Abstract
Due to geologic processes and recent anthropogenic introductions, patterns of genetic and morphological diversity within the Smallmouth Bass (Micropterus dolomieu), which are endemic to the central and eastern United States (USA), are poorly understood. We assessed genetic and morphological differentiation between the widespread Northern Smallmouth Bass (M. d. dolomieu) and the more restricted Neosho Smallmouth Bass (M. d. velox) where their ranges meet in the Central Interior Highlands ecoregion (CIH). Data from 14 microsatellite loci were used to conduct Structure and principal components analyses to evaluate diversity across populations and screen for hybridization with sympatric Spotted Bass (M. punctulatus). We also tested for morphological differences using five morphometric traits and one meristic trait. We found support for three genetic clusters corresponding to previously described taxonomic variation; five clusters largely corresponding to river systems; and nine clusters representing hierarchical population structure within both ranges. We found evidence of a unique genetic cluster in tributaries of the White River within the Northern Smallmouth Bass range and admixture between the subspecies throughout the Neosho range. We also found evidence of morphological differentiation between subspecies; Neosho Smallmouth Bass exhibited larger head length than Northern Smallmouth Bass relative to total length, and there was a significant interaction of subspecies and orbital length, possibly indicating differential growth patterns between subspecies. Our results reveal multiple levels of divergence, suggesting the CIH harbors greater and more complex Smallmouth Bass diversity than previously thought.



Similar content being viewed by others
Data availability
All metadata and raw data used in this study are available on Zenodo (https://doi.org/10.5281/zenodo.3937432).
Change history
24 August 2020
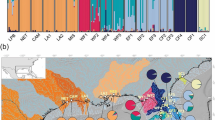
In the original publication of the article, Table 2 was published incorrectly. The correct Table 2 is given in this Correction.
References
Addison J, Spencer S (1971) Preliminary evaluation of three strains of Largemouth Bass, Micropterus salmoides (Lacepede), stocked in ponds in south Alabama. Proc Annu Conf Southeast Assoc Game Fish Comm 25:366–374
Avise JC, Pierce PC, Van Den Avyle MJ, Smith MH, Nelson WS, Asmussen MA (1997) Cytonuclear introgressive swam** and species turnover of bass after an introduction. J Hered 88:14–20
Bagley JC, Mayden RL, Roe KJ, Holznagel W, Harris PM (2011) Congeneric phylogeographical sampling reveals polyphyly and novel biodiversity within the black basses (Centrarchidae: Micropterus). Biol J Linn Soc 104:346–363. https://doi.org/10.1111/j.1095-8312.2011.01720.x
Bailey RM (1956) A revised list of the fishes of Iowa, with keys for identification. In: Harlan JR, Speaker EB (eds) Iowa fish and fishing. Des Moines, IA, pp 327–377
Barton NH (2001) The role of hybridization in evolution. Mol Ecol 10:551–568. https://doi.org/10.1046/j.1365-294x.2001.01216.x
Bermingham E, Rohwer S, Freeman S, Wood C (1992) Vicariance biogeography in the Pleistocene and speciation in North American wood warblers: a test of Mengel’s model. Proc Natl Acad Sci 89:6624–6628. https://doi.org/10.1073/pnas.89.14.6624
Bernatchez L, Wilson CC (1998) Comparative phylogeography of Nearctic and Palearctic fishes. Mol Ecol 7:431–452. https://doi.org/10.1046/j.1365-294x.1998.00319.x
Berner D (2011) Size correction in biology: how reliable are approaches based on (common) principal component analysis? Oecologia 166:961–971. https://doi.org/10.1007/s00442-011-1934-z
Bishop P (1995) Drainage rearrangement by river capture, beheading and diversion. Prog Phys Geog 19:449–473. https://doi.org/10.1177/030913339501900402
Blanca MJ, Alarcón R, Arnau J, Bono R, Bendayan R (2017) Non-normal data: is ANOVA still a valid option? Psicothema 29:552–557. https://doi.org/10.7334/psicothema2016.383
Bonnet S (2009) Shrinking and splitting of drainage basins in orogenic landscapes from the migration of the main drainage divide. Nat Geosci 2:766–771. https://doi.org/10.1038/ngeo666
Borden WC, Krebs RA (2009) Phylogeography and postglacial dispersal of Smallmouth Bass (Micropterus dolomieu) into the Great Lakes. Can J Fish Aquat Sci. https://doi.org/10.1139/F09-155
Boxrucker J, Echelle AA, Van Den Bussche RA (2004) Determining the degree of hybridization in the Smallmouth Bass population of Broken Bow Reservoir and the Mountain Fork River. In: Oklahoma Department of Wildlife Conservation, Oklahoma City, OK
Branson BA (1963) New mollusks from Oklahoma and their zoogeographical significance. Trans Kans Acad Sci 66:501–512. https://doi.org/10.2307/3626549
Branson BA (1967) Fishes of the Neosho River system in Oklahoma. Am Midl Nat 78:126–154. https://doi.org/10.2307/2423375
Brewer SK, Long JM (2015) Biology and ecology of Neosho Smallmouth Bass and the genetically distinct Ouachita lineage. In: Tringali MD, Long JM, Birdson TW, Allen MS (eds) Black bass diversity: multidisciplinary science for conservation. Bethesda, MD, pp 281–295
Brewer SK, Orth DJ (2015) Smallmouth Bass Micropterus dolomieu Lacepede, 1802. In: Tringali MD, Long JM, Birdsong TW, Allen MS (eds) Black bass diversity: multidisciplinary science for conservation. Bethesda, MD, pp 9–26
Bolnick DI, Near TJ (2005) Tempo of hybrid inviability in Centrarchid fishes (Teleostei: Centrarchidae). Evolution 59:1754–1767. https://doi.org/10.1111/j.0014-3820.2005.tb01824.x
Carlander KD (1977) Handbook of freshwater fish biology, vol 2. Ames, IA
Colbourne JK, Neff BD, Wright JM, Gross MR (1996) DNA fingerprinting of Bluegill sunfish (Lepomis macrochirus) using (GT)n microsatellites and its potential for assessment of mating success. Can J Fish Aquat Sci 53:342–349. https://doi.org/10.1139/cjfas-53-2-342
Dudgeon D, Arthington AH, Gessner MO, Kawabata ZI, Knowler DJ, Lévêque C, Naiman RJ et al (2006) Freshwater biodiversity: importance, threats, status and conservation challenges. Biol Rev 81:163–182. https://doi.org/10.1017/S1464793105006950
Dyer RJ (2016) gstudio: tools related to the spatial analysis of genetic marker data. R package version 1.5.0.
Evanno G, Regnaut S, Goudet J (2005) Detecting the number of clusters of individuals using the software structure: a simulation study. Mol Ecol 14:2611–2620. https://doi.org/10.1111/j.1365-294x.2005.02553.x
Francis RM (2017) pophelper: an R package and web app to analyze and visualize population structure. Mol Ecol Resour 17:27–32. https://doi.org/10.1111/1755-0998.12509
Frankel GH (1974) Genetic conservation: our evolutionary responsibility. Genetics 78:53–65
Gharaei A (2012) Morphometric and meristic studies of snow trout Schizothorax zarudnyi (Nikolskii, 1897) as a threatened endemic fish. World J Fish Mar Sci 4:426–429. https://doi.org/10.5829/idosi.wjfms.2012.04.04.63123
Gilbert CR (1998) Type catalogue of recent and fossil North American freshwater fishes: families Cyprinidae, Catostomidae, Ictaluridae, Centrarchidae, and Elassomatidae. Fl Mus Nat Hist Spec Pub 1:1–284. https://doi.org/10.2307/1447374
Hails RS, Morely K (2005) Genes invading new populations: a risk assessment perspective. Trends Ecol Evol 20:245–252. https://doi.org/10.1016/j.tree.2005.02.006
Hale ML, Burg TM, Stevens TE (2012) Sampling for microsatellite-based population genetic studies: 25 to 30 individuals per population is enough to accurately estimate allele frequencies. PLoS ONE 7:e45170
Hansen TF, Houle D (2008) Measuring and comparing evolvability and constraint in multivariate characters. J Evol Biol 21:1201–1219. https://doi.org/10.1111/j.1420-9101.2008.01573.x
Heim GE, Howe WB (1963) Pleistocene drainage and depositional history in northwestern Missouri. Trans Kans Acad Sci 66:378–392. https://doi.org/10.2307/3626530
Hedrick PW (2001) Conservation genetics: where are we now? Trends Ecol Evol 16:629–636. https://doi.org/10.1016/s0169-5347(01)02282-0
Herdegen M, Alexander HJ, Babik W, Mavárez J, Breden F, Radwan J (2014) Population structure of guppies in north-eastern Venezuela, the area of putative incipient speciation. BMC Evol Biol 14:1–14. https://doi.org/10.1186/1471-2148-14-28
Hoyt RD (1974) The effect of stocking on the meristic complement of the Neosho Smallmouth Bass. Proc Annu Conf Southeast Assoc Game Fish Comm 27:643–652
Hubbs CL, Bailey RM (1940) A revision of the black basses (Micropterus and Huro) with descriptions of four new forms. Mich Univ, Mus Zool, Misc Pub 48:1–49. https://doi.org/10.2307/1437996
Ihssen PE, Brooke HE, Casselman JM, McGlade JM, Payne NR, Utter FM (1981) Stock identification: materials and methods. Can J Fish Aquat Sci 38:1838–1855. https://doi.org/10.1139/f81-230
Jackson ZJ, Quist MC (2008) Growth standards for nine North American fish species. Fish Manag Ecol 15:107–118. https://doi.org/10.1111/j.1365-2400.2007.00591.x
Jakobsson M, Rosenberg NA (2007) CLUMPP: a cluster matching and permutation program for dealing with label switching and multimodality in analysis of population structure. Bioinform 23:1801–1806. https://doi.org/10.1093/bioinformatics/btm233
Janes JK, Miller JM, Dupuis JR, Malenfant RM, Gorrell JC, Cullingham CI, Andrew RL (2017) The K = 2 conundrum. Mol Ecol 26:3594–3602. https://doi.org/10.1111/mec.14187
Jennings MJ, Kampa JM, Hatzenbeler GR, Emmons EE (2005) Evaluation of supplemental walleye stocking in northern Wisconsin lakes. N Am J Fish Manage 25:1171–1178. https://doi.org/10.1577/m04-218.1
Johnson BM, Arlinghaus R, Martinez PJ (2009) Introduced species—are we doing all we can to stem the tide of illegal fish stocking? Fisheries 34:389–394. https://doi.org/10.1577/1548-8446-34.8.389
Kalinowski ST (2005) HP-Rare: a computer program for performing rarefaction on measures of allelic diversity. Mol Ecol Notes 5:187–189. https://doi.org/10.1111/j.1471-8286.2004.00845.x
Kassambara, A (2019) ggpubr: ggplot2 based publication ready plots. R Package Version 0.2.1
Kopelman NM, Mayzel J, Jakobsson M, Rosenberg NA, Mayrose I (2015) CLUMPAK: a program for identifying clustering models and packaging population structure inferences across K. Mol Ecol Resour 15:1179–1191. https://doi.org/10.1111/1755-0998.12387
Koppelman JB (1994) Hybridization between Smallmouth Bass, Micropterus dolomieu, and Spotted Bass, M. punctulatus, in the Missouri River System. Missouri Copeia 1994:204–210. https://doi.org/10.2307/1446686
Koppelman JB (2015) Black bass hybrids: a natural phenomenon in an unnatural world. In: Tringali MD, Long JM, Birdsong TW, Allen MS (eds) Black bass diversity: multidisciplinary science for conservation. Bethesda, MD, pp 467–479
Li YL, Liu JX (2018) StructureSelector: a web-based software to select and visualize the optimal number of clusters using multiple methods. Mol Ecol Resour 18:176–177. https://doi.org/10.1111/1755-0998.12719
Li C, Gowan S, Anil A, Beck BH, Thongda W, Kucuktas H et al (2015) Discovery and validation of gene-linked diagnostic SNP markers for assessing hybridization between largemouth bass (Micropterus salmoides) and Florida Bass (M. floridanus). Mol Ecol Resour 15:395–404. https://doi.org/10.1111/1755-0998.12308
Long JM, Allen MS, Porak WF, Suski CD (2015) A historical perspective of black bass management in the United States. In: Tringali MD, Long JM, Birdsong TW, Allen MS (eds) Black bass diversity: multidisciplinary science for conservation. Bethesda, MD, pp 99–122
Longmire JL, Maltbie M, Baker RJ (1997) Use of “lysis buffer” in DNA isolation and its implications for museum collections. Occas pap Mus Tex Tech Univ 163:1–3. https://doi.org/10.5962/bhl.title.143318
Lundberg JG, Kottelat M, Smith GR, Stiassny MLJ, Gill AC (2000) So many fishes, so little time: an overview of recent ichthyological discovery in continental waters. Ann Missouri Bot Gard 87:26–62. https://doi.org/10.2307/2666207
Malloy TP, Van Den Bussche RA, Coughlin WD, Echelle AA (2001) Isolation and characterization of microsatellite loci in Smallmouth Bass Micropterus dolomieu (Teleostei: Centrarchidae), and cross-species amplification in Spotted Bass, M. punctulatus. Mol Ecol 9:1919–1952. https://doi.org/10.1046/j.1365-294x.2000.01096-16.x
Matthews WJ, Robison HW (1988) The distribution of the fishes of Arkansas: a multivariate analysis. Copeia 2:358–374. https://doi.org/10.2307/1445876
Mayden RL (1988) Vicariance biogeography, parsimony, and evolution in North American freshwater fishes. Syst Biol 37:329–355. https://doi.org/10.1093/sysbio/37.4.329
McFadden D (1973) Conditional logit analysis of qualitative choice behavior. In: Zarembka P (ed) Frontiers in econometrics. Academic Press, New York, pp 105–142
Meirmans PG, Van Tienderen PH (2004) GENOTYPE and GENODIVE: two programs for the analysis of genetic diversity of sexual organisms. Mol Ecol Notes 4:792–794. https://doi.org/10.1111/j.1471-8286.2004.00770.x
Nigh TA, Schroeder WA (2002) Atlas of Missouri ecoregions. Missouri Department of Conservation, Jefferson City
Pflieger WL, Fajen OF (1975) Natural hybridization between the Smallmouth Bass and spotted bass. In: Final report, Missouri Department of Conservation, federal aid in fish restoration, Project F-1-R-24, Study S-7. Jefferson City, MO
Pierce PC, Van Den Ayyle MJ (1997) Hybridization between introduced spotted bass and Smallmouth Bass in reservoirs. Trans Am Fish Soc 126:939–947. https://doi.org/10.1577/1548-8659(1997)126<0939:hbisba>2.3.co;2
Pinheiro J, Bates D, DebRoy S, Sarkar D, R Core Team (2018) nlme: Linear and nonlinear mixed effects models. R package version 3.1-137. https://CRAN.R-project.org/package=nlme
Pregler KC, Kanno Y, Rankin D, Coombs JA, Whiteley AR (2018) Characterizing genetic integrity of rear-edge trout populations in the southern Appalachians. Conserv Genet 19:1487–1503. https://doi.org/10.1007/s10592-018-1116-1
Pritchard JK, Stephens M, Donnelly P (2000) Inference of population structure using multilocus genotype data. Genetics 155:945–959.
Puebla O (2009) Ecological speciation in marine v. freshwater fishes. J Fish Biol 75:960–996. https://doi.org/10.1111/j.1095-8649.2009.02358.x
Puechmaille SJ (2016) The program STRUCTURE does not reliably recover the correct population structure when sampling is uneven: subsampling and new estimators alleviate the problem. Mol Ecol Resour 16:608–627. https://doi.org/10.1111/1755-0998.12512
R Core Team (2018) R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria. https://www.R-project.org/
Rahel FJ (2004) Unauthorized fish introductions: fisheries management of the people, for the people, or by the people? In: Nickum MJ, Mazik PM, Nickum JG, MacKinlay DD (eds) Propagated fishes in resource management. Bethesda, MD, pp 431–444
Raymond M, Rousset F (1995) GENEPOP (version 1.2): population genetics software for exact tests and ecumenicism. J Hered 86:248–249. https://doi.org/10.1093/oxfordjournals.jhered.a111573
Rehman FU, Rehman HU, Aman S, Aziz S, Shabir H, Majid A et al (2015) Morphometric and meristic analysis of Silver Carp (Hypophthalmichthys molitrix) from Tanda Dam, Distric Kohat, Pakistan. Glob Vet 15:82–92. https://doi.org/10.5829/idosi.gv.2015.15.01.95217
Rhymer JM, Simberloff D (1996) Extinction by hybridization and introgression. Ann Rev Ecol Evol Syst 27:83–109. https://doi.org/10.1146/annurev.ecolsys.27.1.83
Robbins WH, MacCrimmon HR (1974) The black bass. In: Robbins WH, MacCrimmon HR (eds) America and overseas Sault St. Marie, Ontario, Canada, p 196
Rousset F (2008) Genepop’007: a complete reimplementation of the Genepop software for Windows and Linux. Mol Ecol Resour 8:103–106. https://doi.org/10.1111/j.1471-8286.2007.01931.x
Rundle HD, Nosil P (2005) Ecological speciation. Ecol Lett 8:336–352. https://doi.org/10.1111/j.1461-0248.2004.00715.x
Scott WB, Crossman EJ (1973). Freshwater fishes of Canada. Ottawa, Canada. https://www.dfo-mpo.gc.ca/Library/52369_Pt1.pdf
Seyoum S, Barthel BL, Tringali MD, Davis MC, Schmitt SL, Bellotti PS, Porak WF (2013) Isolation and characterization of eighteen microsatellite loci for the Largemouth Bass, Micropterus salmoides, and cross amplification in congeneric species. Conserv Genet Resour 5:697–701. https://doi.org/10.1007/s12686-013-9885-9
Signorell A, Aho K, Alfons A, Anderegg N, Aragon T, Arppe A et al (2019) DescTools: tools for descriptive statistics. R Package Version 0.99.28.
Smith HM (1956) Handbook of amphibians and reptiles of Kansas. Misc Pub Univ Kans Mus Nat Hist 9:1–356
Sneath PHA, Sokal RR (1973) In: Kennedy D, Park RB (eds) Numerical taxonomy: the principles and practice of numerical classification. W. H. Freeman and Co, San Francisco, pp 1–573
Snow JR (1975) Hatchery propagation of the black basses. In: Clepper H (ed) Black bass: biology and management. Sport Fishing Institute, Washington, pp 344–356
Stark WJ, Echelle AA (1998) Genetic structure and systematics of Smallmouth Bass, with emphasis on interior highlands populations. Trans Am Fish Soc 127:393–416. https://doi.org/10.1577/1548-8659(1998)127<0393:gsasos>2.0.co;2
Stepien CA, Karsiotis SI, Sullivan TJ, Klymus KE (2017) Population genetic structure and comparative diversity of Smallmouth Bass Micropterus dolomieu: Congruent patterns from two genomes. J Fish Biol 90:2125–2147. https://doi.org/10.1111/jfb.13296
Taylor AT, Long JM, Schwemm MR, Brewer SK (2018a) Hybridization and genetic structure of Neosho Smallmouth Bass in the Ozark Highlands. N Am J Fish Manage 38:1226–1240. https://doi.org/10.1002/nafm.10225
Taylor AT, Tringali MD, O’Rouke PM, Long JM (2018b) Shoal bass hybridization in the Chattahoochee River basin near Atlanta, Georgia. J Southeast Assoc Fish Wild Agcys 5:1–9
Taylor AT, Long JM, Tringali MD, Barthel BL (2019) Conservation of black bass diversity: an emerging management paradigm. Fisheries 44:20–36. https://doi.org/10.1002/fsh.10187
Thorpe JE, Koonce JF (1981) Assessing and managing man’s impact on fish genetic resources. Can J Fish Aquat Scis 38:1899–1907. https://doi.org/10.1139/f81-236
Turan C (1999) A note on the examination of morphometric differentiation among fish populations: the Truss system. Turk J Zool 23:259–263
Valiere N (2002) GIMLET: a computer program for analyzing genetic individual identification data. Mol Ecol Notes 2:377–379. https://doi.org/10.1046/j.1471-8286.2002.00228.x-i2
Venables WN, Ripley BD (2002) Modern applied statistics with S. Fourth edition. Springer, New York
Wang J (2017) The computer program Structure for assigning individuals to populations: easy to use but easier to misuse. Mol Ecol Resour 17:981–990. https://doi.org/10.1111/1755-0998.12650
Ward RD, Skibinski WM (1994) A comparison of genetic diversity levels in marine, freshwater, and anadromous fishes. J Fish Biol 44:213–232. https://doi.org/10.1111/j.1095-8649.1994.tb01200.x
Whitt GS, Childers WF, Wheat TE (1971) The inheritance of tissue specific lactate dehydrogenase isozymes in interspecific bass (Micropterus) hybrids. Biochem Genet 5:257–273. https://doi.org/10.1007/bf00485797
Acknowledgements
We thank the students, faculty, and staff at the University of Missouri, Arkansas Game and Fish Commission, Missouri Department of Conservation, and Arkansas Tech University who helped with sample collection. In particular, R. Horton of the Missouri Department of Conservation, J. Quinn of the Arkansas Game and Fish Commission, and C. Gagen at Arkansas Tech University, were essential for support, organization and sample collection. We thank K. Budd, J.J. Burkhart, M. Harris, K. Storey, and C. Titus for their assistance with data interpretation. Undergraduate researcher K. Sulkowski provided invaluable help with sample collection and DNA extraction. The Oklahoma Cooperative Fish and Wildlife Research Unit is a joint collaboration among U.S. Geological Survey, Oklahoma State University, the Oklahoma Department of Wildlife Conservation, the Wildlife Management Institute, and the U.S. Fish and Wildlife Service. Any use of trade, firm, or product names is for descriptive purposes only and does not imply endorsement by the U.S. Government. This project was supported financially by the Missouri Department of Conservation and conducted under the University of Missouri Animal Care and Use Permit #8826.
Funding
This project was funded by the Missouri Department of Conservation.
Author information
Authors and Affiliations
Contributions
Study conception and design were completed by JCG, LSE, LKB, and JK. Sample collection was conducted by JCG, LKB, JK, ATT, SB, and JML. Laboratory genetic work was conducted by JCG and LSE. All authors contributed to statistical analysis and manuscript preparation.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Gunn, J.C., Berkman, L.K., Koppelman, J. et al. Complex patterns of genetic and morphological differentiation in the Smallmouth Bass subspecies (Micropterus dolomieu dolomieu and M. d. velox) of the Central Interior Highlands. Conserv Genet 21, 891–904 (2020). https://doi.org/10.1007/s10592-020-01295-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10592-020-01295-1