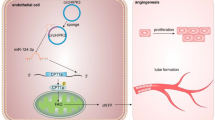
Abstract
Purpose
Intermittent hypoxia (IH), a main characteristic of obstructive sleep apnea (OSA) syndrome, has been known as a dominant cause of OSA-related endothelial dysfunction and hypertension. However, the underlying mechanism still remains unclear. Extracellular vesicles (EVs), small vesicles secreted by various cells, can be absorbed by endothelial cells and then influence vascular function. The aim of this research is to clarify whether and how EVs shedding from red blood cells (RBCs) are involved in IH-induced endothelial dysfunction.
Methods
EVs were extracted by ultracentrifugation. After the identification of property and purity, EVs from IH-exposed RBCs (IH REVs) and normoxia-exposed RBCs (NOR REVs) or from OSA and non-OSA patient RBCs were utilized to treat C57BL/6 mouse aortas or human umbilical vein endothelial cells (HUVECs) for mechanistic exploration.
Results
Functional results demonstrated that REVs from OSA patients dramatically impaired endothelium-dependent relaxations (EDRs). Similarly, in vivo and ex vivo studies showed that IH REVs caused significant endothelial dysfunction compared to control group. Further results presented that IH REVs blocked endothelial nitric oxide synthase (eNOS) phosphorylation through inhibiting PI3K/Akt pathway and enhanced endothelin-1 (ET-1) expression through activating Erk1/2 pathway in endothelial cells. Meanwhile, endothelial dysfunction caused by IH REVs was reversed by Akt activator SC79 as well as Erk kinase inhibitor PD98059, suggesting that PI3K/Akt/eNOS and Erk1/2/ET-1 pathways were implicated in IH REV-induced impaired EDRs.
Conclusions
This study reveals a novel role of REVs in endothelial dysfunction under IH and dissects the relevant mechanism involved in this process, which will help to establish a comprehensive understanding of OSA or IH-related endothelial dysfunction from a new scope.







Similar content being viewed by others
References
Levy P, Kohler M, McNicholas WT, Barbe F, McEvoy RD, Somers VK, et al. Obstructive sleep apnoea syndrome. Nat Rev Disease Primers. 2015;1:15015.
Benjafield AV, Ayas NT, Eastwood PR, Heinzer R, Ip MSM, Morrell MJ, et al. Estimation of the global prevalence and burden of obstructive sleep apnoea: a literature-based analysis. Lancet Respir Med. 2019;7(8):687–98.
Baltzis D, Bakker JP, Patel SR, Veves A. Obstructive sleep apnea and vascular diseases. Compr Physiol. 2016;6(3):1519–28.
Cai A, Wang L, Zhou Y. Hypertension and obstructive sleep apnea. Hypertens Res. 2016;39(6):391–5.
Hou H, Zhao Y, Yu W, Dong H, Xue X, Ding J, et al. Association of obstructive sleep apnea with hypertension: a systematic review and meta-analysis. J Glob Health. 2018;8(1):010405.
Cao MT, Sternbach JM, Guilleminault C. Continuous positive airway pressure therapy in obstuctive sleep apnea: benefits and alternatives. Expert Rev Respir Med. 2017;11(4):259–72.
Schwarz EI, Schlatzer C, Rossi VA, Stradling JR, Kohler M. Effect of CPAP withdrawal on BP in OSA: data from three randomized controlled trials. Chest. 2016;150(6):1202–10.
Lei Q, Lv Y, Li K, Ma L, Du G, **ang Y, et al. Effects of continuous positive airway pressure on blood pressure in patients with resistant hypertension and obstructive sleep apnea: a systematic review and meta-analysis of six randomized controlled trials. J Bras Pneumol. 2017;43(5):373–9.
Dewan NA, Nieto FJ, Somers VK. Intermittent hypoxemia and OSA: implications for comorbidities. Chest. 2015;147(1):266–74.
Atkeson A, Yeh SY, Malhotra A, Jelic S. Endothelial function in obstructive sleep apnea. Prog Cardiovasc Dis. 2009;51(5):351–62.
Chan KCC, Au CT, Chook P, Lee DLY, Lam HS, Wing YK, et al. Endothelial function in children with OSA and the effects of adenotonsillectomy. Chest. 2015;147(1):132–9.
Farooqui FA, Sharma SK, Kumar A, Soneja M, Mani K, Radhakrishnan R, et al. Endothelial function and carotid intima media thickness in obstructive sleep apnea without comorbidity. Sleep Breath. 2017;21(1):69–76.
Song X, Sun Z, Chen G, Shang P, You G, Zhao J, et al. Matrix stiffening induces endothelial dysfunction via the TRPV4/microRNA-6740/endothelin-1 mechanotransduction pathway. Acta Biomater. 2019;100:52–60.
Sharma P, Dong Y, Somers VK, Peterson TE, Zhang Y, Wang S, et al. Intermittent hypoxia regulates vasoactive molecules and alters insulin-signaling in vascular endothelial cells. Sci Rep. 2018;8(1):14110.
Krause BJ, Del Rio R, Moya EA, Marquez-Gutierrez M, Casanello P, Iturriaga R. Arginase-endothelial nitric oxide synthase imbalance contributes to endothelial dysfunction during chronic intermittent hypoxia. J Hypertens. 2015;33(3):515–24.
Kanagy NLWB, Nelin LD. Role of endothelin in intermittent hypoxia-induced hypertension. Hypertension. 2001;37:511–5.
Gjorup PH, Sadauskiene L, Wessels J, Nyvad O, Strunge B, Pedersen EB. Abnormally increased endothelin-1 in plasma during the night in obstructive sleep apnea: relation to blood pressure and severity of disease. Am J Hypertens. 2007;20(1):44–52.
Jelic S, Lederer DJ, Adams T, Padeletti M, Colombo PC, Factor PH, et al. Vascular inflammation in obesity and sleep apnea. Circulation. 2010;121(8):1014–21.
van Niel GDAG, Raposo G. Shedding light on the cell biology of extracellular vesicles. Nat Rev Mol Cell Biol. 2018;19(4):213–28.
Farooqi AA, Desai NN, Qureshi MZ, Librelotto DRN, Gasparri ML, Bishayee A, et al. Exosome biogenesis, bioactivities and functions as new delivery systems of natural compounds. Biotechnol Adv. 2018;36(1):328–34.
Hu Y, Rao SS, Wang ZX, Cao J, Tan YJ, Luo J, et al. Exosomes from human umbilical cord blood accelerate cutaneous wound healing through miR-21-3p-mediated promotion of angiogenesis and fibroblast function. Theranostics. 2018;8(1):169–84.
Fang JH, Zhang ZJ, Shang LR, Luo YW, Lin YF, Yuan Y, et al. Hepatoma cell-secreted exosomal microRNA-103 increases vascular permeability and promotes metastasis by targeting junction proteins. Hepatology. 2018;68(4):1459–75.
Zhang H, Liu J, Qu D, Wang L, Wong CM, Lau CW, et al. Serum exosomes mediate delivery of arginase 1 as a novel mechanism for endothelial dysfunction in diabetes. Proc Natl Acad Sci U S A. 2018;115(29):E6927–E36.
Sender R, Fuchs S, Milo R. Revised estimates for the number of human and bacteria cells in the body. PLoS Biol. 2016;14(8):e1002533.
Jiang L, Gu Y, Du Y, Liu J. Exosomes: diagnostic biomarkers and therapeutic delivery vehicles for Cancer. Mol Pharm. 2019;16(8):3333–49.
Kang J, Li Y, Hu K, Lu W, Zhou X, Yu S, et al. Chronic intermittent hypoxia versus continuous hypoxia: same effects on hemorheology? Clin Hemorheol Microcirc. 2016;63(3):245–55.
Guedes AF, Moreira C, Nogueira JB, Santos NC, Carvalho FA. Fibrinogen-erythrocyte binding and hemorheology measurements in the assessment of essential arterial hypertension patients. Nanoscale. 2019;11(6):2757–66.
Sloop G, Holsworth RE, Weidman JJ, St Cyr JA. The role of chronic hyperviscosity in vascular disease. Ther Adv Cardiovasc Dis. 2015;9(1):19–25.
Zhou Z, Mahdi A, Tratsiakovich Y, Zahorán S, Kövamees O, Nordin F, et al. Erythrocytes from patients with type 2 diabetes induce endothelial dysfunction via arginase I. J Am Coll Cardiol. 2018;72(7):769–80.
Zeng Z, Li Y, Pan Y, Lan X, Song F, Sun J, et al. Cancer-derived exosomal miR-25-3p promotes pre-metastatic niche formation by inducing vascular permeability and angiogenesis. Nat Commun. 2018;9(1):5395.
**g ZT, Liu W, Xue CR, Wu SX, Chen WN, Lin XJ, et al. AKT activator SC79 protects hepatocytes from TNF-α-mediated apoptosis and alleviates d-Gal/LPS-induced liver injury. Am J Physiol Gastrointest Liver Physiol. 2019;316(3):G387–G96.
Potenza MA, Marasciulo FL, Chieppa DM, Brigiani GS, Formoso G, Quon MJ, et al. Insulin resistance in spontaneously hypertensive rats is associated with endothelial dysfunction characterized by imbalance between NO and ET-1 production. Am J Physiol Heart Circ Physiol. 2005;289(2):H813–22.
Ren R, Sun H, Ma C, Liu J, Wang H. Colon cancer cells secrete exosomes to promote self-proliferation by shortening mitosis duration and activation of STAT3 in a hypoxic environment. Cell Biosci. 2019;9:62.
Lee JH, Parveen A, Do MH, Lim Y, Shim SH, Kim SY. Lespedeza cuneata protects the endothelial dysfunction via eNOS phosphorylation of PI3K/Akt signaling pathway in HUVECs. Phytomedicine. 2018;48:1–9.
Wang Y, Cheng KK, Lam KS, Wu D, Wang Y, Huang Y, et al. APPL1 counteracts obesity-induced vascular insulin resistance and endothelial dysfunction by modulating the endothelial production of nitric oxide and endothelin-1 in mice. Diabetes. 2011;60(11):3044–54.
Luan Q, Pan L, He D, Gong X, Zhou H. SC79, the AKT activator protects cerebral ischemia in a rat model of ischemia/reperfusion injury. Med Sci Monit. 2018;24:5391–7.
Durgan DJ, Crossland RF, Lloyd EE, Phillips SC, Bryan RM. Increased cerebrovascular sensitivity to endothelin-1 in a rat model of obstructive sleep apnea: a role for endothelin receptor B. J Cereb Blood Flow Metab. 2015;35(3):402–11.
Torres G, Sanchez-de-la-Torre M, Barbe F. Relationship between OSA and hypertension. Chest. 2015;148(3):824–32.
de Vries GE, Wijkstra PJ, Houwerzijl EJ, Kerstjens HAM, Hoekema A. Cardiovascular effects of oral appliance therapy in obstructive sleep apnea: a systematic review and meta-analysis. Sleep Med Rev. 2018;40:55–68.
Huber HJ, Holvoet P. Exosomes: emerging roles in communication between blood cells and vascular tissues during atherosclerosis. Curr Opin Lipidol. 2015;26(5):412–9.
Trzepizur W, Martinez MC, Priou P, Andriantsitohaina R, Gagnadoux F. Microparticles and vascular dysfunction in obstructive sleep apnoea. Eur Respir J. 2014;44(1):207–16.
Khalyfa A, Gozal D, Chan WC, Andrade J, Prasad B. Circulating plasma exosomes in obstructive sleep apnoea and reverse dip** blood pressure. Eur Respir J. 2020;55(1):1901072.
Khalyfa A, Zhang C, Khalyfa AA, Foster GE, Beaudin AE, Andrade J, et al. Effect on intermittent hypoxia on plasma exosomal micro RNA signature and endothelial function in healthy adults. Sleep. 2016;39(12):2077–90.
Khalyfa A, Kheirandish-Gozal L, Khalyfa AA, Philby MF, Alonso-Alvarez ML, Mohammadi M, et al. Circulating plasma extracellular microvesicle microRNA cargo and endothelial dysfunction in children with obstructive sleep apnea. Am J Respir Crit Care Med. 2016;194(9):1116–26.
Madeddu P, Lobysheva II, Biller P, Gallez B, Beauloye C, Balligand J-L. Nitrosylated hemoglobin levels in human venous erythrocytes correlate with vascular endothelial function measured by digital reactive hyperemia. PLoS One. 2013;8(10):e76457.
Li KY, Zheng L, Wang Q, Hu YW. Characteristics of erythrocyte-derived microvesicles and its relation with atherosclerosis. Atherosclerosis. 2016;255:140–4.
Thompson CB. Into thin air: how we sense and respond to hypoxia. Cell. 2016;167(1):9–11.
Kim JA, Montagnani M, Koh KK, Quon MJ. Reciprocal relationships between insulin resistance and endothelial dysfunction: molecular and pathophysiological mechanisms. Circulation. 2006;113(15):1888–904.
Kheirandish-Gozal L, Khalyfa A, Gozal D, Bhattacharjee R, Wang Y. Endothelial dysfunction in children with obstructive sleep apnea is associated with epigenetic changes in the eNOS gene. Chest. 2013;143(4):971–7.
Varadharaj S, Porter K, Pleister A, Wannemacher J, Sow A, Jarjoura D, et al. Endothelial nitric oxide synthase uncoupling: a novel pathway in OSA induced vascular endothelial dysfunction. Respir Physiol Neurobiol. 2015;207:40–7.
Belaidi E, Joyeux-Faure M, Ribuot C, Launois SH, Levy P, Godin-Ribuot D. Major role for hypoxia inducible factor-1 and the endothelin system in promoting myocardial infarction and hypertension in an animal model of obstructive sleep apnea. J Am Coll Cardiol. 2009;53(15):1309–17.
Chang AL, Kim Y, Seitz AP, Schuster RM, Lentsch AB, Pritts TA. Erythrocyte-derived microparticles activate pulmonary endothelial cells in a murine model of transfusion. Shock. 2017;47(5):632–7.
Kim Y, Goodman MD, Jung AD, Abplanalp WA, Schuster RM, Caldwell CC, et al. Microparticles from aged packed red blood cell units stimulate pulmonary microthrombus formation via P-selectin. Thromb Res. 2020;185:160–6.
Emamian M, Hasanian SM, Tayefi M, Bijari M, Movahedian Far F, Shafiee M, et al. Association of hematocrit with blood pressure and hypertension. J Clin Lab Anal. 2017;31(6):e22124.
Diederich L, Suvorava T, Sansone R, Keller TCS, Barbarino F, Sutton TR, et al. On the effects of reactive oxygen species and nitric oxide on red blood cell deformability. Front Physiol. 2018;9:332.
Acknowledgments
We are grateful to Ms. Yuqing Yang (Bei**g ECHO Biotech. Co., Ltd. China) for transmission electron microscopy assistance.
Funding
This study was supported by Bei**g Natural Science Foundation under Grant No. 7192031, by National Natural Science Foundation of China under Grant No. 81870335, and by Bei**g Key Laboratory of Upper Airway Dysfunction and Related Cardiovascular Diseases under Grant No. BZ0377.
Author information
Authors and Affiliations
Contributions
Lu Peng carried out main experiments, designed some experiments, and wrote the manuscript. Yu Li and **nwei Li helped to do the animal experiments. Yunhui Du, Linyi Li, and Chaowei Hu helped to analyze the data. **g Zhang and Yanwen Qin helped with sample collection. Huina Zhang and Yongxiang Wei conceived the experiments, edited the manuscript, and financed the study.
Corresponding authors
Ethics declarations
Conflict of Interest
The authors declare that they have no conflict of interest.
Ethical Approval
All procedures involving human participants were authorized by the Medical Ethics Committee of Bei**g Anzhen Hospital and adhered to the Declaration of Helsinki and its later amendments or comparable ethical standards. All procedures involving animals were approved by Capital Medical University Animal Experimentation Ethics Committee and carried out in accordance with the National Institutes of Health Guide for the Care and Use of Laboratory Animals.
Informed Consent
Written informed consent was obtained from all participants enrolled in this study.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
ESM 1
(DOCX 424 kb)
Rights and permissions
About this article
Cite this article
Peng, L., Li, Y., Li, X. et al. Extracellular Vesicles Derived from Intermittent Hypoxia–Treated Red Blood Cells Impair Endothelial Function Through Regulating eNOS Phosphorylation and ET-1 Expression. Cardiovasc Drugs Ther 35, 901–913 (2021). https://doi.org/10.1007/s10557-020-07117-3
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10557-020-07117-3