentative production, downstream processing and pharmacological applications. Biotechnol Adv 29(4):418–435. https://doi.org/10.1016/j.biotechadv.2011.03.004
Keklikci U, Soker SI, Sakalar YB, Unlu K, Ozekinci S, Tunik S (2008) Efficacy of topical cyclosporin A 0.05% in conjunctival impression cytology specimens and clinical findings of severe vernal keratoconjunctivitis in children. Jpn J Ophthalmol 52(5):357–562. https://doi.org/10.1007/s10384-008-0577-z.
Yamanaka M, Yokota S, Iwao Y, Noguchi S, Itai S (2014) Development and evaluation of a tacrolimus cream formulation using a binary solvent system. Int J Pharm 464(1–2):19–26. https://doi.org/10.1016/j.ijpharm.2014.01.017
Ebihara N, Ohashi Y, Fujishima H, Fukushima A, Nakagawa Y, Namba K et al (2012) Blood level of tacrolimus in patients with severe allergic conjunctivitis treated by 0.1% tacrolimus ophthalmic suspension. Allergol Int 61(2):275–282. https://doi.org/10.2332/allergolint.11-OA-0349
Harada N, Inada N, Ishimori A, Shoji J, Sawa M (2014) Follow-up study on patients with vernal keratoconjunctivitis undergoing topical 0.1% tacrolimus treatment. Nihon Ganka Gakkai Zasshi 118(4):378–384.
Yazu H, Shimizu E, Aketa N, Dogru M, Okada N, Fukagawa K et al (2019) The efficacy of 0.1% tacrolimus ophthalmic suspension in the treatment of severe atopic keratoconjunctivitis. Ann Allergy Asthma Immunol 122(4):387–392
Won CH, Seo PG, Park YM, Yang JM, Lee KH, Sung KJ et al (2004) A multicenter trial of the efficacy and safety of 0.03% tacrolimus ointment for atopic dermatitis in Korea. J Dermatolo Treatment 15(1):30–34.
Schachner LA, Lamerson C, Sheehan MP, Boguniewicz M, Mosser J, Raimer S et al (2005) Tacrolimus ointment 0.03% is safe and effective for the treatment of mild to moderate atopic dermatitis in pediatric patients: results from a randomized, double-blind, vehicle-controlled study. Pediatrics 116(3):e334–e42.
Virtanen HM, Reitamo S, Kari M, Kari O (2006) Effect of 0.03% tacrolimus ointment on conjunctival cytology in patients with severe atopic blepharoconjunctivitis: a retrospective study. Acta Ophthalmol Scand 84(5):693–695. https://doi.org/10.1111/j.1600-0420.2006.00699.x.
Attas-Fox L, Barkana Y, Iskhakov V, Rayvich S, Gerber Y, Morad Y et al (2008) Topical tacrolimus 0.03% ointment for intractable allergic conjunctivitis: an open-label pilot study. Curr Eye Res 33(7):545–549. https://doi.org/10.1080/02713680802149115.
Kymionis GD, Goldman D, Ide T, Yoo SH. Tacrolimus ointment 0.03% in the eye for treatment of giant papillary conjunctivitis. Cornea. 2008;27(2):228–9. doi: https://doi.org/10.1097/ICO.0b013e318159afbb [doi];00003226–200802000–00020 [pii].
Tam PM, Young AL, Cheng LL, Lam PT (2010) Topical tacrolimus 0.03% monotherapy for vernal keratoconjunctivitis--case series. Br J Ophthalmol 94(10):1405–1406. https://doi.org/10.1136/bjo.2009.172387
Al-Amri AM, Fiorentini SF, Albarry MA, Bamahfouz AY (2017) Long-term use of 0.003% tacrolimus suspension for treatment of vernal keratoconjunctivitis. Oman J Ophthalmol 10(3):145.
Samyukta SK, Pawar N, Ravindran M, Allapitchai F, Rengappa R (2019) Monotherapy of topical tacrolimus 0.03% in the treatment of vernal keratoconjunctivitis in the pediatric population. J Am Assoc Pediatric Ophthalmol Strabismus 23(1):36-e1
Ueda Y, Tomoe H, Takahashi H, Takahashi Y, Yamashita H, Kaneko H et al (2014) Interstitial cystitis associated with primary Sjogren’s syndrome successfully treated with a combination of tacrolimus and corticosteroid: a case report and literature review. Mod Rheumatol. https://doi.org/10.3109/14397595.2014.895283
Vichyanond P, Tantimongkolsuk C, Dumrongkigchaiporn P, Jirapongsananuruk O, Visitsunthorn N, Kosrirukvongs P (2004) Vernal keratoconjunctivitis: result of a novel therapy with 0.1% topical ophthalmic FK-506 ointment. J Allergy Clin Immunol 113(2):355–358.
Zhao M, He F, Yang Y, Lin W, Qiu W, Meng Q et al (2022) Therapeutic efficacy of tacrolimus in vernal keratoconjunctivitis: a meta-analysis of randomised controlled trials. Eur J Hosp Pharm 29(3):129–133
Wan KH-N, Chen LJ, Rong SS, Pang CP, Young AL (2013) Topical cyclosporine in the treatment of allergic conjunctivitis: a meta-analysis. Ophthalmology 120(11):2197–203.
Acknowledgements
Dr. Nir Erdinest is grateful to the Azrieli Foundation for the award of the Post-doc Azrieli Fellowship and Hadassah-Hebrew University Medical Center for the award of post-doctoral fellows.
Funding
No funding was received for this research.
Author information
Authors and Affiliations
Contributions
Conceptualization and Methodology: DBEN, NE, IL; Formal analysis and investigation: DBEN, NE; Writing - original draft preparation: DBEN, NE, NL; Writing - review and editing: NL, NL, DL, AS, YM, SN
Corresponding author
Ethics declarations
Conflict of interest
No conflicts of interest. All authors certify that they have no affiliations with or involvement in any organization or entity with any financial interest (such as honoraria, educational grants, participation in speakers’ bureaus, membership, employment, consultancies, stock ownership or other equity interest and expert testimony or patent-licensing arrangements), or non-financial interest (such as personal or professional relationships, affiliations, knowledge or beliefs) in the subject matter or materials discussed in this manuscript.
Ethical approval
This article contains no studies with human participants or animals performed by any of the authors.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Erdinest, N., Noyman, D.B.E., London, N. et al. Applications of topical immunomodulators enhance clinical signs of vernal keratoconjunctivitis (VKC) and atopic keratoconjunctivitis (AKC): a meta-analysis. Int Ophthalmol 44, 157 (2024). https://doi.org/10.1007/s10792-024-03097-7
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10792-024-03097-7