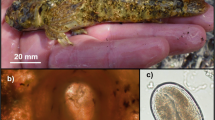
Abstract
Shorthead sculpin (Cottus confusus Bailey and Bond, 1963) in Birch Creek, Idaho, were infected with the trematode Euryhelmis cotti (Simon, 1972). Our objective was to determine if intensity of infection had an influence on growth, reproduction, and mortality of shorthead sculpin at two sites in Birch Creek. Prevalence of trematode infection was 100 % at the upstream site compared to 32 % prevalence at the downstream site; intensity of infection was at least 15 times greater in shorthead sculpin at the upstream site. von Bertalanffy growth models differed significantly between sites; the growth coefficient (K) was higher for sculpin at the upstream site. Gonadosomatic index for both males and females was significantly higher for sculpin found upstream. Females at the upstream site also had significantly more eggs per clutch compared to females at the downstream site. Annual mortality was significantly higher upstream (A = 57 %; 95 % CI = 52–62 %) compared to downstream (A = 42 %; 95 % CI = 38–46 %). Consequently, the density of sculpin was lower at the upstream site (13 fish/100 m2) compared to the downstream site (54 fish/100 m2). We did not find evidence to suggest that growth and reproduction of shorthead sculpin were influenced by infection of E. cotti, but rather were influenced primarily by density dependent factors. Mortality was higher for shorthead sculpin at the upstream site with high prevalence and intensity of infection. However, we do not know if higher mortality was a direct consequence of trematode infection or merely correlated with trematode infection.







Similar content being viewed by others
References
Ameel DJ (1938) The morphology and life cycle of Euryhelmis monorchis n. sp. (Trematoda) from the mink. J Parasitol 24:219. https://doi.org/10.2307/3272291
Amundsen PA, Knudsen R, Klemetsen A (2007) Intraspecific competition and density dependence of food consumption and growth in arctic charr. J Anim Ecol 76:149–158. https://doi.org/10.1111/j.1365-2656.2006.01179.x
Anderson CS (1985) The structure of sculpin populations along a stream size gradient. Environ Biol of Fishes 13(2):93–102. https://doi.org/10.1007/BF00002577
Andrews DA, Minshall GW (1979) Distribution of benthic invertebrates in the lost streams of Idaho. Am Midl Nat 140–148. https://www.jstor.org/stable/2425075
Arranz I, Mehner T, Benejam L, Argillier C, Holmgren K, Jeppesen E, Volta P, Winfield IJ, Brucet S (2016) Density-dependent effects as key drivers of intraspecific size structure of six abundant fish species in lakes across Europe. Can J Fish Aquat Sci 73:519–534. https://doi.org/10.1139/cjfas-2014-0508
Blanar CA, Hewitt M, McMaster M, Kirk J, Wang Z, Norwood W, Marcogliese DJ (2016) Parasite community similarity in Athabasca River trout-perch (Percopsis omiscomaycus) varies with local-scale land use and sediment hydrocarbons, but not distance or linear gradients. Parasitol Res 115(10):3853–3866. https://doi.org/10.1007/s00436-016-5151-x
Blasco-Costa I, Poulin R (2017) Parasite life-cycle studies: a plea to resurrect an old parasitological tradition. J Helminthol 91(6):647–656. https://doi.org/10.1017/S0022149X16000924
Blasco-Costa I, Koehler AV, Martin A, Poulin R (2013) Upstream-downstream gradient in infection levels by fish parasites: a common river pattern? Parasitol 140(2):266. https://doi.org/10.1017/50031182012001527
Bond MJ, Jones NE, Haxton TJ (2016) Growth and life history patterns of a small-bodied stream fish, Cottus cognatus, in hydropeaking and natural rivers of northern Ontario. River Res Appl 32(4):721–733. https://doi.org/10.1002/rra.2886
Chapman DG, Robson DS (1960) The analysis of a catch curve. Biometrics 16:354–368. https://doi.org/10.2307/2527687
Díaz-Rodríguez J, Donaire-Barroso D, Jowers MJ (2018) First report of Euryhelmis parasites (Trematoda, Heterophyidae) in Africa: conservation implications for endemic amphibians. Parasitol Res 117:2569–2576. https://doi.org/10.1007/s00436-018-5946-z
Dobson AP, Hudson PJ (1986) Parasites, disease and the structure of ecological communities. Trends Ecol Evol 1:11–15. https://doi.org/10.1016/0169-5347(86)90060-1
Faust EC, Nishigori M (1926) The life cycles of two new species of Heterophyidae, parasitic in mammals and birds. J Parasitol 13:91–128. https://doi.org/10.2307/3271705
Gebhard AE, Perkin JS (2017) Assessing riverscape-scale variation in fish life history using banded sculpin (Cottus carolinae). Environ Biol of Fish 100(11):1397–1410. https://doi.org/10.1007/s10641-017-0651-9
Grant JWA, Imre I (2005) Patterns of density-dependent growth in juvenile stream-dwelling salmonids. J Fish Biol 67:100–110. https://doi.org/10.1111/j.0022-1112.2005.00916.x
Jacobson KC, Teel D, Van Doornik DM, Casillas E (2008) Parasite-associated mortality of juvenile Pacific salmon caused by the trematode Nanophyetus salmincola during early marine residence. Mar Ecol Prog Ser 354:235–244. https://doi.org/10.3354/meps07232
Johnson PT, Lunde KB, Ritchie EG, Launer AE (1999) The effect of trematode infection on amphibian limb development and survivorship. Science 284:802–804. https://doi.org/10.1126/science.284.5415.802
Johnson PT, Lunde KB, Thurman EM et al (2002) Parasite (Ribeiroia ondatrae) infection linked to amphibian malformations in the western United States. Ecol Monogr 72:151–168. https://doi.org/10.1890/0012-9615(2002)072[0151:PROILT]2.0.CO;2
Johnson PT, Ostfeld RS, Keesing F (2015) Frontiers in research on biodiversity and disease. Ecol Lett 18:1119–1133. https://doi.org/10.1111/ele.12479
Koprivnikar J, Johnson PT (2016) The rise of disease ecology and its implications for parasitology—a review. J Parasitol 102:397–409. https://doi.org/10.1645/15-942
Lafferty KD (2008) Ecosystem consequences of fish parasites. J Fish Biol 73:2083–2093. https://doi.org/10.1111/j.1095-8649.2008.02059.x
Lafferty KD, Morris AK (1996) Altered behavior of parasitized killifish increases susceptibility to predation by bird final hosts. Ecology 77:1390–1397. https://doi.org/10.2307/2265536
Lemly AD, Esch GW (1984) Effects of the trematode Uvulifer ambloplitis on juvenile bluegill sunfish, Lepomis macrochirus: ecological implications. J Parasitol 70:475. https://doi.org/10.2307/3281395
Lester RJG (1984) A review of methods for estimating mortality due to parasites in wild fish populations. Helgolander Meeresun 37:57–64. https://doi.org/10.1007/BF01989295
Lorenzen K, Enberg K (2002) Density-dependent growth as a key mechanism in the regulation of fish populations: evidence from among-population comparisons. Proc R Soc B-Biol Sci 269:49–54. https://doi.org/10.1098/rspb.2001.1853
Marcogliese DJ (2004) Parasites: small players with crucial roles in the ecological theater. EcoHealth 1:151–164. https://doi.org/10.1007/s10393-004-0028-3
Marcogliese DJ (2005) Parasites of the superorganism: are they indicators of ecosystem health? Int J Parasitol 35:705–716. https://doi.org/10.1016/j.ijpara.2005.01.015
Markle DF, Terwilliger MR, Simon DC (2014) Estimates of daily mortality from a neascus trematode in age-0 shortnose sucker (Chasmistes brevirostris) and the potential impact of avian predation. Environ Biol Fish 97:197–207. https://doi.org/10.1007/s10641-013-0141-7
Markle DF, Janik A, Peterson JT, Choudhury A, Simon DC, Tkach VV, Terwilliger MR, Sanders JL, Kent ML (2020) Odds ratios and hurdle models: a long-term analysis of parasite infection patterns in endangered young-of-the-year suckers from Upper Klamath Lake, Oregon, USA. Int J Parasitol 50:315–330. https://doi.org/10.1016/j.ijpara.2020.02.001
McCurdy D, Forbes M, Boates J (1999) Testing alternative hypotheses for variation in amphipod behaviour and life history in relation to parasitism. Int J Parasitol 29:1001–1009. https://doi.org/10.1016/s0020-7519(99)00067-3
Merrill TES, Johnson PT (2020) Towards a mechanistic understanding of competence: a missing link in diversity–disease research. Parasitol 147:1159–1170. https://doi.org/10.1017/S0031182020000943
Minchella DJ, Scott ME (1991) Parasitism: a cryptic determinant of animal community structure. Trends Ecol Evol 6:250–254. https://doi.org/10.1016/0169-5347(91)90071-5
Mitchell AJ, Smith C, Hoffman G (1982) Pathogenicity and histopathology of an unusually intense infection of white grubs (Posthodiplostomum m. minimum) in the fathead minnow (Pimephales promelas). J Wildl Dis 18:51–57. https://doi.org/10.7589/0090-3558-18.1.51
Mitchell AJ, Overstreet RM, Goodwin AE, Brandt TM (2005) Spread of an exotic fish-gill trematode. Fish 30:11–16. https://doi.org/10.1577/1548-8446(2005)30[11:soaeft]2.0.co;2
Myers RA, Bowen KG, Barrowman NJ (1999) Maximum reproductive rate of fish at low population sizes. Can J Fish Aquat Sci 56:2404–2419. https://doi.org/10.1139/f99-201
Ogle DH (2016) Introductory fisheries analyses with R. CRC Press, Florida
Ondračková M, Šimková A, Gelnar M, Jurajda P (2004) Posthodiplostomum cuticola (Digenea: Diplostomatidae) in intermediate fish hosts: factors contributing to the parasite infection and prey selection by the definitive bird host. Parasitol 129:761–770. https://doi.org/10.1017/s0031182004006456
Ondračková M, Dávidová M, Pečínková M et al (2005) Metazoan parasites of Neogobius fishes in the Slovak section of the River Danube. J Appl Ichthyol 21:345–349. https://doi.org/10.1111/j.1439-0426.2005.00682.x
Ortega C, Fajardo R, Enríquez R (2009) Trematode Centrocestus formosanus infection and distribution in ornamental fishes in Mexico. J Aquat Anim Health 21:18–22. https://doi.org/10.1577/h07-022.1
Quist MC, Pegg MA, DeVries DR (2012) Age and growth. In: Zale AV, Parrish DL, Sutton TM (eds) Fisheries techniques, 3rd edn. American Fisheries Society, Bethesda, pp 677–731
R Development Core Team (2019) R: A language and environment for statistical computing. R Foundation for Statistical Computing, Vienna. http://www.R-project.org
Ricker WE (1975) Computation and interpretation of biological statistics of fish populations. Technical Report Bulletin 191, Bulletin of the Fisheries Research Board of Canada
Sandland GJ, Goater CP (2001) Parasite-induced variation in host morphology: brain-encysting trematodes in fathead minnows. J Parasitol 87:267. https://doi.org/10.2307/3285040
Sato H, Ihara S, Inaba O, Une Y (2010) Identification of Euryhelmis costaricensis metacercariae in the skin of Tohoku hynobiid salamanders (Hynobius lichenatus), northeastern Honshu, Japan. J Wildl Dis 46:832–842. https://doi.org/10.7589/0090-3558-46.3.832
Simon MJ (1972) Euryhelmis cotti N. Sp. (Trematoda: Heterophyidae) with observations on its life cycle. Dissertation and Theses, Paper 960, Portland State University
Smith MW, Then AY, Wor C, Ralph G, Polluck KH, Hoenig JM (2012) Recommendations for catch-curve analysis. N Am J Fish Manag 32:956–967
Taylor DW (1985) Evolution of freshwater drainages and mollusks in western North America. In: Smiley CJ (ed) Late Cenozoic history of the Pacific Northwest. Pacific Division of the American Association for the Advancement of Science, San Francisco
Tobler M, Schlupp I (2006) Influence of black spot disease on shoaling behaviour in female western mosquitofish, Gambusia affinis (Poeciliidae, Teleostei). Environ Biol Fish 81:29–34. https://doi.org/10.1007/s10641-006-9153-
Tobler M, Schlupp I (2008) Influence of black spot disease on shoaling behaviour in female western mosquitofish, Gambusia affinis (Poeciliidae, Teleostei). Environ Biol Fish 81:29–34. https://doi.org/10.1007/s10641-006-9153-x
Wood CL, Johnson PT (2015) A world without parasites: exploring the hidden ecology of infection. Front Ecol Environ 13:425–434. https://doi.org/10.1890/140368
Wootton D (1957) The life history of Cryptocotyle concavum (Creplin, 1825) Fischoeder, 1903 (Trematoda: Heterophyidae). J Parasitol 43:271–279
Zippin C (1958) The removal method of population estimation. J Wildl Manag 22:82–90
Acknowledgements
We are grateful to Jason Spillett, Gabe Schultz, Darcy McCarrick, and Renata Santos for help with field sampling. Idaho Department of Fish and Game provided use of the low-speed saw. Brigham Young University-Idaho provided funding for the project. All fish were handled in full compliance with Brigham Young University-Idaho Institutional Animal Care and Use Committee protocols. Samples were collected in compliance with an Idaho Department of Fish and Game scientific collection permit (F-14-67-17) issued to EJB.
Funding
Funding was provided by Brigham Young University-Idaho.
Author information
Authors and Affiliations
Contributions
All authors have contributed to this manuscript sufficiently to warrant authorship.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they do not have any conflicts of interest.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Byington, D., Flinders, J. & Billman, E. Effect of a trematode infection on growth, reproduction, and mortality of shorthead sculpin. Environ Biol Fish 104, 265–276 (2021). https://doi.org/10.1007/s10641-021-01072-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10641-021-01072-0