Abstract
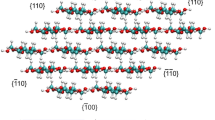
Phosphoric acid is widely used for the swelling and hydrolysis of cellulose. The detailed description of molecular interactions between cellulose and phosphoric acid is essential for understanding and controlling these processes. Here, to obtain structural insights into the swelling behavior, we investigated the structural evolution of cellulose swollen in concentrated phosphoric acid solution using X-ray fiber diffraction and solid-state NMR spectroscopy. We observed the formation of a crystalline complex of cellulose and phosphoric acid at − 40 °C, where cellulose molecules adopt a seven-fold helical conformation. This structure is the second known cellulose-acid crystalline complex and the first cellulosic crystal consisting of seven-fold helical chains. Our observation highlights the conformational flexibility of cellulose molecules in the solvated states and the strong influence of cellulose-acid interactions on the packing and conformation of cellulose molecules.
Graphical abstract






Similar content being viewed by others
Data availability
All data generated or analyzed during this study are included in this article.
References
Billès E, Onwukamike KN, Coma V et al (2016) Cellulose oligomers production and separation for the synthesis of new fully bio-based amphiphilic compounds. Carbohydr Polym 154:121–128. https://doi.org/10.1016/j.carbpol.2016.07.107
Boerstoel H, Maatman H, Westerink JB, Koenders BM (2001) Liquid crystalline solutions of cellulose in phosphoric acid. Polymer 42:7371–7379. https://doi.org/10.1016/S0032-3861(01)00210-5
Camarero Espinosa S, Kuhnt T, Foster EJ, Weder C (2013) Isolation of thermally stable cellulose nanocrystals by phosphoric acid hydrolysis. Biomacromolecules 14:1223–1230. https://doi.org/10.1021/bm400219u
Endo T, Yoshida S, Kimura Y (2020) Self-assembly and complexation of cellulose/ionic liquid at high cellulose concentration: anion dependence. Cryst Growth Des 20:6267–6271. https://doi.org/10.1021/acs.cgd.0c00346
Falcoz-Vigne L, Ogawa Y, Molina-Boisseau S et al (2017) Quantification of a tightly adsorbed monolayer of xylan on cellulose surface. Cellulose 24:3725–3739. https://doi.org/10.1007/s10570-017-1401-z
French AD (2017) Glucose, not cellobiose, is the repeating unit of cellulose and why that is important. Cellulose 24:4605–4609. https://doi.org/10.1007/s10570-017-1450-3
French AD, Johnson GP (2004) What crystals of small analogs are trying to tell us about cellulose structure. Cellulose 11:5–22. https://doi.org/10.1023/B:CELL.0000014765.94239.fe
French AD, Johnson GP (2009) Cellulose and the twofold screw axis: modeling and experimental arguments. Cellulose 16:959–973. https://doi.org/10.1007/s10570-009-9347-4
French AD, Johnson GP, Cramer CJ, Csonka GI (2012) Conformational analysis of cellobiose by electronic structure theories. Carbohydr Res 350:68–76. https://doi.org/10.1016/j.carres.2011.12.023
French AD, Montgomery DW, Prevost NT, Edwards JV, Woods RW (2021) Comparison of cellooligosaccharide conformations in complexes with proteins with energy maps for cellobiose. Carbohydr Polym 264:118004
Hall M, Bansal P, Lee JH et al (2010) Cellulose crystallinity: a key predictor of the enzymatic hydrolysis rate. FEBS J 277:1571–1582. https://doi.org/10.1111/j.1742-4658.2010.07585.x
Horii F, Hirai A, Kitamaru R (1983) Solid-state 13C-NMR study of conformations of oligosaccharides and cellulose. Polym Bull 10:357–361. https://doi.org/10.1007/BF00281948
Isogai A, Usuda M (1991) Preparation of low-molecular weight celluloses using phosphoric acid. Mokuzai Gakkaishi. 37:339–344
Lee DM, Blackwell J, Litt MH (1983) Structure of a cellulose II-hydrazine complex. Biopolymers 22:1383–1399. https://doi.org/10.1002/bip.360220510
Li W, Ogawa Y, Perez J et al (2022) Fivefold helical cellulose trapped in a sulfuric acid framework. Cryst Growth Des 22:20–25. https://doi.org/10.1021/acs.cgd.1c00664
Moffat J, Morris VJ, Al-Assaf S, Gunning AP (2016) Visualisation of xanthan conformation by atomic force microscopy. Carbohydr Polym 148:380–389. https://doi.org/10.1016/j.carbpol.2016.04.078
Morris ER (2019) Ordered conformation of xanthan in solutions and “weak gels”: Single helix, double helix–or both? Food Hydrocoll 86:18–25. https://doi.org/10.1016/j.foodhyd.2017.11.036
Nishiyama Y, Langan P, Chanzy H (2002) Crystal structure and hydrogen-bonding system in cellulose Iβ from synchrotron X-ray and neutron fiber diffraction. J Am Chem Soc 124:9074–9082. https://doi.org/10.1021/ja0257319
Nishiyama Y, Mazeau K, Morin M, Cardoso MB, Chanzy H, Putaux J-L (2010) Molecular and crystal structure of 7-fold V-amylose complexed with 2-propanol. Macromolecules 43(20):8628–8636
Nishiyama Y, Asaadi S, Ahvenainen P, Sixta H (2019) Water-induced crystallization and nano-scale spinodal decomposition of cellulose in NMMO and ionic liquid dope. Cellulose 26:281–289. https://doi.org/10.1007/s10570-018-2148-x
Northolt MG, Boerstoel H, Maatman H et al (2001) The structure and properties of cellulose fibres spun from an anisotropic phosphoric acid solution. Polymer 42:8249–8264. https://doi.org/10.1016/S0032-3861(01)00211-7
Okano T, Sarko A (1984) Mercerization of cellulose. I. X-ray diffraction evidence for intermediate structures. J Appl Polym Sci 29:4175–4182. https://doi.org/10.1002/app.1984.070291247
Okano T, Sarko A (1985) Mercerization of cellulose. II. Alkali–cellulose intermediates and a possible mercerization mechanism. J Appl Polym Sci 30:325–332. https://doi.org/10.1002/app.1985.070300128
Porro F, Bédué O, Chanzy H, Heux L (2007) Solid-state 13C NMR study of Na-cellulose complexes. Biomacromolecules 8:2586–2593. https://doi.org/10.1021/bm0702657
Schrödinger L, DeLano W (2020) PyMOL. http://www.pymol.org/pymol
Smith JP, Brown WE, Lehr JR (1955) Structure of crystalline phosphoric acid. J Am Chem Soc 77:2728–2730. https://doi.org/10.1021/ja01615a013
Sobue H, Kiessig H, Hess K (1939) Das system cellulose–natriumhydroxyd–wasser in Abhängigkeit von der temperatur. Z Für Phys Chem 43B:309–328. https://doi.org/10.1515/zpch-1939-4324
Song G, Yu J, Ding M, Zhang J (2018) A novel cellulose/ionic liquid complex crystal. Cryst Growth Des 18:4260–4264. https://doi.org/10.1021/acs.cgd.8b00754
Veregin RP, Fyfe CA, Marchessault RH, Taylor MG (1987) Correlation of 13C chemical shifts with torsional angles from high-resolution, 13C-C.P.-M.A.S. N.M.R. studies of crystalline cyclomalto-oligosaccharide complexes, and their relation to the structures of the starch polymorphs. Carbohydr Res 160:41–56. https://doi.org/10.1016/0008-6215(87)80302-6
Wada M, Heux L, Nishiyama Y, Langan P (2009) The structure of the complex of cellulose I with ethylenediamine by X-ray crystallography and cross-polarization/magic angle spinning 13C nuclear magnetic resonance. Cellulose 16:943–957. https://doi.org/10.1007/s10570-009-9338-5
Zhang Y-HP, Cui J, Lynd LR, Kuang LR (2006) A transition from cellulose swelling to cellulose dissolution by o-phosphoric acid: evidence from enzymatic hydrolysis and supramolecular structure. Biomacromolecules 7:644–648. https://doi.org/10.1021/bm050799c
Acknowledgments
We acknowledge Agence Nationale de la Recherche (ANR Grant Number: ANR-21-CE29-0016-1 and ANR-11-EQPX-0010 under the “Investissements d’Avenir” program) and Glyco@Alps (ANR-15-IDEX-02) for the financial support and the NanoBio-ICMG platform (FR 2607) for granting access to the NMR facility. ESRF is acknowledged for the provision of beamtime (experiment number A02-1-902, D2AM beamline). We acknowledge Benjamin Richter and Yves Watier at the Sample Environment Service of ESRF, who designed the temperature-controlled specimen holder.
Author information
Authors and Affiliations
Contributions
IM and YO designed the experiments. JHL, IM, IJ, YN, and YO performed the experiments. JHL, ADF, YN, and YO analyzed the data. JHL, ADF, and YO prepared the figures and wrote the manuscript. All the authors approved the final version of the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Consent for publication
All authors have provided their consent for publication.
Ethics approval and consent to participate
Not applicable.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Lim, J.H., Morfin, I., Jeacomine, I. et al. The occurrence of seven-fold helical molecular conformation in cellulose-phosphoric acid complex. Cellulose 30, 8063–8073 (2023). https://doi.org/10.1007/s10570-023-05346-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10570-023-05346-x