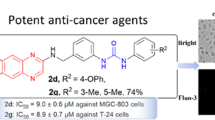
5′-Nitro- (2) and 5′-aminodubamine (3) were sequentially synthesized from the alkaloid dubamine. Condensation of 3 with 15 aldehydes gave imines, reduction of which produced secondary amines. Studies of the cytotoxicities for HeLa and HEp-2 cells of the compounds at a concentration of 100 μM revealed several trends. The activity decreased significantly or disappeared at lower concentrations although compound 5o at concentrations of 1–10 μM was active for HEp-2 cells at the level of cisplatin.
Similar content being viewed by others
References
A. P. G. Macabeo and A. M. Aguinaldo, Pharmacogn. Rev., 2, 317 (2008).
V. Nadaraj and S. T. Selvi, J. Chem. Pharm. Res., 4, 2850 (2012).
R. V. Solomon and H. Lee, Curr. Med. Chem., 18, 1508 (2011).
B. Pati and S. Banerjee, J. PharmaSciTech, 3, 59-67 (2014).
T. A. K. Prescott, I. H. Sadler, R. Kiapranis, and S. K. Maciver, J. Ethnopharmacol., 109, 289 (2007).
J. Koyama, I. Toyokuni, and K. Tagahara, Chem. Pharm. Bull., 47, 1038 (1999).
J. Yoon and Ch. Cheon, Asian J. Org. Chem., 8, 7 (2019).
M. Martinez-Grueiro, C. Gimenez-Pardo, A. Gomez-Barrio, X. Franck, A. Fournet, R. Hocquemiller, B. Figadere, and N. Casado-Escribano, Farmaco, 60, 219 (2005).
K. Gopaul, S. A. Shintre, and N. A. Koorbanally, Anti-Cancer Agents Med. Chem., 15, 631 (2015).
O. Afzal, S. Kumar, M. R. Haider, M. R. Ali, R. Kumar, M. Jaggi, and S. Bawa, Eur. J. Med. Chem., 97, 871 (2015).
S. Yu. Yunusov, Alkaloids [in Russian], FAN UzSSR, Tashkent, 1981.
A. A. Szewczyk, K. Glowniak, and T. Baj, Curr. Issues Pharm. Med. Sci., 29, 33 (2016).
N. Hassanloe, B. Zeynizadeh, and Sh. Ashuri, Org. Chem.: Indian J., 12, 6 (2016).
B. Zh. Elmuradov, A. Sh. Abdurazakov, and Kh. M. Shakhidoyatov, Chem. Nat. Compd., 46, 262 (2010).
A. Vass, J. Dudas, J. Toth, and R. S. Varma, Tetrahedron Lett., 42, 5347 (2001).
I. P. Tsypysheva, A. V. Koval’skaya, and A. N. Lobov, Chem. Nat. Compd., 49, 902 (2013).
Kh. M. Shakhidoyatov, N. I. Mukarramov, and F. R. Utaeva, Chem. Nat. Compd., 44, 625 (2008).
M. Niks and M. Otto, J. Immunol. Methods, 130, 149 (1990).
Acknowledgment
The work was financially supported by the Ministry of Innovative Development, Cabinet of Ministers, Republic of Uzbekistan (Grant MRU-FA-21/2017).
Author information
Authors and Affiliations
Corresponding author
Additional information
Translated from Khimiya Prirodnykh Soedinenii, No. 3, May–June, 2020, pp. 439–444.
Rights and permissions
About this article
Cite this article
Niyazmetov, A.R., Terent’eva, E.O., Khamidova, U.B. et al. Synthesis of Derivatives of the 2-Arylquinoline Alkaloid Dubamine and their Cytotoxicity. Chem Nat Compd 56, 511–517 (2020). https://doi.org/10.1007/s10600-020-03074-3
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10600-020-03074-3