Abstract
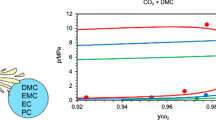
This paper investigates the phase equilibrium conditions in the carbon dioxide hydrate forming system. Carbon dioxide hydrate can be utilized for carbon capture, salt manufacture, carbonated solid foods and tritium water concentration, so the phase equilibrium conditions have been substantially reported so far. However, the data from previous studies were inconsistent with each other, such as there is a difference of 1.0 K in the phase equilibrium temperature at 2 MPa. In this study, the newly three-phase (water rich liquid + hydrate + guest rich vapor) equilibrium conditions in the carbon dioxide hydrate forming system were measured at twenty different temperature conditions within the range of (271.9-282.7) K in the two different laboratories. The six pairs of the three-phase equilibrium condition data measured under equivalent pressure conditions were consistent within mutual uncertainties. The internal consistency of the data measured in this study was evaluated by the Clausius-Clapeyron equation. The data measured in this study existed within the uncertainty range of the data from several previous studies.









Similar content being viewed by others
Data Availability
The data and materials used in this study are available upon request to the corresponding author.
References
H. Kiyokawa, S. Horii, S. Alavi, R. Ohmura, Fuel. 278, 118330 (2020). https://doi.org/10.1016/j.fuel.2020.118330
M. Maruyama, S. Kao, H. Kiyokawa, S. Takeya, R. Ohmura, Energy Fuels. 36(18), 10601–10609 (2022). https://doi.org/10.1021/acs.energyfuels.2c01355
B. Castellani, F. Rossi, M. Filipponi, A. Nicolini, Biomass Bioenergy. 70, 330–338 (2014). https://doi.org/10.1016/j.biombioe.2014.08.026
M. Tanaka, K. Tsugane, D. Suga, S. Tomura, R. Ohmura, K. Yasuda, ACS Sustainable Chem. Eng. 9, 9078–9084 (2021). https://doi.org/10.1021/acssuschemeng.1c023
A. Gibo, S. Nakano, S. Shiraishi, S. Takeya, S. Tomura, R. Ohmura, K. Yasuda, Desalination. 539, 115937 (2022). https://doi.org/10.1016/j.desal.2022.115937
M. Maruyama, S. Tomura, K. Yasuda, R. Ohmura, J. Clean. Prod. 383, 135425 (2023). https://doi.org/10.1016/j.jclepro.2022.135425
S. Shigehara, R. Ohmura, Food Chem. 371, 131369 (2022). https://doi.org/10.1016/j.foodchem.2021.131369
T.B. Peters, J.L. Smith, J.G. Brisson, J. Food Eng. 100(4), 669–677 (2010). https://doi.org/10.1016/j.jfoodeng.2010.05.017
S. Nakamura, T. Awata, H. Kiyokawa, H. Ito, R. Ohmura, Chem. Eng. J. (2023). https://doi.org/10.1016/j.cej.2023.142979
S. Adisasmito, R.J. Frank, E.D. Sloan, J. Chem. Eng. Data. 36, 68–71 (1991). https://doi.org/10.1021/je00001a020
K. Yasuda, R. Ohmura, J. Chem. Eng. Data. 53(9), 2182–2188 (2008). https://doi.org/10.1021/je800396v
Y. Nema, R. Ohmura, I. Senaha, K. Yasuda, Fluid Phase Equilib. 441, 49–53 (2017). https://doi.org/10.1016/j.fluid.2016.12.014
M.K. Chun, J.H. Yoon, H. Lee, J. Chem. Eng. Data. 41(5), 1114–1116 (1996). https://doi.org/10.1021/je9601218
K. Ohgaki, Y. Makihara, K. Takano, J. Chem. Eng. Japan. 26(5), 558–564 (1993). https://doi.org/10.1252/jcej.26.558
M.M. Mooijer-van den, R. Heuvel, C.J. Witteman, Peters, Fluid Phase Equilib. 182, 97–110 (2001). https://doi.org/10.1016/s0378-3812(01)00384-3
S.S. Fan, T.M. Guo, J. Chem. Eng. Data. 44(4), 829–832 (1999). https://doi.org/10.1021/je990011b
H.J. Ng, D.B. Robinson, Fluid Phase Equilib. 21(1–2), 145–155 (1985). https://doi.org/10.1016/0378-3812(85)90065-2
Y.T. Seo, H. Lee, J.H. Yoon, J. Chem. Eng. Data. 46(2), 381–384 (2001). https://doi.org/10.1021/je000237a
T. Maekawa, Fluid Phase Equilib. 303(1), 76–79 (2011). https://doi.org/10.1016/j.fluid.2011.01.011
M. Wang, Z.G. Sun, X.H. Qiu, M.G. Zhu, C.H. Li, A.J. Zhang, J. Li, C.M. Li, H.F. Huang, J. Chem. Eng. Data. 62(2), 812–815 (2017). https://doi.org/10.1021/acs.jced.6b00848
M.L. Dai, Z.G. Sun, J. Lim, H.F. Huang, J. Chem. Thermodyn. 148 (2020). https://doi.org/10.1016/j.jct.2020.106144
S.S. Fan, G.J. Chen, Q.L. Ma, T.M. Guo, Chem. Eng. J. 78, 173–178 (2000). https://doi.org/10.1016/S1385-8947(00)00157-1
A.H. Mohammadi, R. Anderson, B. Tohidi, AIChE J. 51(10), 2825–2833 (2005). https://doi.org/10.1002/aic.10526
C.H. Unruh, D.L. Katz, Trans. AIME. 4, 83–86 (1949). https://doi.org/10.2118/949983-G
P.F. Ferrari, A.Z. Guembaroski, M.A.M. Neto, R.E.M. Morales, A.K. Sum, Fluid Phase Equilib. 412, 176–183 (2016). https://doi.org/10.1016/j.fluid.2015.10.008
K.M. Sabil, G. Witkamp, C.J. Peters, Fluid Phase Equilib. 284, 38–43 (2009). https://doi.org/10.1016/j.fluid.2009.06.006
P. Englezos, S. Hall, Can. J. Chem. Eng. 72, 887 (1994). https://doi.org/10.1002/cjce.5450720516
K. Tezuka, R. Shen, T. Watanabe, S. Takeya, S. Alavi, J.A. Ripmeester, R. Ohmura, Chem. Com. 49, 505–507 (2013). https://doi.org/10.1039/c2cc37717a
B. Tohidi, R.W. Burgass, A. Danesh, K.K. Ostergaard, A.C. Todd, Ann. N Y Acad. Sci. 912(1), 924–931 (2000). https://doi.org/10.1111/j.1749-6632.2000.tb06846.x
Funding
Not applicable.
Author information
Authors and Affiliations
Contributions
K.Y. and R.O. conceived the idea of the study. A.G., S.S., and H.I. are conducted the measurements and interpreted the data. H.I. wrote the main manuscript text. K.Y. and R.O. provided supervision and made corrections to the manuscript. All authors reviewed it.
Corresponding author
Ethics declarations
Compering Interest
Not applicable.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Ito, H., Gibo, A., Shiraishi, S. et al. Renewed Measurements of Carbon Dioxide Hydrate Phase Equilibrium. Int J Thermophys 44, 128 (2023). https://doi.org/10.1007/s10765-023-03241-y
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10765-023-03241-y