Abstract
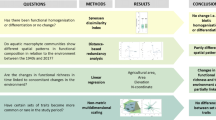
In this study, we aimed at investigating the mechanisms underlying the coexistence of a native and an invasive macrophyte (Egeria najas and Hydrilla verticillata), by analyzing the overlap and breadth of their functional and environmental niches in the Upper Paraná River basin (Brazil). We adopted a probabilistic hypervolume approach using relative niche size and uniqueness as metrics of niche breadth and divergence. We compared the two species niches and the variance within each species’ populations when occurring in monospecific stands or in mixed growth conditions. We found that the two species have partially different functional niches and they both expand their functional niche when co-occurring, revealing increased traits variability. This suggests that both species have a different and higher ability to exploit available resources when co-occurring with each other, which might explain their coexistence. However, H. verticillata was not greatly affected by the presence of E. najas since it does not show any environmental niche shift in its presence; nonetheless the latter species is still able to thrive given its more generalist behavior in terms of environmental conditions. Our results suggest that coexistence between the investigated native and invasive species is facilitated more profoundly by functional rather than environmental adaptations.







Similar content being viewed by others
Data availability
The authors declare that the data supporting the findings of this study are available within the paper and its Supplementary Information file. Should any raw data files be needed in another format they are available from the corresponding author upon reasonable request.
References
Alahuhta, J. & J. Heino, 2013. Spatial extent, regional specificity and metacommunity structuring in lake macrophytes. Journal of Biogeography 40: 1572–1582. https://doi.org/10.1111/jbi.12089.
Agostinho, A. A., Thomaz, S. M., & Gomes, L. C., 2004. Threats for biodiversity in the floodplain of the Upper Paraná River: effects of hydrological regulation by dams. International Journal of Ecohydrology & Hydrobiology 4(3): 267–280. https://www.cabidigitallibrary.org/doi/full/10.5555/20043189621.
Anderson, L. G., P. O. J. Hall, A. Iverfeldt, M. M. R. Van Der Loeff, B. Sundby & S. F. G. Westerlund, 1986. Benthic respiration measured by total carbonate production. Limnology and Oceanography 31: 319–329.
Aspila, K. I., H. Agemian & A. S. Y. Chau, 1976. A semiauto- mated method for the determination of inorganic, organic and total phosphate in sediments. Analyst 101: 187–197.
Blonder, B., C. B. Morrow, B. Maitner, D. J. Harris, C. Lamanna, C. Violle, B. J. Enquist & A. J. Kerkhoff, 2018. New approaches for delineating n-dimensional hypervolumes. Methods in Ecology and Evolution 9(2): 305–319. https://doi.org/10.1111/2041-210X.12865.
Blonder, B., C. B. Morrow, S. Brown, G. Butruille, D. Chen, A. Laini, & D. J. Harris, 2023. hypervolume: High Dimensional Geometry, Set Operations, Projection, and Inference Using Kernel Density Estimation, Support Vector Machines, and Convex Hulls. R package version 3.1.3, https://CRAN.R-project.org/package=hypervolume.
Bremner, J. M., 1965. Inorganic forms of nitrogen. Methods of soil analysis: Part 2 chemical and microbiological properties 9: 1179–1237.
Chmara, R., E. Pronin, & J. Szmeja, 2021. Functional macrophyte trait variation as a response to the source of inorganic carbon acquisition. PeerJ 9: e12584, https://peerj.com/articles/12584.
Cook, C. D. K., & R. Lüönd, 1982. A revision of the genus Hydrocharis (hydrocharitaceae). Aquatic Botany 14: 177–204, https://linkinghub.elsevier.com/retrieve/pii/0304377082900973.
Croft, H., J. Chen, X. Luo, P. Bartlett, B. Chen & R. M. Staebler, 2017. Leaf chlorophyll content as a proxy for leaf photosynthetic capacity. Global Change Biology 23(9): 3513–3524.
Dalla Vecchia, A., P. Villa & R. Bolpagni, 2020. Functional traits in macrophyte studies: Current trends and future research agenda. Aquatic Botany 167: 103290. https://doi.org/10.1016/j.aquabot.2020.103290.
Dalle Fratte, M., R. Bolpagni, G. Brusa, M. Caccianiga, S. Pierce, M. Zanzottera & B. E. L. Cerabolini, 2019. Alien plant species invade by occupying similar functional spaces to native species. Flora 257: 151419. https://doi.org/10.1016/j.flora.2019.151419.
Donovan, L. A., H. Maherali, C. M. Caruso, H. Huber & H. de Kroon, 2011. The evolution of the worldwide leaf economics spectrum. Trends in Ecology and Evolution 26: 88–95.
Efremov, A., Y. Bolotova, A. Mesterházy, & C. Toma, 2018. Features of Distribution of Hydrilla verticillata (L. fil.) Royle (Hydrocharitaceae) in North Eurasia. Journal of Coastal Research 34: 675, https://bioone.org/journals/journal-of-coastal-research/volume-34/issue-3/JCOASTRES-D-17–00072.1/Features-of-Distribution-of-Hydrilla-verticillata-L-fil-Royle-Hydrocharitaceae/https://doi.org/10.2112/JCOASTRES-D-17-00072.1.full.
Fasoli, J. V. B., R. P. Mormul, E. R. Cunha & S. M. Thomaz, 2018. Plasticity responses of an invasive macrophyte species to inorganic carbon availability and to the interaction with a native species. Hydrobiologia 817: 227–237. https://doi.org/10.1007/s10750-018-3543-x.
Ferrareze, M., L. Casatti & M. G. Nogueira, 2014. Spatial heterogeneity affecting fish fauna in cascade reservoirs of the Upper Parana´ Basin, Brazil. Hydrobiologia 738: 97.
Firn, J., J. M. McGree, E. Harvey, H. Flores-Moreno, M. Schütz, Y. M. Buckley, E. T. Borer, E. W. Seabloom, K. J. La Pierre, A. M. MacDougall, S. M. Prober, C. J. Stevens, L. L. Sullivan, E. Porter, E. Ladouceur, C. Allen, K. H. Moromizato, J. W. Morgan, W. S. Harpole, Y. Hautier, N. Eisenhauer, J. P. Wright, P. B. Adler, C. A. Arnillas, J. D. Bakker, L. Biederman, A. A. D. Broadbent, C. S. Brown, M. N. Bugalho, M. C. Caldeira, E. E. Cleland, A. Ebeling, P. A. Fay, N. Hagenah, A. R. Kleinhesselink, R. Mitchell, J. L. Moore, C. Nogueira, P. L. Peri, C. Roscher, M. D. Smith, P. D. Wragg, & A. C. Risch, 2019. Leaf nutrients, not specific leaf area, are consistent indicators of elevated nutrient inputs. Nature Ecology & Evolution 3: 400–406, https://www.nature.com/articles/s41559-018-0790-1.
Florêncio, F. M., D. C. Alves, F. M. Lansac-Tôha, M. J. Silveira, & S. M. Thomaz, 2021a. The success of the invasive macrophyte Hydrilla verticillata and its interactions with the native Egeria najas in response to environmental factors and plant abundance in a subtropical reservoir. Aquatic Botany 175: 103432. https://linkinghub.elsevier.com/retrieve/pii/S0304377021000814
Florêncio, F. M., M. J. Silveira, & S. M. Thomaz, 2021b. Niche differentiation between a native and an invasive species of submersed macrophyte in a subtropical reservoir. Acta Botanica Brasilica 35: 132–139, http://www.scielo.br/scielo.php?script=sci_arttext&pid=S0102-33062021000600132&tlng=en.
Fu, H., J. Zhong, G. Yuan, L. Ni, P. **e & T. Cao, 2014. Functional traits composition predict macrophytes community productivity along a water depth gradient in a freshwater lake. Ecology and Evolution 4: 1516–1523.
Fu, H., J. Zhong, G. Yuan, C. Guo, Q. Lou, W. Zhang, J. Xu, L. Ni, P. **e & T. Cao, 2015. Predicting changes in macrophyte community structure from functional traits in a freshwater lake: A test of maximum entropy model. Plos One 10: e0131630. https://doi.org/10.1371/journal.pone.0131630.
Haug, E. J., J. T. Harris & R. J. Richardson, 2019. Monoecious Hydrilla verticillata development in complete darkness. Aquatic Botany Elsevier 154: 28–34. https://doi.org/10.1016/j.aquabot.2018.12.005.
Hess, M. C. M., F. Mesléard & E. Buisson, 2019. Priority effects: Emerging principles for invasive plant species management. Ecological Engineering 127: 48–57. https://doi.org/10.1016/j.ecoleng.2018.11.011.
Huston, M. A., 1999. Local process and regional patterns: Appropiate scales for understanding variation in the diversity of plants and animals. Oikos 86: 393–401. https://doi.org/10.2307/3546645.
Hutchinson, G., 1957. Concluding remarks. Cold Spring Harbor Symposia on Quantitative Biology 22: 415–427.
Laini, A., T. Datry & B. W. Blonder, 2023. N-dimensional hypervolumes in trait-based ecology: Does occupancy rate matter? Functional Ecology. 37: 1802–1814. https://doi.org/10.1111/1365-2435.14344.
Langeland, K. A., 1996. Hydrilla tuber formation in response to single and sequential bensulfuron methyl exposures at different times. Hydrobiologia 340: 247–251. https://doi.org/10.1007/BF00012763.
Letten, A. D., P.-J. Ke & T. Fukami, 2017. Linking modern coexistence theory and contemporary niche theory. Concepts and Synthesis 87: 161–177.
Lienig, D., H. L. Golterman, R. S. Clymo & M. A. M. Ohnstadt, 1978. Methods for physical and chemical analysis of freshwaters—1BP Handbook No 8, 2nd ed. Blackwell Scientific Publications, Oxford:
Liu, H., W. Zhou, X. Li, Q. Chu, N. Tang, B. Shu, G. Liu & W. **ng, 2020. How many submerged macrophyte species are needed to improve water clarity and quality in Yangtze floodplain lakes? Science of the Total Environment 724: 138267. https://doi.org/10.1016/j.scitotenv.2020.138267.
Liu, Q., F. J. Sterck, J. L. Zhang, A. Scheire, E. Konings, M. Cao, L. Q. Sha & L. Poorter, 2021. Traits, strategies, and niches of liana species in a tropical seasonal rainforest. Oecologia 196: 499–514. https://doi.org/10.1007/S00442-021-04937-4/FIGURES/4.
Lorenzen, C. J., 1967. Determination of chlorophyll and phaeopigments: spectrophotometric equations. Limnology and Oceanography 12: 343–346.
Lucio, L. C., S. M. Thomaz, S. Prioli, A. Maria, T. A. Boni, A. V. Oliveira & A. J. Prioli, 2019. Molecular characterization of the invasive aquatic macrophyte Hydrilla verticillata (Hydrocharitaceae) in Brazil. Anais Da Academia Brasileira De Ciências 91: e20180494.
Ma, F., Z. Zuo, L. Yang, D. Li, H. Wang, F. Li, S. Fan, C. Liu & D. Yu, 2022. The effect of trait-based diversity on productivity results mainly from intraspecific trait variability in the macrophyte community. Freshwater Biology 67: 1137–1149. https://doi.org/10.1111/fwb.13906.
MacArthur, R. & R. Levins, 1967. The limiting similarity, convergence, and divergence of coexisting species. The American Naturalist 101: 377–385. https://doi.org/10.2307/2459090.
Marcondes D. A. S., A. L. Mustafá & R. H. Tanaka, 2003. Estudos para manejo integrado de plantas aquáticas no reservatório de Jupiá. In: Thomaz SM, Bini LM (eds) Ecologia e manejo de macrófitas aquáticas. EDUEM, Maringá, pp 299–317.
Mayfield, M. M. & J. M. Levine, 2010. Opposing effects of competitive exclusion on the phylogenetic structure of communities. Ecology Letters 13: 1085–1093. https://doi.org/10.1111/j.1461-0248.2010.01509.x.
Mi, X., Z. Sun, Y. Song, X. Liu, J. Yang, J. Wu, X. Ci, J. Li, L. Lin, M. Cao & K. Ma, 2021. Rare tree species have narrow environmental but not functional niches. Functional Ecology 35: 511–520. https://doi.org/10.1111/1365-2435.13714/SUPPINFO.
Michelan, T. S., S. M. Thomaz, R. P. Mormul, & P. Carvalho, 2010. Effects of an exotic invasive macrophyte (tropical signalgrass) on native plant community composition, species richness and functional diversity. Freshwater Biology 55: 1315–1326, http://doi.wiley.com/https://doi.org/10.1111/j.1365-2427.2009.02355.x.
Mony, C., T. J. Koschnick, W. T. Haller, & S. Muller, 2007. Competition between two invasive Hydrocharitaceae (Hydrilla verticillata (L.f.) (Royle) and Egeria densa (Planch)) as influenced by sediment fertility and season. Aquatic Botany 86: 236–242, https://linkinghub.elsevier.com/retrieve/pii/S0304377006001744.
Mormul, R. P., S. M. Thomaz & E. Jeppesen, 2020. Do interactions between eutrophication and CO2 enrichment increase the potential of elodeid invasion in tropical lakes? Biological Invasions 22: 2787–2795. https://doi.org/10.1007/s10530-020-02284-8.
Mormul, R. P., F. A. Ferreira, T. S. Michelan, P. Carvalho, M. J. Silveira, & S. M. Thomaz, 2010. Aquatic macrophytes in the large, sub-tropical Itaipu Reservoir, Brazil. Revista de Biologia Tropical 58: 1437–1452, http://revistas.ucr.ac.cr/index.php/rbt/article/view/5422.
Patrick, A. E. S. & S. Florentine, 2021. Factors affecting the global distribution of Hydrilla verticillata (L. fil.) Royle: A review. Weed Research 61: 253–271. https://doi.org/10.1111/wre.12478.
Pelicice, F. M., P. S. Pompeu & A. A. Agostinho, 2015. Large reservoirs as ecological barriers to downstream movements of Neotropical migratory fish. Fish and Fisheries 16(4): 697–715. https://doi.org/10.1111/faf.12089.
Pérez-Harguindeguy, N., S. Díaz, E. Garnier, S. Lavorel, H. Poorter, P. Jaureguiberry, M. S. Bret-Harte, W. K. Cornwell, J. M. Craine, D. E. Gurvich, C. Urcelay, E. J. Veneklaas, P. B. Reich, L. Poorter, I. J. Wright, P. Ray, L. Enrico, J. G. Pausas, A. C. de Vos, et al., 2013. New handbook for standardised measurement of plant functional traits worldwide. Australian Journal of Botany 61: 167–234. https://doi.org/10.1071/BT12225.
Pierini, S. A. & S. M. Thomaz, 2009. Effects of limnological and morphometric factors upon Zmin, Zmax and width of Egeria spp stands in a tropical reservoir. Brazilian Archives of Biology and Technology 52: 387–396. https://doi.org/10.1590/S1516-89132009000200016.
Puglielli, G., A. Bricca, S. Chelli, F. Petruzzellis, A. T. R. Acosta, G. Bacaro, E. Beccari, L. Bernardo, G. Bonari, R. Bolpagni, F. Boscutti, G. Calvia, G. Campetella, L. Cancellieri, R. Canullo, M. Carbognani, M. Carboni, M. L. Carranza, M. B. Castellani, et al., 2024. Intraspecific variability of leaf form and function across habitat types. Ecology Letters 27: e14396. https://doi.org/10.1111/ELE.14396.
Pulzatto, M. M., E. R. Cunha, M. S. Dainez-Filho & S. M. Thomaz, 2019. Association between the success of an invasive macrophyte, environmental variables and abundance of a competing native macrophyte. Frontiers in Plant Science 10: 1–11. https://doi.org/10.3389/fpls.2019.00514/full.
R Core Team, 2023. R: A language and environment for statistical computing. R Foundation for Statistical.
Riis, T., B. Olesen, J. S. Clayton, C. Lambertini, H. Brix & B. K. Sorrell, 2012. Growth and morphology in relation to temperature and light availability during the establishment of three invasive aquatic plant species. Aquatic Botany 102: 56–64. https://doi.org/10.1016/j.aquabot.2012.05.002.
Robichaud, C. D. & R. C. Rooney, 2022. Differences in above-ground resource acquisition and niche overlap between a model invader (Phragmites australis) and resident plant species: measuring the role of fitness and niche differences in the field. Biological Invasions. 24: 649–682. https://doi.org/10.1007/S10530-021-02674-6/TABLES/4.
Rosenfeld, J. S., 2002. Functional redundancy in ecology and conservation. Oikos 98: 156–162.
Schneider, C. A., W. S. Rasband & K. W. Eliceiri, 2012. NIH Image to ImageJ: 25 years of image analysis. Nature Methods 9: 671–675. https://doi.org/10.1038/nmeth.2089.
Silveira, M. J. & S. M. Thomaz, 2015. Growth of a native versus an invasive submerged aquatic macrophyte differs in relation to mud and organic matter concentrations in sediment. Aquatic Botany 124: 85–91. https://doi.org/10.1016/j.aquabot.2015.03.004.
Silveira, M. J. & S. M. Thomaz, 2019. Interspecific associations between Hydrilla verticillata and three dominant native genera of submerged macrophytes are taxa dependent. Aquatic Sciences 81: 21. https://doi.org/10.1007/s00027-018-0614-z.
Silveira, M. J. & S. M. Thomaz, 2023. Effects of interactions between abiotic and biotic factors on growth of a non-native macrophyte. Biological Invasions 25: 431–440. https://doi.org/10.1007/s10530-022-02924-1.
Silveira, M. J., S. M. Thomaz, R. P. Mormul & F. P. Camacho, 2009. Effects of desiccation and sediment type on early regeneration of plant fragments of three species of aquatic macrophytes. International Review of Hydrobiology 94: 169–178. https://doi.org/10.1002/iroh.200811086.
Silveira, M. J., D. C. Alves & S. M. Thomaz, 2018. Effects of the density of the invasive macrophyte Hydrilla verticillata and root competition on growth of one native macrophyte in different sediment fertilities. Ecological Research 33: 927–934. https://doi.org/10.1007/s11284-018-1602-4.
Sousa, W. T. Z., 2011. Hydrilla verticillata (Hydrocharitaceae), a recent invader threatening Brazil’s freshwater environments: a review of the extent of the problem. Hydrobiologia 669: 1–20. https://doi.org/10.1007/s10750-011-0696-2.
Sousa, W. T. Z., S. M. Thomaz, K. J. Murphy, M. J. Silveira & R. P. Mormul, 2009. Environmental predictors of the occurrence of exotic Hydrilla verticillata (L.f.) Royle and native Egeria najas Planch in a sub-tropical river floodplain: the Upper River Paraná, Brazil. Hydrobiologia 632: 65–78. https://doi.org/10.1007/s10750-009-9828-3.
Sousa, W. T. Z., S. M. Thomaz, & K. J. Murphy, 2010. Response of native Egeria najas Planch. and invasive Hydrilla verticillata (L.f.) Royle to altered hydroecological regime in a subtropical river. Aquatic Botany 92: 40–48, https://linkinghub.elsevier.com/retrieve/pii/S030437700900120X.
Souza, D. C., E. R. Cunha, R. D. A. Murillo, S. J. Silveira, M. M. Pulzatto, M. S. Dainez-Filho, L. A. Lolis & S. M. Thomaz, 2017. Species inventory of aquatic macrophytes in the last undammed stretch of the Upper Paraná River, Brazil. Acta Limnologica Brasiliensia. https://doi.org/10.1590/S2179-975X6017.
Thomaz, S. M., 2023. Ecosystem services provided by freshwater macrophytes. Hydrobiologia. https://doi.org/10.1007/s10750-021-04739-y.
Thomaz, S. M., P. Carvalho, R. P. Mormul, F. A. Ferreira, M. J. Silveira, & T. S. Michelan, 2009. Temporal trends and effects of diversity on occurrence of exotic macrophytes in a large reservoir. Acta Oecologica 35: 614–620, https://linkinghub.elsevier.com/retrieve/pii/S1146609X09000630.
Trémolières, M., 2004. Plant response strategies to stress and disturbance: the case of aquatic plants. Journal of Biosciences 29: 461–470. https://doi.org/10.1007/BF02712119.
Urban, R. A., J. E. Titus & W.-X. Zhu, 2006. An invasive macrophyte alters sediment chemistry due to suppression of a native isoetid. Oecologia 148: 455–463. https://doi.org/10.1007/s00442-006-0393-4.
Van, T. K., W. T. Haller, & G. Bowes, 1976. Comparison of the Photosynthetic Characteristics of Three Submersed Aquatic Plants. Plant Physiology 58: 761–768, https://academic.oup.com/plphys/article/58/6/761-768/6075292.
Vilas, M. P., C. L. Marti, M. P. Adams, C. E. Oldham & M. R. Hipsey, 2017. Invasive macrophytes control the spatial and temporal patterns of temperature and dissolved oxygen in a shallow lake: a proposed feedback mechanism of macrophyte loss. Frontiers in Plant Science 8: 1–14. https://doi.org/10.3389/fpls.2017.02097/full.
Voesenek, L. A. C. J., T. D. Colmer, R. Pierik, F. F. Millenaar & A. J. M. Peeters, 2006. How plants cope with complete submergence. New Phytologist 170: 213–226. https://doi.org/10.1111/j.1469-8137.2006.01692.x.
Vukov, D., M. Ilić, M. Ćuk, & R. Igić, 2023. Environmental Drivers of Functional Structure and Diversity of Vascular Macrophyte Assemblages in Altered Waterbodies in Serbia. Diversity 15: 231, https://www.mdpi.com/1424-2818/15/2/231.
Wahl, C., M. Kaller & R. Diaz, 2021. Invasion of floating fern alters freshwater macroinvertebrate community structure with implications for bottom-up processes. Hydrobiologia 848: 2523–2537. https://doi.org/10.1007/s10750-021-04571-4.
Wang, L., X. Wang, X. Han, Y. Gao, B. Liu, X. Zhang & G. Wang, 2021. Potamogeton crispus responses to varying water depth in morphological plasticity and physiological traits. Environmental Science and Pollution Research 28: 4253–4261. https://doi.org/10.1007/s11356-020-10806-z.
Wellburn, A. R., 1994. The spectral determination of chlorophylls A and B, as well as Total caroteinds, using various solvents with Spectrophotometers of different resolution. Journal of Plant Physiology 144: 307–313.
Williams, J. D. H., J. K. Syers & T. W. Walker, 1967. Fractionation of soil inorganic phosphate by a modification of Chang and Jackson’s procedure. Soil Science Society of American Journal 31(6): 736–739.
Wright, I. J., P. B. Reich, M. Westoby, D. D. Ackerly, Z. Baruch, F. Bongers, J. Cavender-Bares, T. Chapin, J. H. C. Cornelissen, M. Diemer, J. Flexas, E. Garnier, P. K. Groom, J. Gulias, K. Hikosaka, B. B. Lamont, T. Lee, W. Lee, C. Lusk, J. J. Midgley, M.-L. Navas, Ü. Niinemets, J. Oleksyn, N. Osada, H. Poorter, P. Poot, L. Prior, V. I. Pyankov, C. Roumet, S. C. Thomas, M. G. Tjoelker, E. J. Veneklaas, & R. Villar, 2004a. The worldwide leaf economics spectrum. Nature 428: 821–827, http://www.nature.com/articles/nature02403.
Yofukuji, K. Y., A. L. P. Cardozo, B. A. Quirino, M. H. F. Aleixo & R. Fugi, 2021. Macrophyte diversity alters invertebrate community and fish diet. Hydrobiologia 848: 913–927. https://doi.org/10.1007/s10750-020-04501-w.
Acknowledgements
FM Florêncio and RP Leal acknowledge the Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES) for providing doctoral scholarships. ADV has benefited from the equipment and framework of the COMP-HUB and COMP-R Initiative, funded by the ‘Departments of Excellence’ program of the Italian Ministry for Education, University and Research (MIUR, 2018-2022 and MUR, 2023-2027, respectively). We thank MSc. Matteo Amoruso, MSc. Beatrice Fois and all the researchers from the Aquatic Macrophyte Ecology Laboratory of the Limnology, Ichthyology and Aquaculture Research Center at the State University of Maringá for their assistance during sampling and data processing. We also offer special appreciation to Dr. Rossano Bolpagni and Dr. Sidinei Magela Thomaz for participating in the development of the ideas for this paper and for reviewing its first version. Finally, we thank the two anonymous reviewers whose comments contributed to improve this paper.
Funding
Partial financial support was received from the Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES) by providing doctoral scholarships to FM Florêncio and RP Leal.
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Material preparation, data collection and analysis were performed by Alice Dalla Vecchia, Aline Rosado, Fernanda Florêncio and Rodrigo Leal. The first draft of the manuscript was written by Fernanda Florêncio, and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Handling editor: Andre A. Padial
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Guest editors: Sidinei M. Thomaz, Cécile Fauvelot, Lee B. Kats, Jonne Kotta & Fernando M. Pelicice / Aquatic Invasive Species IV
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Florêncio, F.M., Rosado, A., Leal, R.P. et al. Unexpected coexistence of a native and an invasive macrophyte: a functional versus environmental niche perspective. Hydrobiologia (2024). https://doi.org/10.1007/s10750-024-05606-2
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10750-024-05606-2