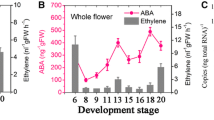
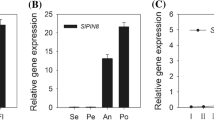
Abstract
The plant hormone abscisic acid (ABA) plays an important role in many aspects of plant growth and development, and a growing number of evidences show that ABA may play an important role in the development of tomato pollen development. To further analyze the role of ABA in the development of tomato pollen, the SlNCED1 gene encoding 9-cis-epoxycarotenoid dioxygenase (NCED), a key enzyme in the ABA biosynthesis, was suppressed in tomato plants by transformation with RNA interference (RNAi). Endogenous ABA content and SlNCED1 transcript levels in the transgenic anther were down-regulated to between 20 and 55% of the levels measured in the wild-type anther. The significant reduction of NCED activity led to a down-regulation in the transcription of specific genes positively related to anther development in tomato, such as TomA92b9, TomA108, SlbHLH080 and 5B-CRP. However, leading to an up-regulation in the transcription of specific genes negatively related to anther development in tomato, such as adh-3a, LAT58, LePro1 and SlPMEI. This resulted in abnormal anther development, which led to the development of a large number of abnormal pollens. In conclusion, ABA affects pollen maturation by regulating the expression of anther-specific genes.









Similar content being viewed by others
References
Alexander MP (1969) Differential staining of aborted and non-aborted Pollen. Biotech Histochem 44:117–122
Antoni R, Gonzalez-Guzman M, Rodriguez L, Rodrigues A, Pizzio GA, Rodriguez PL (2012) Selective inhibition of clade A phosphatases type 2 C by PYR/PYL/RCAR abscisic acid receptors1. Physiol Plant 158:970–980
Barthe P, Garello G, Biancotrinchant J, Pagedegivry MTL (2000) Oxygen availability and ABA metabolism in Fagus sylvatica seeds. Plant Growth Regul 30:185–191
Chandra Sekhar KN, Sawhney VK (1991) Role of ABA in stamen and pistil development in the normal and solanifolia mutant of tomato (Lycopersicon esculentum). Sex Plant Reprod 4:279–283
Chen R, Aguirre PJ, Smith AG (1994) Characterization of an anther- and tapetum-specific gene encoding a glycine-rich protein from tomato. J Plant Physiol 143:651–658
Cheng ZJ, Zhao XY, Shao XX, Wang F, Zhou C, Liu YG, Zhang Y, Zhang XS (2014) Abscisic acid regulates early seed development in Arabidopsis by ABI5-mediated transcription of SHORT HYPOCOTYL UNDER BLUE1. Plant Cell 26:1053–1068
Chernys JT, Zeevaart JAD (2000) Characterization of the 9-cis-epoxycarotenoid dioxygenase gene family and the regulation of abscisic acid biosynthesis in avocado. Plant Physiol 124:343–353
Dai S, Kai W, Liang B, Wang J, Jiang L, Du Y, Sun Y, Leng P (2018) The functional analysis of SlNCED1 in tomato pollen development. Cell Mol Life Sci 75:3457–3472
Dong Y, Guo J (2012) Transcriptional analysis of 9-cis-epoxycarotenoid dioxygenase, glucosyltransferase, 8’-hydroxylase and β-glucosidase genes that regulate abscisic acid homeostasis around the onset of grape berry ripening. J Agric Sci Technol A 7:873–881
Dorffling K (2015) The Discovery of Abscisic Acid: A Retrospect. J Plant Growth Regul 34:795–808
Finkelstein RR, Gampala SS, Rock CD (2002) Abscisic acid signaling in seeds and seedlings. Plant Cell 14:S15–S45
Foster E, Gleddie S, Robert LS (2001) Tapetal gene expression reflects the complex role of the tapetum in pollen development. Recent Res Develop Plant Physiol 2:219–239
Geiger D, Scherzer S, Mumm P, Marten I, Ache P, Matschi S, Liese A, Wellmann C, Al-Rasheid KAS, Grili E, Romeis T, Hedrich R (2010) Guard cell anion channel SLAC1 is regulated by CDPK protein kinases with distinct Ca2+ affinities. Proc Natl Acad Sci USA 107:8023–8028
Hauser F, Waadt R, Schroeder JI (2011) Evolution of abscisic acid synthesis and signaling mechanisms. Curr Biol 21:R346–R355
Ingersoll JC, Rothenberg M, Liedl BE, Folkerts K, Garvin D, Hanson MR, Doyle JJ, Mutschler MA (1994) A novel anther-expressed adh-homologous gene in Lycopersicon esculentum. Plant Mol Biol 26:1875–1891
Jia HF, **e ZQ, Wang C, Shangguan LF, Qian N, Cui MJ, Liu ZJ, Zheng T, Wang MQ, Fang JG (2017) Abscisic acid, sucrose, and auxin coordinately regulate berry ripening process of the Fujiminori grape. Funct Integr Genomics 17:441–457
Kai W, Fu Y, Wang J, Liang B, Li Q, Leng P (2019) Functional analysis of SlNCED1 in pistil development and fruit set in tomato (Solanum lycopersicum L.). Sci Report 9:16943–16955
Kanno Y, Jikumaru Y, Hanada A, Nambara E, Abrams SR, Kamiya Y, Seo M (2010) Comprehensive hormone profiling in develo** Arabidopsis seeds: examination of the site of abscisic acid biosynthesis, abscisic acid transport and hormone interactions. Plant Cell Physiol 51:1988–2001
Kenneth J, McNeil, Alan G, Smith (2005) An anther-specific cysteine-rich protein of tomato localized to the tapetum and microspores. J Plant Physiol 162:457–464
Kim JE, Lee B, Kim S, Lee B, Lee J, Jol S (2013) Genome-wide SNP database for marker-assisted background selection in Tomato. Korean J Breed Sci 45:232–239
Kim WB, Lim CJ, Jang HA, Yi SY, Oh SK, Lee HY, Kim HA, Park YI, Kwon SY (2014) SlPMEI, a pollen-specific gene in tomato. Canadian J Plant Sci 94, 73–83
Koltunow AM, Truettner J, Cox KH, Wallroth M, Goldberg RB (1990) Different temporal and spatial gene expression patterns occur during anther development. Plant Cell 2:1201–1224
Kushiro T, Okamoto M, Nakabayashi K, Yamagishi K, Kitamura S, Asami T, Hirai N, Koshiba T, Kamiya Y, Nambara E (2004) The Arabidopsis cytochrome P450 CYP707A encodes ABA 8-hydroxylases: key enzymes in ABA catabolism. EMBO J 23:1647–1656
Li Q, Ji K, Sun Y, Luo H, Wang H, Leng P (2013) The role of FaBG3 in fruit ripening and B. cinerea fungal infection of strawberry. Plant J 76:24–35
Liao X, Li M, Liu B, Yan M, Yu X, Zi H, Liu H, Yamamuro RC (2018) Interlinked regulatory loops of ABA catabolism and biosynthesis coordinate fruit growth and ripening in woodland strawberry. Proc Natl Acad Sci USA 115:e11542–e11550
Miao Y, Lv D, Wang P, Wang XC, Chen J, Miao C, Song CP (2006) An arabidopsis glutathione peroxidase functions as both a redox transducer and a scavenger in abscisic acid and drought stress responses. Plant Cell 18:2749–2766
Nacken WK0, Huijser P, Beltran JP, Saedler H, Sommer H (1991) Molecular characterization of two stamen-specific genes, tap1 and fil1, that are expressed in the wild type, but not in the deficiens mutant of Antirrhinum majus. Mol Gen Genet 229:129–136
Nambara E, Marion-Poll A (2005) Abscisic acid biosynthesis and catabolism. Annu Rev Plant Biol 56:165–185
Nitsch LMC, Oplaat C, Feron R, Qian M, Wolters-Arts M, Hedden P, Vriezen MWH (2009) Abscisic acid levels in tomato ovaries are regulated by LeNCED1 and SlCYP707A1. Planta 229:1335–1346
Oh HD, Yu DJ, Chung SW, Chea S, Lee HJ (2017) Abscisic acid stimulates anthocyanin accumulation in ‘Jersey’ highbush blueberry fruits during ripening. Food Chem 244:403–407
Polle E, Konzak CF, Kittrick JA (1978) Visual detection of aluminum tolerance levels in wheat by hematoxylin staining of seedling roots. Crop Sci 18:823–827
Priest DM, Ambrose SJ, Vaistij FE, Elias L, Higgins GS (2006) Use of the glucosyltransferase UGT71B6 to disturb abscisic acid homeostasis in Arabidopsis thaliana. Plant J 46:492–502
Qin XQ, Zeevaart JAD (1999) The 9-cis-epoxycarotenoid cleavage reaction is the key regulatory step of abscisic acid biosynthesis in water-stressed bean. Proc Natl Acad Sci USA 96:15354–15361
Sauter A, Wichert K, Hartung W (2000) Extracellular β-glucosidase activity in barley involved in the hydrolysis of ABA glucose conjugate in leaves. J Exp Bot 51:937–944
Sun L, Sun Y, Zhang M, Wang L, Ren J, Cui M, Wang Y, Ji K, Li P, Li Q, Chen P, Dai S, Duan C, Wu Y, Leng P (2012) Suppression of 9-cis-epoxycarotenoid dioxygenase, which encodes a key enzyme in abscisic acid biosynthesis, alters fruit texture in transgenic tomato. Plant Physiol 158:283–298
Sun L, Yuan B, Zhang M, Wang L, Cui M, Wang Q, Leng P (2012) Fruit-specific RNAi-mediated suppression of SlNCED1 increases both lycopene and β-carotene contents in tomato fruit. J Exp Bot 63:3097–3108
Sun Y, Ji K, Liang B, Du Y, Jiang L, Wang J, Kai W, Zhang Y, Zhai X, Chen P, Wang H, Leng P (2017) Suppressing ABA uridine diphosphate glucosyltransferase (SlUGT75C1) alters fruit ripening and the stress response in tomato. Plant J 91:574–589
Trapnell C, Roberts A, Goff LA, Pertea G, Kim D, Kelley DR, Pimentel H, Salzberg SL, Rinn JL, Pachter L (2012) Differential gene and transcript expression analysis of RNA-seq experiments with TopHat and Cufflinks. Nat Protoc 7:562–578
Ursin VM, Yamaguchi J, McCormick S (1989) Gametophytic and sporophytic expression of anther-specific genes in develo** tomato anthers. Plant Cell 1:727–736
Van Bergen S, Kottenhagen MJ, Van D, Meulen RM, Wang M (1999) The role of abscisic acid in induction of androgenesis: a comparative study between Hordeum vulgare L. cvs. Igri and Digger. J Plant Growth Regul 18:135–143
Villalobos-González L, Peña-Neira A, Ibáñez F, Pastenes C (2016) Long-term effects of abscisic acid (ABA) on the grape berry phenylpropanoid pathway: gene expression and metabolite content. Plant Physiol Biochem 105:213–223
Wang X, Yin W, Wu J, Chai LJ, Yi HL (2016) Effects of exogenous abscisic acid on the expression of citrus fruit ripening-related genes and fruit ripening. Sci Hortic 201:175–183
Wilkinson S, Davies WJ (2002) ABA-based chemical signalling: the co-ordination of responses to stress in plants. Plant Cell Environ 25:195–210
Wright SY, Suner MM, Bell PJ, Vaudin M, Greenland AJ (1993) Isolation and characterization of male flower cDNAs from maize. Plant J 3:41–49
**ao HS, Lv LX, Chen ZT (2003) Dynamic changes of endogenous hormone in litchi (Litchi chinensis sonn.) pistil and stamen during flower development. Chin J Appl Environ Biol 9:279–283
Xu SX, Liu GS, Chen RD (2006) Characterization of an anther- and tapetum-specific gene and its highly specific promoter isolated from tomato. Plant Cell Reports 25:231–240
Yu LX, Parthasarathy MV (2014) Molecular and cellular characterization of the tomato pollen profilin, LePro1. Plos One. 9: e 86505
Zhang YS, Li Q, Jiang L, Kai WB, Liang B, Wang J, Du YW, Zhai XW, Wang JL, Zhang YQ, Sun YF, Zhang LS, Leng P (2018) Suppressing type 2 C protein phosphatases alters fruit ripening and the stress response in tomato. Plant Cell Physiol 59:142–154
Zou J, Abrams GD, Barton DL, Taylor DC, Pomeroy MK, Abramset SR (1995) Induction of lipid and oleosin biosynthesis by (+)-Abscisic Acid and its metabolites in microspore-derived embryos of Brassica napus L.cv reston (Biological responses in the presence of 8’-Hydroxyabscisic Acid). Plant Physiol 108:563–571
Acknowledgements
This work was financially supported by grants from the National Natural Science Foundation of China (Grant numbers 31902018, 31801868) and Natural Science Foundation of Shandong Province (Grant numbers ZR2019PC016, ZR2018PC023).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Additional information
Communicated by Guosheng **ong.
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic Supplementary Material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Wang, X., Xu, N., Dong, K. et al. SlNCED1 affects pollen maturation in tomato by regulating the expression of anther-specific genes. Plant Growth Regul 95, 191–205 (2021). https://doi.org/10.1007/s10725-021-00732-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10725-021-00732-6