Abstract
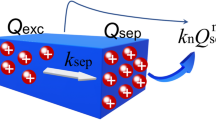
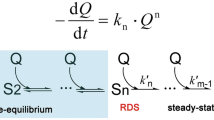
For artificial photosynthesis, the lower photon conversion efficiencies of the photogenerated charges hinder the practical application in solar energy harvesting. The challenges are commonly ascribed to the sluggish surface redox reactions and significant surface charge recombination. Although there are several transient techniques applied for monitoring the surface charges evolution, how the experimental spectroscopy data is interpreted still involves ambiguous explanations on the complicated reactions. Here, we firstly developed a parallel 1st–nth order reaction model, which could be applied to quantitatively analyze the surface charge reactions. The microkinetic calculations were carried out by varying the reaction order, rate constants as well as the contribution of the competitive ones. Interestingly, the simplified reaction model can successfully demonstrate the charges react rate changes, which can be further applied in mechanism study complementary with those experimental methods.
Graphic Abstract





Similar content being viewed by others
Data Availability
The datasets generated during and/or analysed during the current study are not publicly available due financial support, but are available from the corresponding author on reasonable request.
References
Fujishima A, Honda K (1972) Electrochemical photolysis of water at a semiconductor electrode. Nature 238:37–38
Walter MG, Warren EL, McKone JR, Boettcher SW, Mi Q, Santori EA, Lewis NS (2010) Solar water splitting cells. Chem Rev 110:6446–6473
Wang Q, Domen K (2020) Particulate photocatalysts for light-driven water splitting: mechanisms, challenges, and design strategies. Chem Rev 120:919–985
Wang Z, Li C, Domen K (2019) Recent developments in heterogeneous photocatalysts for solar-driven overall water splitting. Chem Soc Rev 48:2109–2125
Lewis NS (2016) Research Opportunities to Advance Solar Energy Utilization. Science 351:aad1920
Yang X, Wang D (2017) Chapter two—photophysics and photochemistry at the semiconductor/electrolyte interface for solar water splitting. In: Mi Z, Wang L, Jagadish C (eds) Semiconductors and semimetals, vol 97. Elsevier, Amsterdam, pp 47–80. https://doi.org/10.1016/bs.semsem.2017.03.001
Yang X, Wang D (2018) Photocatalysis: from fundamental principles to materials and applications. ACS Appl Energy Mater 1:6657–6693
Kafizas A, Ma Y, Pastor E, Pendlebury SR, Mesa C, Francàs L, Le Formal F, Noor N, Ling M, Sotelo-Vazquez C, Carmalt CJ, Parkin IP, Durrant JR (2017) Water oxidation kinetics of accumulated holes on the surface of a TiO2 photoanode: a rate law analysis. ACS Catal 7:4896–4903
Zhang P, Wang T, Gong J (2018) Current mechanistic understanding of surface reactions over water-splitting photocatalysts. Chem 4:223–245
Suen N-T, Hung S-F, Quan Q, Zhang N, Xu Y-J, Chen HM (2017) Electrocatalysis for the oxygen evolution reaction: recent development and future perspectives. Chem Soc Rev 46:337–365
Takata T, Jiang J, Sakata Y, Nakabayashi M, Shibata N, Nandal V, Seki K, Hisatomi T, Domen K (2020) Photocatalytic water splitting with A quantum efficiency of almost unity. Nature 581:411–414
Mesa CA, Francàs L, Yang KR, Garrido-Barros P, Pastor E, Ma Y, Kafizas A, Rosser TE, Mayer MT, Reisner E, Grätzel M, Batista VS, Durrant JR (2020) Multihole water oxidation catalysis on haematite photoanodes revealed by operando spectroelectrochemistry and DFT. Nat Chem 12:82–89
Zhang Y, Zhang H, Liu A, Chen C, Song W, Zhao J (2018) Rate-limiting O-O bond formation pathways for water oxidation on hematite photoanode. J Am Chem Soc 140:3264–3269
Chatman S, Zarzycki P, Rosso KM (2015) Spontaneous water oxidation at hematite (α-Fe2O3) crystal faces. ACS Appl Mater Interfaces 7:1550–1559
Li J, Wan W, Triana CA, Chen H, Zhao Y, Mavrokefalos CK, Patzke GR (2021) Reaction kinetics and interplay of two different surface states on hematite photoanodes for water oxidation. Nat Commun 12:255
Zhang H, Liu D, Ren S, Zhang H (2017) Kinetic studies of direct blue photodegradation over flower-like TiO2. Res Chem Intermed 43:1529–1542
Guo H, Ke Y, Wang D, Lin K, Shen R, Chen J, Weng W (2013) Efficient adsorption and photocatalytic degradation of congo red onto hydrothermally synthesized nis nanoparticles. J Nanopart Res 15:1475
Kosaka T, Teduka Y, Ogura T, Zhou Y, Hisatomi T, Nishiyama H, Domen K, Takahashi Y, Onishi H (2020) Transient kinetics of O2 evolution in photocatalytic water-splitting reaction. ACS Catal 10:13159–13164
Ollis DF (2005) Kinetics of liquid phase photocatalyzed reactions: an illuminating approach. J Phys Chem B 109:2439–2444
Le Formal F, Pastor E, Tilley SD, Mesa CA, Pendlebury SR, Grätzel M, Durrant JR (2015) Rate law analysis of water oxidation on a hematite surface. J Am Chem Soc 137:6629–6637
Ma Y, Mesa CA, Pastor E, Kafizas A, Francàs L, Le Formal F, Pendlebury SR, Durrant JR (2016) Rate law analysis of water oxidation and hole scavenging on a BiVO4 photoanode. ACS Energy Lett 1:618–623
Corby S, Pastor E, Dong Y, Zheng X, Francàs L, Sachs M, Selim S, Kafizas A, Bakulin AA, Durrant JR (2019) Charge separation, band-bending, and recombination in WO3 photoanodes. J Phys Chem Lett 10:5395–5401
Mesa CA, Rao RR, Francas L, Corby S, Durrant JR (2020) Reply to: questioning the rate law in the analysis of water oxidation catalysis on haematite photoanodes. Nat Chem 12:1099–1101
Zhang S, Leng W (2020) Questioning the rate law in the analysis of water oxidation catalysis on haematite photoanodes. Nat Chem 12:1097–1098
Gamelin DR (2012) Catalyst or spectator? Nat Chem 4:965–967
Li P, Chen X, He H, Zhou X, Zhou Y, Zou Z (2018) Polyhedral 30-faceted BiVO4 microcrystals predominantly enclosed by high-index planes promoting photocatalytic water-splitting activity. Adv Mater 30:1703119
Wang S, Liu G, Wang L (2019) Crystal facet engineering of photoelectrodes for photoelectrochemical water splitting. Chem Rev 119:5192–5247
Wang D, Sheng T, Chen J, Wang H-F, Hu P (2018) Identifying the key obstacle in photocatalytic oxygen evolution on rutile TiO2. Nat Catal 1:291–299
Ouhbi H, Aschauer U (2019) Water oxidation catalysis on reconstructed NaTaO3 (001) surfaces. J Mater Chem A 7:16770–16776
Choi JY, Jeong D, Lee SJ, Kang D-g, Kim SK, Nam KM, Song H (2017) Engineering reaction kinetics by tailoring the metal tips of metal-semiconductor nanodumbbells. Nano Lett 17:5688–5694
Ardagh MA, Shetty M, Kuznetsov A, Zhang Q, Christopher P, Vlachos DG, Abdelrahman OA, Dauenhauer PJ (2020) Catalytic resonance theory: parallel reaction pathway control. Chem Sci 11:3501–3510
Mesa CA, Kafizas A, Francas L, Pendlebury SR, Pastor E, Ma YM, Le Formal F, Mayer MT, Gratzel M, Durrant JR (2017) Kinetics of photoelectrochemical oxidation of methanol on hematite photoanodes. J Am Chem Soc 139:11537–11543
Zhang S, Zhao Y, Shi R, Zhou C, Waterhouse GIN, Wang Z, Weng Y, Zhang T (2021) Sub-3 nm ultrafine Cu2O for visible light driven nitrogen fixation. Angew Chem Int Ed 60:2554–2560
Katoh R, Furube A, Yamanaka K-i, Morikawa T (2010) Charge separation and trap** in N-doped TiO2 photocatalysts: a time-resolved microwave conductivity study. J Phys Chem Lett 1:3261–3265
Sandberg OJ, Tvingstedt K, Meredith P, Armin A (2019) Theoretical perspective on transient photovoltage and charge extraction techniques. J Phys Chem C 123:14261–14271
Li S, Hou L, Zhang L, Chen L, Lin Y, Wang D, **e T (2015) Direct evidence of the efficient hole collection process of the CoOx cocatalyst for photocatalytic reactions: a surface photovoltage study. J Mater Chem A 3:17820–17826
Bhosale SS, Kharade AK, Jokar E, Fathi A, Chang S-m, Diau EW-G (2019) Mechanism of photocatalytic CO2 reduction by bismuth-based perovskite nanocrystals at the gas-solid interface. J Am Chem Soc 141:20434–20442
Bian Z, Tachikawa T, Kim W, Choi W, Majima T (2012) Superior electron transport and photocatalytic abilities of metal-nanoparticle-loaded TiO2 superstructures. J Phys Chem C 116:25444–25453
Elbanna O, Fujitsuka M, Kim S, Majima T (2018) Charge carrier dynamics in TiO2 mesocrystals with oxygen vacancies for photocatalytic hydrogen generation under solar light irradiation. J Phys Chem C 122:15163–15170
Lu K, Lei Y, Qi R, Liu J, Yang X, Jia Z, Liu R, ** for efficient ITO/perovskite junction solar cells. J Mater Chem A 5:25211–25219
Li L, Yang X, Lei Y, Yu H, Yang Z, Zheng Z, Wang D (2018) Ultrathin Fe-NiO nanosheets as catalytic charge reservoirs for a planar mo-doped BiVO4 photoanode. Chem Sci 9:8860–8870
Le Formal F, Pendlebury SR, Cornuz M, Tilley SD, Grätzel M, Durrant JR (2014) Back electron-hole recombination in hematite photoanodes for water splitting. J Am Chem Soc 136:2564–2574
Schneider J, Bahnemann DW (2013) Undesired role of sacrificial reagents in photocatalysis. J Phys Chem Lett 4:3479–3483
Upul Wijayantha KG, Saremi-Yarahmadi S, Peter LM (2011) Kinetics of oxygen evolution at a-Fe2O3 photoanodes: a study by photoelectrochemical impedance spectroscopy. Phys Chem Chem Phys 13:5264–5270
Thorne JE, Jang J-W, Liu EY, Wang D (2016) Understanding the origin of photoelectrode performance enhancement by probing surface kinetics. Chem Sci 7:3347–3354
Galland C, Ghosh Y, Steinbrück A, Sykora M, Hollingsworth JA, Klimov VI, Htoon H (2011) Two types of luminescence blinking revealed by spectroelectrochemistry of single quantum dots. Nature 479:203–207
Tang L, Ji R, Li X, Teng KS, Lau SP (2013) Energy-level structure of nitrogen-doped graphene quantum dots. J Mater Chem C 1:4908–4915
Yang X, Wang Y, Li CM, Wang D (2021) Mechanisms of water oxidation on heterogeneous catalyst surfaces. Nano Res. https://doi.org/10.1007/s12274-12021-13607-12275
Acknowledgements
This work is financially supported from National Natural Science Foundation of China (Project. Nos. 61504117, 52072327, 21673200 and U1604121), Natural Science Foundation of Jiangsu Province (BK20180103).
Author information
Authors and Affiliations
Contributions
The manuscript was written through contributions of all authors. All authors have given approval to the final version of the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Supporting Information
The Supporting Information is available free of charge on at DOI: The linear relationship of ln(Qexc0/Qsep) vs time and (Qexc0/Qsep)n−1 ~ t curves (PDF).
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Wang, X., Hu, J., Qu, J. et al. Chemical Kinetics of Parallel Consuming Processes for Photogenerated Charges at the Semiconductor Surfaces: A Theoretical Classical Calculation. Catal Lett 152, 2470–2479 (2022). https://doi.org/10.1007/s10562-021-03832-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10562-021-03832-0