Abstract
Background
Urinary extracellular vesicles (EVs) have gained increasing interest in recent years as a potential source of noninvasive biomarkers of diseases related to urinary organs, but knowledge of the mechanism is still limited. The current study sought to clarify the mechanism of urinary EVs behind di-(2-ethylhexyl) phthalate (DEHP)-induced hypospadias via PFN2 delivery.
Method
PFN2 expression in hypospadias was predicted by bioinformatics analysis. Following the induction of a hypospadias rat model using DEHP, rats were injected with EVs and/or underwent alteration of PFN2 and TGF-β1 to assess their effects in vivo. The extracted rat urothelial cells (UECs) were co-cultured with EVs extracted from urine for in vitro experiments.
Result
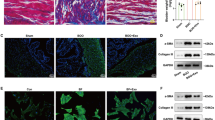
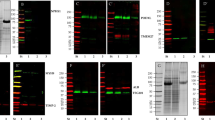
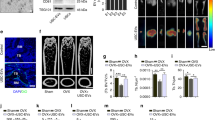
Microarray analysis predicted poor PFN2 expression in hypospadias. Upregulated PFN2 was found in urinary EVs, and restrained epithelial-mesenchymal transition (EMT) was observed in DEHP-exposed rats. Urinary EVs or PFN2 overexpression increased SMAD2, SMAD3, and TGF-β1 protein expression and SMAD2 and SMAD3 phosphorylation in UECs and DEHP-exposed rats. UEC migration, invasion, and EMT were augmented by EV co-culture or upregulation of PFN2. Of note, the silencing of TGF-β1 counterweighed the effect of PFN2. Besides, EV co-culture or overexpression of PFN2 or TGF-β1 elevated the body weight, anal–genital distance (AGD), anal–genital index (AGI), and EMT of DEHP-exposed rats.
Conclusion
In summary, urinary EVs activated the SMAD/TGF-β1 pathway to induce EMT via PFN2 delivery, thus protecting against DEHP-induced hypospadias.
Graphical Abstract
(1) EMT in epithelial cells inhibits DEHP-induced hypospadias.
(2) Urine-derived EVs deliver PFN2 to promote EMT in epithelial cells.
(3) PFN2 can activate the SMAD/TGF-β1 signaling axis.
(4) Urine-derived EVs can transmit PFN2 to activate the SMAD/TGF-β1 signaling axis, thus promoting EMT and inhibiting the occurrence of hypospadias.










Similar content being viewed by others
Data Availability
The data and materials of the study can be obtained from the corresponding author upon request.
Code availability
Not applicable.
References
Cao Q, Liu Y, Wu Y, Hu C, Sun L, Wang J, Li C, Guo M, Liu X, Lv J, Huo X, Yue J, Du X, Chen Z. Profilin 2 promotes growth, metastasis, and angiogenesis of small cell lung cancer through cancer-derived exosomes. Aging (Albany NY). 2020;12:25981–99. https://doi.org/10.18632/aging.202213.
Chen X, Chen RX, Wei WS, Li YH, Feng ZH, Tan L, Chen JW, Yuan GJ, Chen SL, Guo SJ, **ao KH, Liu ZW, Luo JH, Zhou FJ, **e D. PRMT5 circular RNA promotes metastasis of urothelial carcinoma of the bladder through sponging miR-30c to induce epithelial-mesenchymal transition. Clin Cancer Res. 2018;24:6319–30. https://doi.org/10.1158/1078-0432.CCR-18-1270.
Cui XB, Zhang SM, Xu YX, Dang HW, Liu CX, Wang LH, Yang L, Hu JM, Liang WH, Jiang JF, Li N, Li Y, Chen YZ, Li F. PFN2, a novel marker of unfavorable prognosis, is a potential therapeutic target involved in esophageal squamous cell carcinoma. J Transl Med. 2016;14:137. https://doi.org/10.1186/s12967-016-0884-y.
Ding H, Li LX, Harris PC, Yang J, Li X. Extracellular vesicles and exosomes generated from cystic renal epithelial cells promote cyst growth in autosomal dominant polycystic kidney disease. Nat Commun. 2021;12:4548. https://doi.org/10.1038/s41467-021-24799-x.
Erdbrugger U, Blijdorp CJ, Bijnsdorp IV, Borras FE, Burger D, Bussolati B, Byrd JB, Clayton A, Dear JW, Falcon-Perez JM, Grange C, Hill AF, Holthofer H, Hoorn EJ, Jenster G, Jimenez CR, Junker K, Klein J, Knepper MA, Koritzinsky EH, Luther JM, Lenassi M, Leivo J, Mertens I, Musante L, Oeyen E, Puhka M, van Royen ME, Sanchez C, Soekmadji C, Thongboonkerd V, van Steijn V, Verhaegh G, Webber JP, Witwer K, Yuen PST, Zheng L, Llorente A, Martens-Uzunova ES. Urinary extracellular vesicles: a position paper by the urine task force of the International Society for Extracellular Vesicles. J Extracell Vesicles. 2021;10:e12093. https://doi.org/10.1002/jev2.12093.
Franzen CA, Blackwell RH, Todorovic V, Greco KA, Foreman KE, Flanigan RC, Kuo PC, Gupta GN. Urothelial cells undergo epithelial-to-mesenchymal transition after exposure to muscle invasive bladder cancer exosomes. Oncogenesis. 2015;4:e163. https://doi.org/10.1038/oncsis.2015.21.
Huan J, Hornick NI, Shurtleff MJ, Skinner AM, Goloviznina NA, Roberts CT Jr, Kurre P. RNA trafficking by acute myelogenous leukemia exosomes. Cancer Res. 2013;73:918–29. https://doi.org/10.1158/0008-5472.CAN-12-2184.
Jiang M, Qiu N, **a H, Liang H, Li H, Ao X. Long noncoding RNA FOXD2AS1/miR1505p/PFN2 axis regulates breast cancer malignancy and tumorigenesis. Int J Oncol. 2019;54:1043–52. https://doi.org/10.3892/ijo.2019.4671.
Joodi M, Amerizadeh F, Hassanian SM, Erfani M, Ghayour-Mobarhan M, Ferns GA, Khazaei M, Avan A. The genetic factors contributing to hypospadias and their clinical utility in its diagnosis. J Cell Physiol. 2019;234:5519–23. https://doi.org/10.1002/jcp.27350.
Karpman D, Tontanahal A. Extracellular vesicles in renal inflammatory and infectious diseases. Free Radic Biol Med. 2021;171:42–54. https://doi.org/10.1016/j.freeradbiomed.2021.04.032.
Lamouille S, Xu J, Derynck R. Molecular mechanisms of epithelial-mesenchymal transition. Nat Rev Mol Cell Biol. 2014;15:178–96. https://doi.org/10.1038/nrm3758.
Li M, Qiu L, Zhang Y, Hua Y, Tu S, He Y, Wen S, Wang Q, Wei G. Dose-related effect by maternal exposure to di-(2-ethylhexyl) phthalate plasticizer on inducing hypospadiac male rats. Environ Toxicol Pharmacol. 2013;35:55–60. https://doi.org/10.1016/j.etap.2012.10.006.
Ling Y, Cao Q, Liu Y, Zhao J, Zhao Y, Li K, Chen Z, Du X, Huo X, Kang H, Chen Z. Profilin 2 (PFN2) promotes the proliferation, migration, invasion and epithelial-to-mesenchymal transition of triple negative breast cancer cells. Breast Cancer. 2021;28:368–78. https://doi.org/10.1007/s12282-020-01169-x.
Liu Z, Sun J, Li C, Xu L, Liu J. MKL1 regulates hepatocellular carcinoma cell proliferation, migration and apoptosis via the COMPASS complex and NF-kappaB signaling. BMC Cancer. 2021;21:1184. https://doi.org/10.1186/s12885-021-08185-w.
Mathivanan S, Ji H, Simpson RJ. Exosomes: extracellular organelles important in intercellular communication. J Proteomics. 2010;73:1907–20. https://doi.org/10.1016/j.jprot.2010.06.006.
Merchant ML, Rood IM, Deegens JKJ, Klein JB. Isolation and characterization of urinary extracellular vesicles: implications for biomarker discovery. Nat Rev Nephrol. 2017;13:731–49. https://doi.org/10.1038/nrneph.2017.148.
Mizutani K, Kawakami K, Horie K, Fujita Y, Kameyama K, Kato T, Nakane K, Tsuchiya T, Yasuda M, Masunaga K, Kasuya Y, Masuda Y, Deguchi T, Koie T, Ito M. Urinary exosome as a potential biomarker for urinary tract infection. Cell Microbiol. 2019;21:e13020. https://doi.org/10.1111/cmi.13020.
Moon PG, You S, Lee JE, Hwang D, Baek MC. Urinary exosomes and proteomics. Mass Spectrom Rev. 2011;30:1185–202. https://doi.org/10.1002/mas.20319.
Raimondo F, Corbetta S, Morosi L, Chinello C, Gianazza E, Castoldi G, Di Gioia C, Bombardi C, Stella A, Battaglia C, Bianchi C, Magni F, Pitto M. Urinary exosomes and diabetic nephropathy: a proteomic approach. Mol Biosyst. 2013;9:1139–46. https://doi.org/10.1039/c2mb25396h.
Ren J, Ding L, Zhang D, Shi G, Xu Q, Shen S, Wang Y, Wang T, Hou Y. Carcinoma-associated fibroblasts promote the stemness and chemoresistance of colorectal cancer by transferring exosomal lncRNA H19. Theranostics. 2018;8:3932–48. https://doi.org/10.7150/thno.25541.
Ricklefs FL, Maire CL, Reimer R, Duhrsen L, Kolbe K, Holz M, Schneider E, Rissiek A, Babayan A, Hille C, Pantel K, Krasemann S, Glatzel M, Heiland DH, Flitsch J, Martens T, Schmidt NO, Peine S, Breakefield XO, Lawler S, Chiocca EA, Fehse B, Giebel B, Gorgens A, Westphal M, Lamszus K. Imaging flow cytometry facilitates multiparametric characterization of extracellular vesicles in malignant brain tumours. J Extracell Vesicles. 2019;8:1588555. https://doi.org/10.1080/20013078.2019.1588555.
Rikkert LG, de Rond L, van Dam A, van Leeuwen TG, Coumans FAW, de Reijke TM, Terstappen L, Nieuwland R. Detection of extracellular vesicles in plasma and urine of prostate cancer patients by flow cytometry and surface plasmon resonance imaging. PLoS ONE. 2020;15:e0233443. https://doi.org/10.1371/journal.pone.0233443.
Shang Y, Kang Y, Sun J, Wei P, Yang J, Zhang H. MiR-145-modulated SOX9-mediated hypospadias through acting on mitogen-activated protein kinase signaling pathway. J Cell Physiol. 2019;234:10397–410. https://doi.org/10.1002/jcp.27708.
Song Y, Wang Z, Jiang J, Piao Y, Li L, Xu C, Piao H, Li L, Yan G. DEK-targeting aptamer DTA-64 attenuates bronchial EMT-mediated airway remodelling by suppressing TGF-beta1/SMAD, MAPK and PI3K signalling pathway in asthma. J Cell Mol Med. 2020;24:13739–50. https://doi.org/10.1111/jcmm.15942.
Street JM, Koritzinsky EH, Glispie DM, Yuen PST. Urine exosome isolation and characterization. Methods Mol Biol. 2017;1641:413–23. https://doi.org/10.1007/978-1-4939-7172-5_23.
Sun J, Han S, Ma L, Zhang H, Zhan Z, Aguilar HA, Zhang H, **ao K, Gu Y, Gu Z, Tao WA. Synergistically bifunctional paramagnetic separation enables efficient isolation of urine extracellular vesicles and downstream phosphoproteomic analysis. ACS Appl Mater Interfaces. 2021;13:3622–30. https://doi.org/10.1021/acsami.0c19400.
Tang YN, Ding WQ, Guo XJ, Yuan XW, Wang DM, Song JG. Epigenetic regulation of SMAD2 and SMAD3 by profilin-2 promotes lung cancer growth and metastasis. Nat Commun. 2015;6:8230. https://doi.org/10.1038/ncomms9230.
Tian RH, Guo KM, Han GH, Bai Y. Downregulation of MicroRNA-494 inhibits the TGF-beta1/SMADs signaling pathway and prevents the development of hypospadias through upregulating NEDD4L. Exp Mol Pathol. 2020;115:104452. https://doi.org/10.1016/j.yexmp.2020.104452.
van der Horst HJ, de Wall LL. Hypospadias, all there is to know. Eur J Pediatr. 2017;176:435–41. https://doi.org/10.1007/s00431-017-2864-5.
Wang Y, Wen M, Kwon Y, Xu Y, Liu Y, Zhang P, He X, Wang Q, Huang Y, Jen KY, LaBarge MA, You L, Kogan SC, Gray JW, Mao JH, Wei G. CUL4A induces epithelial-mesenchymal transition and promotes cancer metastasis by regulating ZEB1 expression. Cancer Res. 2014;74:520–31. https://doi.org/10.1158/0008-5472.CAN-13-2182.
Wei R, Zhao L, Kong G, Liu X, Zhu S, Zhang S, Min L. Combination of size-exclusion chromatography and ultracentrifugation improves the proteomic profiling of plasma-derived small extracellular vesicles. Biol Proced Online. 2020;22:12. https://doi.org/10.1186/s12575-020-00125-5.
Wu ZR, Yan L, Liu YT, Cao L, Guo YH, Zhang Y, Yao H, Cai L, Shang HB, Rui WW, Yang G, Zhang XB, Tang H, Wang Y, Huang JY, Wei YX, Zhao WG, Su B, Wu ZB. Inhibition of mTORC1 by lncRNA H19 via disrupting 4E-BP1/raptor interaction in pituitary tumours. Nat Commun. 2018;9:4624. https://doi.org/10.1038/s41467-018-06853-3.
Yan J, Ma C, Gao Y. MicroRNA-30a-5p suppresses epithelial-mesenchymal transition by targeting profilin-2 in high invasive non-small cell lung cancer cell lines. Oncol Rep. 2017;37:3146–54. https://doi.org/10.3892/or.2017.5566.
Yao Y, Chen R, Wang G, Zhang Y, Liu F. Exosomes derived from mesenchymal stem cells reverse EMT via TGF-beta1/SMAD pathway and promote repair of damaged endometrium. Stem Cell Res Ther. 2019;10:225. https://doi.org/10.1186/s13287-019-1332-8.
Zhang W, Zhou X, Zhang H, Yao Q, Liu Y, Dong Z. Extracellular vesicles in diagnosis and therapy of kidney diseases. Am J Physiol Renal Physiol. 2016;311:F844–51. https://doi.org/10.1152/ajprenal.00429.2016.
Zhang Y, Meng W, Yue P, Li X. M2 macrophage-derived extracellular vesicles promote gastric cancer progression via a microRNA-130b-3p/MLL3/GRHL2 signaling cascade. J Exp Clin Cancer Res. 2020;39:134. https://doi.org/10.1186/s13046-020-01626-7.
Zhou Y, Huang F, Liu Y, Li D, Zhou Y, Shen L, Long C, Liu X, Wei G. TGF-beta1 relieves epithelial-mesenchymal transition reduction in hypospadias induced by DEHP in rats. Pediatr Res. 2020;87:639–46. https://doi.org/10.1038/s41390-019-0622-2.
Zhu YP, Zhao W, Sun WL, Zhao S, Chen M, Pan L, Zhou Z, **a SJ, Jiang JT. Di-n-butyl phthalate (DBP) reduces epithelial-mesenchymal transition via IP3R in hypospadias during maternal exposure. Ecotoxicol Environ Saf. 2020;192:110201. https://doi.org/10.1016/j.ecoenv.2020.110201.
Funding
This study was funded by the Science and Technology Projects in Guangzhou (202102020097, 202201020598) and the Guangzhou Institute of Pediatrics/Guangzhou Women and Children’s Medical Center (pre‐NSFC‐2018‐016, YIP‐2018‐021, GWCMC2020‐4‐009) and the Natural Science Foundation of Guangdong Province, China (2019A1515011178).
Author information
Authors and Affiliations
Contributions
Shibo Zhu and **angliang Tang contributed to the conception and design of the study; ** Zhang and Dian Li contributed to the acquisition of data; **hua Hu contributed to the analysis and interpretation of data; Shibo Zhu and **aofeng Gao contributed to drafting the article; **angliang Tang and Wei Jia contributed to revising the article critically for important intellectual content; all of the authors approved the final version to be submitted.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
This study was approved by Guangzhou Women and Children’s Medical Center, Guangzhou Medical University and in strict accordance with the recommendations of the Guide for the Care and Use of Laboratory Animals published by the US National Institutes of Health. Adequate measures were taken to minimize suffering of the included animals. Consent to participate is not applicable.
Consent for publication
Not applicable.
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Zhu, S., Tang, X., Zhang, J. et al. Urinary extracellular vesicles prevent di-(2-ethylhexyl) phthalate-induced hypospadias by facilitating epithelial–mesenchymal transition via PFN2 delivery. Cell Biol Toxicol 39, 2569–2586 (2023). https://doi.org/10.1007/s10565-023-09838-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10565-023-09838-1