Abstract
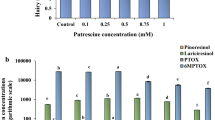
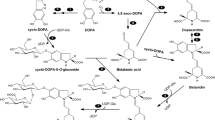
L-3,4-Dihydroxyphenylalanine (L-DOPA) is a compound with strong allelopathic effects on Brassicaceae, Asteraceae, Cucurbitaceae and Hydrophyllaceae species. Although Gramineae are less affected by L-DOPA with respect to their root growth, metabolic routes that protect them from L-DOPA toxicity are poorly understood. We identified a DOPA glucoside in maize (Zea mays L., Gramineae) roots and leaves, but DOPA aglycon was not detectable in maize. Accordingly, when maize seedlings were exposed to L-DOPA solution, DOPA glucoside concentrations increased in maize leaves, suggesting that absorbed L-DOPA is rapidly converted to glucoside conjugate. When DOPA glucoside solution was applied to lettuce seeds (Lactuca sativa; Asteraceae), lettuce radicle growth was less inhibited compared to free L-DOPA. Considering that maize radicle growth is less affected by free L-DOPA, it is likely that maize seedlings protect themselves from toxicity by L-DOPA glucosylation. Interestingly, a developmental stage dependent variation in DOPA glucoside concentration was observed with highest level of metabolite detected in L2 stage maize leaves. As DOPA glucoside also increased in maize during herbivory by the bird cherry-oat aphid (Rhopalosiphum padi L.) and the Graminae generalist armyworm (Mythimna loreyi), as well as in response to treatment with the plant hormones, we propose that DOPA glucoside might be involved in various stress responses and/or defense in maize seedlings.





Similar content being viewed by others
References
Andrews RS, Pridham JB (1965) Structure of a Dopa glucoside from Vicia faba. Nature 205:1213–1214
Brunet PCJ (1963) Tyrosine metabolism in insects. Ann N Y Acad Sci 15:1020–1034
Ciepiela AP, Sempruch C (1999) Effect of L-3,4-dihydroxyphenylalanine, ornithine and γ-aminobutyric acid on winter wheat resistance to grain aphid. J Appl Entomol 123:285–288. https://doi.org/10.1046/j.1439-0418.1999.00356.x
Cohen SA, Michaud DP (1993) Synthesis of a fluorescent derivatizing reagent, 6-aminoquinolyl-N-hydroxysuccinimidyl carbamate, and its application for the analysis of hydrolysate amino acids via high-performance liquid chromatography. Anal Biochem 211:279–287. https://doi.org/10.1006/abio.1993.1270
Fujii Y, Shibuya T, Yasuda T (1991) L-3,4-Dihydroxyphenylalanine as an allelochemical candidate from Mucuna pruriens (L.) DC. var. utilis. Agric Biol Chem 55:617–618. https://doi.org/10.1271/bbb1961.55.617
Hachinohe M, Matsumoto H (2005) Involvement of reactive oxygen species generated from melanin synthesis pathway in phytotoxicity of L-DOPA. J Chem Ecol 31:237–246. https://doi.org/10.1007/s10886-005-1338-9
Hachinohe M, Matsumoto H (2007) Mechanism of Selective phytotoxicity of l-3,4-dihydroxyphenylalanine (L-DOPA) in barnyardglass and lettuce. J Chem Ecol 33:1919–1926. https://doi.org/10.1007/s10886-007-9359-1
Hachinohe M, Sunohara Y, Matsumoto H (2004) Absorption, translocation and metabolism of L-DOPA in barnyardgrass and lettuce: their involvement in species-selective phytotoxic action. Plant Growth Regul 43:237–243. https://doi.org/10.1023/B:GROW.0000045996.72922.1b
Hennion F, Martin-Tanguy J (2000) Amines of the subantarctic crucifer Pringlea antiscorbutica are responsive to temperature conditions. Physiol Plant 109:232–243. https://doi.org/10.1034/j.1399-3054.2000.100303.x
Hopkins TL, Morgan TD, Mueller DD, Tomer KB, Kramer KJ (1995) Identification of catecholamine β-glucosides in the hemolymph of the tobacco hornworm, Manduca sexta (L.), during development. Insect Biochem Mol Biol 25:29–37. https://doi.org/10.1016/0965-1748(94)00043-H
Jiao Y, Chen Y, Ma C, Qin J, Nguyen THN, Liu D, Gan H, Ding S, Luo Z (2018) Phenylalanine as a nitrogen source induces root growth and nitrogen-use efficiency in Populus × canescens. Tree Physiol 38:66–82. https://doi.org/10.1093/treephys/tpx109
Kulma A, Szopa J (2007) Catecholamines are active compounds in plants. Plant Sci 172:433–440. https://doi.org/10.1016/j.plantsci.2006.10.013
Macías FA, Mejías FJR, Molinillo JMG (2019) Recent advances in allelopathy for weed control: from knowledge to applications. Pest Manag Sci 75:2413–2436. https://doi.org/10.1002/ps.5355
Matsumoto H (2011) The mechanisms of phytotoxic action and selectivity of non-protein aromatic amino acids L-DOPA and m-tyrosine. J Pestic Sci 36:1–8. https://doi.org/10.1584/jpestics.R10-15
Nagasawa T, Takagi H, Kawakami K, Suzuki T, Sahashi Y (1961) Studies on the browning compounds of broad bean Vicia faba L. Agr Biol Chem 25:441
Nakajima N, Hiradate S, Fujii Y (1999) Characteristics of growth inhibitory effect of L-3, 4-dihydroxyphenylalanine (L-DOPA) on cucumber seedlings. J Weed Sci Tech (in Japanese) 44:132–138. https://doi.org/10.3719/weed.44.132
Niculaes C, Abramov A, Hannemann L, Frey M (2019) Plant Protection by Benzoxazinoids—Recent Insights into Biosynthesis and Function. Agronomy 8:143. https://doi.org/10.3390/agronomy8080143
Nishihara E, Parvez MM, Araya H, Fujii Y (2004) Germination growth response of different plant species to the allelochemical L-3,4-dihydroxyphenylalanine (L-DOPA). Plant Growth Regul 42:181–189. https://doi.org/10.1023/B:GROW.0000017483.76365.27
Nishihara E, Parvez M, Araya H, Kawashima S, Fujii Y (2005) L-3-(3,4-Dihydroxyphenyl)alanine (L-DOPA), an allelochemical exuded from velvetbean (Mucuna pruriens) roots. Plant Growth Regul 45:113–120. https://doi.org/10.1007/s10725-005-0610-x
Offen D, Panet H, Galili-Mosberg R, Melamed E (2001) Catechol-O-methyltransferase decreases levodopa toxicity in vitro. Clin Neuropharmacol 24:27–30
Rehr SS, Janzen DH, Feeny PP (1973) L-DOPA in legume seeds: a chemical barrier to insect attack. Science 181:81–82
Schulz M, Schütz V, Bigler L, Sicker D, Laschke L (2019) Conversions of benzoxazinoids and downstream metabolites by soil microorganisms. Front Ecol Evol 7:238. https://doi.org/10.3389/fevo.2019.00238
Shitana S, Yazaki K (2020) Dynamism of vacuoles toward survival strategy in plants. Biochim Biophys Acta Biomembr 1862:183127. https://doi.org/10.1016/j.bbamem.2019.183127
Sicker D, Frey M, Schulz M, Gierl A (2000) Role of natural benzoxazinones in the survival strategy of plants. Int Rev Cytol 198:319–346. https://doi.org/10.1016/S0074-7696(00)98008-2
Sivakumar R, Ponrasu T, Divakar S (2009) Syntheses of dopa glycosides using glucosidases. Glycoconj J 26:199–209. https://doi.org/10.1007/s10719-008-9176-y
Swiedrych A, Lorenc-Kukula K, Skirycz A, Szopa J (2004) The catecholamine biosynthesis route in potato is affected by stress. Plant Physiol Biochem 42:593–600. https://doi.org/10.1016/j.plaphy.2004.07.002
Szopa J, Wilczynski G, Fiehn O, Wenczel, a., and Willmitzer, L. (2001) Identification and quantification of catecholamines in potato plants (Solanum tuberosum) by GC–MS. Phytochemistry 58:315–320. https://doi.org/10.1016/S0031-9422(01)00232-1
Tarawali G, Manyong VM, Carsky RJ, Vissoh PV, Osei-Bonsu P, Galiba M (1999) Adoption of improved fallows in West Africa: lessons from mucuna and stylo case studies. Agrofor Syst 47:93–122. https://doi.org/10.1023/A:1006270122255
Wari D, Aboshi T, Shinya T, Galis I (2022) Integrated view of plant metabolic defense with particular focus on chewing herbivores. J Integr Plant Biol 64:449–475. https://doi.org/10.1111/jipb.13204
Acknowledgements
We thank Mr. Hitomi Miyazaki (Zennoh Agricultural Research and Development Center) for providing R. padi. This study was supported by the Joint Research Program of the Institute of Plant Science and Resources at Okayama University, by a Grant-in-Aid for Scientific Research (No. 18K14397 to T.A., No. 21H02196 to I.G.) from the Japan Society for the Promotion of Sciences.
Funding
Japan Society for the Promotion of Science,18K14397,Takako Aboshi, 21H02196, Ivan Galis,Joint Research Program of the Institute of Plant Science and Resources at Okayama University, 3026, Takako Aboshi
Author information
Authors and Affiliations
Contributions
TA, KI and IG contributed to the study’s design. Data collection and analysis were performed by TA and KI with the aid of TS and TM. The first draft of the manuscript was written by TA with the aid of IG. All authors commented on previous versions of the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflicts of interest associated with this manuscript.
Additional information
Communicated by Zhong-Hua Chen.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Aboshi, T., Ittou, K., Galis, I. et al. Glucosylation prevents autotoxicity of stress inducible DOPA in maize seedlings. Plant Growth Regul 101, 159–167 (2023). https://doi.org/10.1007/s10725-023-01009-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10725-023-01009-w