Abstract
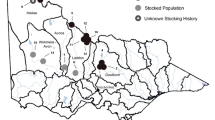
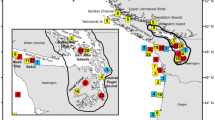
Glacial cycles and pre-glacial drainage patterns have imbued the Interior Highlands of the United States with a rich suite of freshwater taxa and phylogroups. However, supplementation of sportfish from the Great Lakes into the waterbodies of this region, including the Missouri, Ouachita, Black, White, and Little Red River drainages, may have obscured phylogeographic patterns. Walleye (Sander vitreus), one of the most common sportfish in the eastern US, inhabit the Interior Highlands but their population genetic composition and structure in this region has received little attention. We examined the genetic composition of walleye (n = 643) in the Interior Highlands using microsatellite markers and a subsample (n = 188) with mitochondrial DNA and found significant genetic differences among walleye in our study area. Walleye from the Missouri, Ouachita, White, and Little Red drainages were most closely related to a Great Lakes reference sample, a common stocking source. However, the Black River, in the easternmost portion of the Interior Highlands, contained walleye with mitochondrial DNA that was closely related to walleye from the Eastern Highlands. The remainder of the study area drainages contained a mix of walleye groups, more closely related to Great Lakes rather than Highlands walleye but not definitively the product of stocking. Though managers have relied on mitochondrial markers for stock identification in the past, we recommend that walleye in regions receiving little research attention be analyzed with nuclear markers to better understand and preserve genetic diversity and that managers stock with local walleye only within the drainages that we identified as genetically distinct: Black, Missouri, and White/Little Red/Ouachita.




Similar content being viewed by others
References
Allen BE, Bowles E, Morris MR, Rogers SM (2018) Loss of SNP genetic diversity following population collapse in a recreational walleye (Sander vitreus) fishery. Can J of Fish Aquat Sci 75:1644–1651. https://doi.org/10.1139/cjfas-2017-0164
Arkansas Game and Fish Commission (2017) Walleye, Sauger and Saugeye Management Plan.
April J, Hanner RH, Dion-Côté AM, Bernatchez L (2013) Glacial cycles as an allopatric speciation pump in north-eastern American freshwater fishes. Mol Ecol 22:409–422. https://doi.org/10.1111/mec.12116
Barila, TY (1980) Stizostedion vitreum (Mitchill), walleye. In: Lee, DS et al (eds) Atlas of North American freshwater fishes, North Carolina State Museum of Natural History, Raleigh, pp 747–748
Berendzen PB, Simons AM, Wood RM (2003) Phylogeography of the northern hogsucker, Hypentelium nigricans (Teleostei: Cypriniformes): genetic evidence for the existence of the ancient Teays River. J Biogeogr 30:1139–1152. https://doi.org/10.1046/j.1365-2699.2003.00888.x
Berendzen PB, Simons AM, Wood RM, Dowling TE, Secor CL (2008a) Recovering cryptic diversity and ancient drainage patterns in eastern North America: historical biogeography of the Notropis rubellus species group (Teleostei: Cypriniformes). Mol Phylogenetics Evol 46:721–737. https://doi.org/10.1016/j.ympev.2007.07.008
Berendzen PB, Gamble T, Simons AM (2008b) Phylogeography of the bigeye chub Hybopsis amblops (Teleostei: Cypriniformes): early Pleistocene diversification and post-glacial range expansion. J Fish Biol 73:2021–2039. https://doi.org/10.1111/j.1095-8649.2008.02046.x
Bermingham E, Avise JC (1986) Molecular zoogeography of freshwater fishes in the southeastern United States. Genet 113:939–965. https://doi.org/10.1093/genetics/113.4.939
Bernatchez L, Danzmann RG (1993) Congruence in control-region sequence and restriction-site variation in mitochondrial DNA of brook charr (Salvelinus fontinalis Mitchill). Mol Biol Evol 10:1002–1114
Bernatchez L, Wilson CC (1998) Comparative phylogeography of Nearctic and Palearctic fishes. Mol Ecol 7:431–452. https://doi.org/10.1046/j.1365-294x.1998.00319.x
Billington N (1996). Geographical distribution of mitochondrial DNA (mtDNA) variation in walleye, sauger, and yellow perch. In Annales Zoologici Fennici (pp. 699–706). Finnish Zoological and Botanical Publishing Board. https://www.jstor.org/stable/23736117
Billington N, Barrette RJ, Hebert PD (1992) Management implications of mitochondrial DNA variation in walleye stocks. N Am J Fish Manag 12:276–284. https://doi.org/10.1577/1548-8675(1992)012%3C0276:MIOMDV%3E2.3.CO;2
Billington N, Hebert PD (1988) Mitochondrial DNA variation in Great Lakes walleye (Stizostedion vitreum) populations. Can J of Fish Aquat Sci 45:643–654. https://doi.org/10.1139/f88-078
Billington N, Hebert PD (1991) Mitochondrial DNA diversity in fishes and its implications for introductions. Can J of Fish Aquat Sci 48:80–94. https://doi.org/10.1139/f91-306
Billington N, Sloss BL (1998) Mitochondrial DNA and allozyme analysis of walleyes from the Rockcastle River and the Cumberland River (Big South Fork). Kentucky. Technical Report, Kentucky Department of Fish and Wildlife Resources.
Billington N, Strange RM (1995) Mitochondrial DNA analysis confirms the existence of a genetically divergent walleye population in northeastern Mississippi. Trans Am Fish Soc 124:770–776. https://doi.org/10.1577/1548-8659(1995)124%3C0770:NMDACT%3E2.3.CO;2
Black, JD 1940. The distribution of the fishes of Arkansas. Dissertation, University of Michigan, Ann Arbor, Michigan
Borden WC, Krebs RA (2009) Phylogeography and postglacial dispersal of smallmouth bass (Micropterus dolomieu) into the Great Lakes. Can J of Fish Aquat Sci 66:2142–2156. https://doi.org/10.1139/F09-155
Borer SO, Miller LM, Kapuscinski AR (1999) Microsatellites in walleye Stizostedion vitreum. Mol Ecol 8:336–338
Chapuis MP, Estoup A (2007) Microsatellite null alleles and estimation of population differentiation. Mol Biol Evol 24:621–631. https://doi.org/10.1093/molbev/msl191
Cornforth A, Russell B, Settle R (2010) Black River Strain Walleye Final Report. Report to the Missouri Department of Conservation, Jefferson City, MO.
DiStefano RJ, Hiebert JF (2000) Distribution and movement of walleye in reservoir tailwaters during spawning season. J Freshw Ecol 15:145–155. https://doi.org/10.1080/02705060.2000.9663732
Duvernell DD, Westhafer E, Schaefer JF (2019) Late Pleistocene range expansion of North American topminnows accompanied by admixture and introgression. J Biogeogr 46:2126–2140. https://doi.org/10.1111/jbi.13597
Eldridge WH, Bacigalupi MD, Adelman IR, Miller LM, Kapuscinski AR (2002) Determination of relative survival of two stocked walleye populations and resident natural-origin fish by microsatellite DNA parentage assignment. Can J of Fish Aquat Sci 59:282–290. https://doi.org/10.1139/f02-007
Evanno G, Regnaut S, Goudet J (2005) Detecting the number of clusters of individuals using the software STRUCTURE: a simulation study. Mol Ecol 14:2611–2620. https://doi.org/10.1111/j.1365-294X.2005.02553.x
Excoffier L, Lischer HEL (2010) Arlequin suite ver 3.5: A new series of programs to perform population genetics analyses under Linux and Windows. Mol Ecol Resour 10:564–567. https://doi.org/10.1111/j.1755-0998.2010.02847.x
Falush D, Stephens M, Pritchard JK (2003) Inference of population structure using multilocus genotype data: linked loci and correlated allele frequencies. Genet 164:1567–1587. https://doi.org/10.1093/genetics/164.4.1567
Gatt MH, Ferguson MM, Liskauskas AP (2000) Comparison of control region sequencing and fragment RFLP analysis for resolving mitochondrial DNA variation and phylogenetic relationships among Great Lakes walleyes. Trans Am Fish Soc 129:1288–1299. https://doi.org/10.1577/1548-8659(2000)129%3C1288:COCRSA%3E2.0.CO;2
Gatt MH, Fraser DJ, Liskauskas AP, Ferguson MM (2002) Mitochondrial DNA variation and stock structure of walleyes from eastern Lake Huron: an analysis of contemporary and historical samples. Trans Am Fish Soc 131:99–108. https://doi.org/10.1577/1548-8659(2002)131%3C0099:MDVASS%3E2.0.CO;2
George AL, Kuhajda BR, Williams JD, Cantrell MA, Rakes PL, Shute JR (2009) Guidelines for propagation and translocation for freshwater fish conservation. Fisheries 34:529–545. https://doi.org/10.1577/1548-8446-34.11.529
Gunn JC, Berkman LK, Koppelman J, Taylor AT, Brewer S, Long JM, Eggert LS (2020) Complex patterns of genetic and morphological differentiation in the Smallmouth Bass subspecies (Micropterus dolomieu dolomieu and M. d. velox) of the Central Interior Highlands. Conserv Genet 21:891–904. https://doi.org/10.1007/s10592-020-01295-1
Gunn JC, Berkman LK, Koppelman J, Taylor AT, Brewer SK, Long JM, Eggert LS (2022) Genomic divergence, local adaptation, and complex demographic history may inform management of a popular sportfish species complex. Ecol Evol 12 e9370. https://doi.org/10.1002/ece3.9370
Hall T (1999) BioEdit: a user-friendly biological sequence alignment editor and analysis program for Windows 95/98/NT. Nucleic Acids Symp Ser 41:95–98
Hammen JJ, Sloss BL (2019) Walleye genetic characterization in the northern Ceded Territory of Wisconsin: Implications for stocking using conservation strategies. N Am J Fish Manag 39:693–704. https://doi.org/10.1002/nafm.10302
Haponski AE, Stepien CA (2013) Phylogenetic and biogeographical relationships of the Sander pikeperches (Percidae: Perciformes): patterns across North America and Eurasia. Biol J Linn Soc 110:156–179. https://doi.org/10.1111/bij.12114
Haponski AE, Stepien CA (2014a) A population genetic window into the past and future of the walleye Sander vitreus: relation to historic walleye and the extinct “blue pike” S. v.“glaucus”. BMC Evol Biol 14:133. https://doi.org/10.1186/1471-2148-14-133
Haponski AE, Stepien CA (2014b) Genetic connectivity and diversity of walleye (Sander vitreus) spawning groups in the Huron-Erie Corridor. J Gt Lakes Res 40:89–100. https://doi.org/10.1016/j.jglr.2012.12.006
Henry SD, Barkley SW, Koppelman JB, Johnson RL (2008) Assessment of stocking success of walleye in the Eleven Point River, Arkansas. N Am J Fish Manag 28:1498–1505. https://doi.org/10.1577/M07-111.1
Hoagstrom CW, Berry CR Jr (2010) The native range of walleyes in the Missouri River drainage. N Am J Fish Manag 30:642–654. https://doi.org/10.1577/M09-081.1
Jakobsson M, Rosenberg NA (2007) CLUMPP: a cluster matching and permutation program for dealing with label switching and multimodality in analysis of population structure. Bioinform 23:1801–1806. https://doi.org/10.1093/bioinformatics/btm233
Janes JK, Miller JM, Dupuis JR, Malenfant RM, Gorrell JC, Cullingham CI, Andrew RL (2017) The K=2 conundrum. Mol Ecol 26:3594–3602. https://doi.org/10.1111/mec.14187
Jones OR, Wang J (2010) COLONY: a program for parentage and sibship inference from multilocus genotype data. Mol Ecol Resour 10:551–555. https://doi.org/10.1111/j.1755-0998.2009.02787.x
Kalinowski ST (2005) HP-RARE 1.0: a computer program for performing rarefaction on measures of allelic richness. Mol Ecol Notes 5:187–189. https://doi.org/10.1111/j.1471-8286.2004.00845.x
Li YL, Liu JX (2018) StructureSelector: A web-based software to select and visualize the optimal number of clusters using multiple methods. Mol Ecol Resour 18:176–177. https://doi.org/10.1111/1755-0998.12719
Mandrak NE, Crossman EJ (1992) Postglacial dispersal of freshwater fishes into Ontario. Can J Zool 70:2247–2259. https://doi.org/10.1139/z92-302
Mayden RL (1988) Vicariance biogeography, parsimony, and evolution in North American freshwater fishes. Syst Biol 37:329–355. https://doi.org/10.1093/sysbio/37.4.329
Missouri Department of Conservation (2017) Missouri’s Walleye Management Plan 2017–2026.
Near TJ, Page LM, Mayden RL (2001) Intraspecific phylogeography of Percina evides (Percidae: Etheostomatinae): an additional test of the Central Highlands pre-Pleistocene vicariance hypothesis. Mol Ecol 10:2235–2240. https://doi.org/10.1046/j.1365-294X.2001.01362.x
Palmer GC, Culver C, Dutton D, Murphy BR, Hallerman EM, Billington N, Williams J (2006) Genetic distinct walleye stocks in Claytor Lake and the upper New River, Virginia. Proc Annu Conf Southeast Assoc Fish Wildl Agencies 60:125–131
Peakall R, Smouse PE (2006) GENALEX 6: genetic analysis in Excel. Population genetic software for teaching and research. Mol Ecol Notes 6:288–295. https://doi.org/10.1111/j.1471-8286.2005.01155.x
Peakall R, Smouse PE (2012) GENALEX 6.5: genetic analysis in Excel. Population genetic software for teaching and research-an update. Bioinform 28:2537–2539. https://doi.org/10.1111/j.1471-8286.2005.01155.x
Puechmaille SJ (2016) The program structure does not reliably recover the correct population structure when sampling is uneven: subsampling and new estimators alleviate the problem. Mol Ecol Resour 16:608–627. https://doi.org/10.1111/1755-0998.12512
Pritchard JK, Stephens M, Donnelly P (2000) Inference of population structure using multilocus genotype data. Genet 155:945–959. https://doi.org/10.1093/genetics/155.2.945
Raymond M, Rousset F (1995) An exact test for population differentiation. Evol 49:1280–1283. https://doi.org/10.2307/2410454
Robison HW, Buchanan TM (2020) Fishes of Arkansas. University of Arkansas Press.
Rodriguez-Ramilo ST, Wang J (2012) The effect of close relatives on unsupervised Bayesian clustering algorithms in population genetic structure analysis. Mol Ecol Resour 12:873–884. https://doi.org/10.1111/j.1755-0998.2012.03156.x
Ronquist F, Teslenko M, Van Der Mark P et al (2012) MRBAYES 3.2: efficient Bayesian phylogenetic inference and model choice across a large model space. Syst Biol 61:539–542. https://doi.org/10.1093/sysbio/sys029
Rousset F (2008) GENEPOP’007: a complete re-implementation of the genepop software for Windows and Linux. Mol Ecol Resour 8:103–106. https://doi.org/10.1111/j.1471-8286.2007.01931.x
Rozas J, Ferrer-Mata A, Sánchez-DelBarrio JC, Guirao-Rico S, Librado P, Ramos-Onsins SE, Sánchez-Gracia A (2017) DnaSP 6: DNA sequence polymorphism analysis of large data sets. Mol Biol Evol 34:3299–3302
Stegman CE (2013) Distribution and History of Walleye (Sander vitreus) in the North American Central Highlands. Ohio University, Thesis
Stepien CA, Faber JE (1998) Population genetic structure, phylogeography and spawning philopatry in walleye (Stizostedion vitreum) from mitochondrial DNA control region sequences. Mol Ecol 7:1757–1769. https://doi.org/10.1046/j.1365-294x.1998.00512.x
Stepien CA, Murphy DJ, Lohner RN, Sepulveda-Villet OJ, Haponski AE (2009) Signatures of vicariance, postglacial dispersal and spawning philopatry: population genetics of the walleye Sander vitreus. Mol Ecol 18:3411–3428. https://doi.org/10.1111/j.1365-294X.2009.04291.x
Strange RM, Burr BM (1997) Intraspecific phylogeography of North American highland fishes: a test of the Pleistocene vicariance hypothesis. Evol 51:885–897. https://doi.org/10.1111/j.1558-5646.1997.tb03670.x
Wahlund S (1928) Zusammensetzung von Populationen und Korrelationerscheinungen vom Standpunkt der Vererbungslehre aus betrachtet. Hereditas 11:65–106
Waples RS, Anderson EC (2017) Purging putative siblings from population genetic data sets: a cautionary view. Mol Ecol 26:1211–1224. https://doi.org/10.1111/mec.14022
Ward RD, Billington N, Hebert PD (1989) Comparison of allozyme and mitochondrial DNA variation in populations of walleye, Stizostedion vitreum. Can J of Fish Aquat Sci 46:2074–2084. https://doi.org/10.1139/f89-257
Weir BS (1996) Genetic Data Analysis II: Methods for Discrete Population Genetic Data.
Sinauer Assoc., Inc., Sunderland, MA, USA.
Welsh A, Hill T, Quinlan H, Robinson C, May B (2008) Genetic assessment of lake sturgeon population structure in the Laurentian Great Lakes. N Am J Fish Manag 28:572–591. https://doi.org/10.1577/M06-184.1
White MM, Faber JE, Zipfel KJ (2012) Genetic identity of walleye in the Cumberland River. Am Midl Nat 167:373–383. https://doi.org/10.1674/0003-0031-167.2.373
White MM, Kassler TW, Philipp DP, Schell SA (2005) A genetic assessment of Ohio River walleyes. Trans Am Fish Soc 134:661–675. https://doi.org/10.1577/T03-218.1
Wiley EO, Mayden RL (1985) Species and speciation in phylogenetic systematics, with examples from the North American fish fauna. Ann Mo Bot Gard 72:596–635. https://doi.org/10.2307/2399217
Acknowledgements
We thank the many field staff who contributed samples and provided research support for this study including Kelly Winningham, Matt Schroeder, Nathan Recktenwald, Chris Brooke, Jason Persinger, Christina Kelsay, Dave Woods, Shane Bush, Blake Stephens, Mary Scott, Allison Asher, Brent Timmons, Jeremy Risley, Katrina Knott, and Jon Stein. Michael Sandel generously provided Lake Erie and North Dakota walleye samples. We also thank Jonathan Orech, Jessica Brooks, Hilary K. Canada, and Michelle Anderson for support in the lab. Joe Gunn, Eric Hallerman, Robert Hrabik, and two anonymous reviewers provided helpful comments on the manuscript. We are grateful for administrative project support from John Ackerson, Andy Cornforth, David Duvernell, Dave Herzog, Chris Kennedy, Tom Kulowiec, A. J. Pratt, Tony Spicci, and Kasey Whiteman. Supplemental funding was provided by the Arkansas State University Department of Biological Sciences. We thank the Arkansas Center for Biodiversity Collections for cataloging a subset of voucher specimens of the Black River Walleye used in this study into the Arkansas State University Museum of Zoology (catalog numbers ASUMZ 20281-20287).
Author information
Authors and Affiliations
Contributions
L.B. C.T. and B.F. wrote the main manuscript text. L.B prepared figures 1-3, B.F. prepared figure 4. P.C. D.K. J.K. and L.B. conceived of the study and secured funding. P.C. and D.K. collected samples and organized collection efforts. C.T., D.T., and L.E. performed lab work and provided lab support. All authors reviewed the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Berkman, L.K., Titus, C.L., Thomas, D.R. et al. Genetic differences among the Interior Highlands walleye (Sander vitreus) with mitochondrial and nuclear markers indicate the need for updated stocking practices. Conserv Genet 24, 347–359 (2023). https://doi.org/10.1007/s10592-023-01504-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10592-023-01504-7