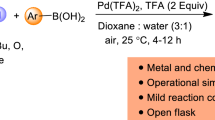
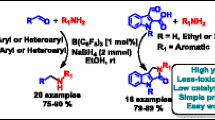
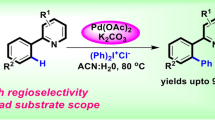
Abstract
A regioselective C2 arylation of indoles, benzothiophene and benzofuran without directing group has been accomplished using economically cheap Pd NPs and NHC-Pd@MNPs catalyst. The reusable catalyst is efficiently employed to access C2 arylated heterocycles in good to excellent yield. The reusability of the catalyst is studied up to five cycles and a gram-scale synthesis has been achieved. The reaction mechanism is well supported by control experiments and literature precedents.
Grapic Abstract





Similar content being viewed by others
References
Wang D-H, Engle KM, Shi B-F, Yu J-Q (2010) Science 327:315–319
Daugulis O, Do H-Q, Shabashov D (2009) Acc Chem Res 42:1074–1086
Ackermann L, Vicente R, Kapdi AR (2009) Angew Chem Int Ed 48:9792–9826
Lyons TW, Sanford MS (2010) Chem Rev 110:1147–1169
McGlacken GP, Bateman LM (2009) Chem Soc Rev 38:2447–2464
Alberico D, Scott ME, Lautens M (2007) Chem Rev 107:174–238
Godula K, Sames D (2006) Science 312:67–72
Colby DA, Bergman RG, Ellman JA (2010) Chem Rev 110:624–655
Kuhl N, Hopkinson MN, Delord JW, Glorius F (2012) Angew Chem Int Ed 51:2–21
Girard SA, Knauber T, Li CJ (2014) Angew Chem Int Ed 53:74–100
Ackermann L (2011) Chem Rev 111:1315–1345
Kochanowska-Karamyan AJ, Hamann MT (2010) Chem Rev 110:4489–4497
Somei M, Yamada F (2004) Nat Prod Rep 21:278–311
Ambros R, Schneider MR, von Angerer S (1990) J Med Chem 33:153–160
Polossek T, Ambros R, von Angerer S, Brandl G, Mannschreck A, von Angerer E (1992) J Med Chem 35:3537–3547
Faust R, Garratt PJ, Jones R, Yeh LK (2000) J Med Chem 43:1050–1061
Kraus GA, Gupta V, Kohut M, Singh N (2009) Bio Org Med Chem Lett 19:5539–5542
Zhao J, Zhang Y, Cheng K (2008) J Org Chem 73:7428–7431
Yang S-D, Sun C-L, Fang Z, Li B-J, Li Y-Z, Shi Z-J (2008) Angew Chem Int Ed 47:1473–1476
Liang Z, Yao B, Zhang Y (2010) Org Lett 12:3185–3187
Cornella J, Lu P, Larrosa I (2009) Org Lett 11:5506–5509
Vogler T, Studer A (2008) Org Lett 10:129–131
Ban I, Sudo T, Taniguchi T, Itami K (2008) Org Lett 10:3607–3609
Hachiya H, Hirano K, Satoh T, Miura M (2010) Angew Chem Int Ed 49:2202–2205
**e K, Yang Z, Zhou X, Li X, Wang S, Tan Z, An X, Guo C-C (2010) Org Lett 12:1564–1567
Stuart DR, Fagnou K (2007) Science 316:1172–1175
** P, Yang F, Qin S, Zhao D, Lan J, Gao G, Hu C, You J (2010) J Am Chem Soc 132:1822–1824
Kita Y, Morimoto K, Ito M, Ogawa C, Goto A, Dohi T (2009) J Am Chem Soc 131:1668–1669
Dwight TA, Rue NR, Charyk D, Josselyn R, DeBoef B (2007) Org Lett 9:3137–3139
Suzuki A (2005) Chem Commun 38:4759–4763
Nicolaou KC, Bulger PG, Sarlah D (2005) Angew Chem Int Ed 44:4442–4489
Miura M (2004) Angew Chem Int Ed 43:2201–2203
Stanforth SP (1998) Tetrahedron 54:263–303
Art SP, Satoh T, Kawamura Y, Miura M, Nomura M (1998) Bull Chem Soc Jpn 71:467–473
Rieth RD, Mankad NP, Calimano E, Sadighi JP (2004) Org Lett 63:981–3983
Chiong HA, Daugulis O (2007) Org Lett 9:1449–1451
Lebrasseur N, Larrosa I (2008) J Am Chem Soc 130:2926–2927
Lane BS, Sames D (2004) Org Lett 6:2897–2900
Deprez NR, Kalyani D, Krause A, Sanford MS (2006) J Am Chem Soc 128:4972–4973
Gryko DT, Vakuliuk O, Gryko D, Koszarna B (2009) J Org Chem 74:9517–9520
Ca ND, Maestri G, Catellani M (2009) Chem. Eur. J 15:7850–7853
Gracia S, Cazorla C, Metay E, Rostaing P, Lemaire M (2009) J Org Chem 74:3160–3163
Battace A, Lemhadri M, Zair T, Doucet H, Santelli M (2007) Organometallics 26:472–474
Yang L, Zhao L, Li C-J (2010) Chem Comm 46:4184–4186
Sauermann N, González MJ, Ackermann L (2015) Org Lett 17:5316–5319
Nadres ET, Lazareva A, Daugulis O (2011) J Org Chem 76:471–483
Tolnai GL, Ganss S, Brand JP, Waser J (2013) Org Lett 15:112–115
**e F, Qi Z, Yu S, Li X (2014) J Am Chem Soc 136:4780–4787
Zhang ZZ, Liu B, Wang CY, Shi BF (2015) Org Lett 17:4094–4097
Li T, Wang Z, Qin WB, Wen TB (2016) ChemCatChem 8:2146–2154
Ruan Z, Sauermann N, Manoni E, Ackermann L (2017) Angew Chem Int Ed 56:1–6
Wu W, Fang S, Jiang G, Li M, Jiang H (2019) Org. Chem. Front 6:2200–2204
Khake SM, Soni V, Gonnade RG, Punji B (2017) Chem Eur J 23:2907–2916
Jumn MA, Donia B, Piet WNM, Van L, Baruno C (2020) Chem Rev 120:1042–1084
Yuanbiao H, Zu** L, Rong C (2011) Chem Eur J 17:12706–12712
Liang W, Wen-bin Y, Chun C (2011) Chem Commun 47:806–808
Joel M, Anuja N, Cheuk-Wai T, Jan-E B, Berit O (2014) Chem Eur J 20:13531–13535
Linlin D, Rao F, Bingsen Z, Wen S, Shangjun C, Ying W (2016) ACS Catal 6:1062–1074
Christophe VD, Rong Y, Walter R, Dean FT, Gabor AS (2017) J Am Chem Soc 139:18084–18092
Yuan BH, Qiang W, Jun L, Xusheng W, Rong C (2016) J Am Chem Soc 138:10104
Yuan BH, Min S, Xusheng W, ** H, Rui** C, Zu JL, Rong C (2016) J Catal 333:1–7
Wang L, Yi WB, Cai C (2011) Chem Commun 47:806–808
Huang Y, Ma T, Huang P, Wu D, Lin Z, Cao R (2013) ChemCatChem 5:1877–1883
Zhang L, Li P, Liu C, Yang J, Wanga M, Wang L (2014) Catal Sci Technol 4:1979–1988
Huang Y, Lin Z, Cao R (2011) Chem Eur J 17:12706–12712
Malmgren J, Nagendiran A, Tai CW, Backvall JE, Olofsson B (2014) Chem Eur J 20:1–6
Duan L, Fu R, Zhang B, Shi W, Chen S, Wan Y (2016) ACS Catal 6:1062–1074
Lu GP, Cai C (2012) Synlett 23:2992–2966
Xu Z, Xu Y, Lu H, Yang T, Lin X, Shao L, Ren F (2015) Tetrahedron 71:2616–2621
Joucla L, Batail N, Djakovitcha L (2010) Adv Synth Catal 352:2929–2936
Islam S, Larrosa I (2013) Chem Eur J 19:15093–15096
Moncea O, Poinsot D, Fokin AA, Schreiner PR, Hierso JC (2018) ChemCatChem 10:2915–2922
Kandathil V, Dateer RB, Sasidhar BS, Patil SA, Patil SA (2018) Catal Lett 148:1562–1578
Nasrollahzadeh M, Sajadi SM, Maham M, Ehsani A (2015) RSC Adv 5:2562–2567
Kandathil V, Kempasiddaiaha M, Sasidhar BS, Patil SA (2019) Carbohydr Polym 223:1150
Garai C, Hasan SN, Barai AC, Ghorai S, Panja SK, Bag BG (2019) J Nanostr Chem 8:462
Hegde RV, Ghosh A, Patil SA, Dateer RB (2019) Tetrahedron 75:13077
Vishal K, Fahlman BD, Sasidhar BS, Patil SA, Patil SA (2017) Catal Lett 147:900–918
Das D, Bhutia ZT, Chatterjee A, Banerjee M (2019) J Org Chem 84:10764–10774
Colletto C, Panigrahi A, Casado J-F, Larrosa I (2018) J Am Chem Soc 140:9638–9643
Jayarajan R, Kumar R, Gupta J, Dev G, Kadu P, Chatterjee D, Bahadur D, Maiti D, Maji SK (2019) J Mater Chem A 7:4486–4493
Domingo GC, Paula DM, Ataualpa AC, Braga FM, Antonio ME (2007) J Am Chem Soc 129:6880–6886
Sudha K, Michael B, Stanley EG, Kendra B, Frank B, G, Keith, CE, (2017) Chem Commun 53:7022–7025
Acknowledgements
Author thank to, SERB-DST, Government of India, for the financial support through the research Grant: File Nos. SB/S2/RJN-042/2017 and ECR/2017/002207. Author also thanks to Jain University, India, for financial support.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of interest
There are no conflicts to declare.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Hegde, R.V., Ong, TG., Ambre, R. et al. Regioselective Direct C2 Arylation of Indole, Benzothiophene and Benzofuran: Utilization of Reusable Pd NPs and NHC-Pd@MNPs Catalyst for C–H Activation Reaction. Catal Lett 151, 1397–1405 (2021). https://doi.org/10.1007/s10562-020-03390-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10562-020-03390-x