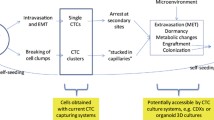
Abstract
Metastasis remains the principal trigger for relapse and mortality across diverse cancer types. Circulating tumor cells (CTCs), which originate from the primary tumor or its metastatic sites, traverse the vascular system, serving as precursors in cancer recurrence and metastasis. Nevertheless, before CTCs can establish themselves in the distant parenchyma, they must overcome significant challenges present within the circulatory system, including hydrodynamic shear stress (HSS), oxidative damage, anoikis, and immune surveillance. Recently, there has been a growing body of compelling evidence suggesting that a specific subset of CTCs can persist within the bloodstream, but the precise mechanisms of their survival remain largely elusive. This review aims to present an outline of the survival challenges encountered by CTCs and to summarize the recent advancements in understanding the underlying survival mechanisms, suggesting their implications for cancer treatment.



Similar content being viewed by others
Data availability
Not applicable.
References
Fares, J., et al. (2020). Molecular principles of metastasis: A hallmark of cancer revisited. Signal Transduction and Targeted Therapy, 5(1), 28.
Ashworth, T. R. (1869). A case of cancer in which cells similar to those in the tumors were seen in the blood after death. Australas Med J, 14, 146–149.
Taddei, M. L., et al. (2012). Anoikis: An emerging hallmark in health and diseases. The Journal of Pathology, 226(2), 380–393.
Kanwar, N., et al. (2015). Identification of genomic signatures in circulating tumor cells from breast cancer. International Journal of Cancer, 137(2), 332–344.
Chang, T. Y. (2021). Comparison of genetic profiling between primary tumor and circulating Tumor cells captured by Microfluidics in Epithelial Ovarian Cancer: Tumor Heterogeneity or Allele Dropout? Diagnostics (Basel) 11 (6).
Zou, L., et al. (2020). Genome–wide copy number analysis of circulating tumor cells in breast cancer patients with liver metastasis. Oncology Reports, 44(3), 1075–1093.
Chiu, C. G., et al. (2014). Genome-wide characterization of circulating tumor cells identifies novel prognostic genomic alterations in systemic melanoma metastasis. Clinical Chemistry, 60(6), 873–885.
Steinert, G., et al. (2014). Immune escape and survival mechanisms in circulating Tumor cells of Colorectal Cancer. Cancer Research, 74(6), 1694–1704.
Müller, V., et al. (2005). Circulating tumor cells in breast cancer: Correlation to bone marrow micrometastases, heterogeneous response to systemic therapy and low proliferative activity. Clinical Cancer Research, 11(10), 3678–3685.
Gires, O. A. O. Expression and function of epithelial cell adhesion molecule EpCAM: Where are we after 40 years? (1573–7233 (Electronic)).
Thiery, J. P. (2003). Epithelial–mesenchymal transitions in development and pathologies. Current Opinion in Cell Biology, 15(6), 740–746.
Huang, Y., et al. (2022). The molecular mechanisms and therapeutic strategies of EMT in tumor progression and metastasis. Journal of Hematology & Oncology, 15(1), 129.
Huaman, J. (2019). Fibronectin regulation of integrin B1 and SLUG in circulating Tumor cells. Cells 8 (6).
Lecharpentier, A., et al. (2011). Detection of circulating tumour cells with a hybrid (epithelial/mesenchymal) phenotype in patients with metastatic non-small cell lung cancer. British Journal of Cancer, 105(9), 1338–1341.
Yu, M., et al. (2013). Circulating breast tumor cells exhibit dynamic changes in epithelial and mesenchymal composition. Science, 339(6119), 580–584.
Armstrong, A. J., et al. (2011). Circulating tumor cells from patients with advanced prostate and breast cancer display both epithelial and mesenchymal markers. Molecular Cancer Research, 9(8), 997–1007.
Kallergi, G., et al. (2011). Epithelial to mesenchymal transition markers expressed in circulating tumour cells of early and metastatic breast cancer patients. Breast Cancer Research, 13(3), R59.
Liu, X., et al. (2019). Epithelial-type systemic breast carcinoma cells with a restricted mesenchymal transition are a major source of metastasis. Science Advances, 5(6), eaav4275.
Chaffer, C. L., et al. (2006). Mesenchymal-to-epithelial transition facilitates bladder cancer metastasis: Role of fibroblast growth factor receptor-2. Cancer Research, 66(23), 11271–11278.
Yamamoto, M., et al. (2017). Intratumoral bidirectional transitions between epithelial and mesenchymal cells in triple-negative breast cancer. Cancer Science, 108(6), 1210–1222.
Yang, M. H., et al. (2015). Circulating cancer stem cells: The importance to select. Chinese Journal of Cancer Research, 27(5), 437–449.
Theodoropoulos, P. A., et al. (2010). Circulating tumor cells with a putative stem cell phenotype in peripheral blood of patients with breast cancer. Cancer Letters, 288(1), 99–106.
Wan, S., et al. (2019). New Labyrinth Microfluidic device detects circulating Tumor cells expressing Cancer stem cell marker and circulating Tumor Microemboli in Hepatocellular Carcinoma. Scientific Reports, 9(1), 18575.
Morel, A. P. (2008). Generation of breast cancer stem cells through epithelial-mesenchymal transition. PLoS One 3 (8), e2888.
Mani, S. A., et al. (2008). The epithelial-mesenchymal transition generates cells with properties of stem cells. Cell, 133(4), 704–715.
Papadaki, M. A., et al. (2019). Circulating Tumor cells with stemness and Epithelial-To-Mesenchymal Transition Features Are Chemoresistant and Predictive of poor outcome in metastatic breast Cancer. Molecular Cancer Therapeutics, 18(2), 437–447.
Follain, G., et al. (2020). Fluids and their mechanics in tumour transit: Sha** metastasis. Nature Reviews Cancer, 20(2), 107–124.
Headley, M. B., et al. (2016). Visualization of immediate immune responses to pioneer metastatic cells in the lung. Nature, 531(7595), 513–517.
Chang, S. F., et al. (2008). Tumor cell cycle arrest induced by shear stress: Roles of integrins and smad. Proc Natl Acad Sci U S A, 105(10), 3927–3932.
Fu, A., et al. (2016). High expression of MnSOD promotes survival of circulating breast cancer cells and increases their resistance to doxorubicin. Oncotarget, 7(31), 50239–50257.
Regmi, S., et al. (2017). High Shear stresses under Exercise Condition destroy circulating Tumor cells in a Microfluidic System. Scientific Reports, 7, 39975.
Kienast, Y., et al. (2010). Real-time imaging reveals the single steps of brain metastasis formation. Nature Medicine, 16(1), 116–122.
Marrella, A. (2021). High blood flow shear stress values are associated with circulating tumor cells cluster disaggregation in a multi-channel microfluidic device. PLoS One 16 (1), e0245536.
Jasuja, H., et al. (2023). Interstitial fluid flow contributes to prostate cancer invasion and migration to bone; study conducted using a novel horizontal flow bioreactor. Biofabrication, 15(2), 025017.
Kim, O. H., et al. (2022). Fluid shear stress facilitates prostate cancer metastasis through Piezo1-Src-YAP axis. Life Sciences, 308, 120936.
Piskounova, E., et al. (2015). Oxidative stress inhibits distant metastasis by human melanoma cells. Nature, 527(7577), 186–191.
Schafer, Z. T., et al. (2009). Antioxidant and oncogene rescue of metabolic defects caused by loss of matrix attachment. Nature, 461(7260), 109–113.
Redza-Dutordoir, M., & Averill-Bates, D. A. (2016). Activation of apoptosis signalling pathways by reactive oxygen species. Biochimica Et Biophysica Acta, 1863(12), 2977–2992.
Que, Z., et al. (2019). **gfukang induces anti-cancer activity through oxidative stress-mediated DNA damage in circulating human lung cancer cells. Bmc Complementary and Alternative Medicine, 19(1), 204.
Ubellacker, J. M., et al. (2020). Lymph protects metastasizing melanoma cells from ferroptosis. Nature, 585(7823), 113–118.
Paoli, P., et al. (2013). Anoikis molecular pathways and its role in cancer progression. Biochimica Et Biophysica Acta, 1833(12), 3481–3498.
Khan, S. U., et al. (2022). Understanding the cell survival mechanism of anoikis-resistant cancer cells during different steps of metastasis. Clinical & Experimental Metastasis, 39(5), 715–726.
Mohme, M., et al. (2017). Circulating and disseminated tumour cells - mechanisms of immune surveillance and escape. Nature Reviews. Clinical Oncology, 14(3), 155–167.
Vivier, E., et al. (2018). Innate lymphoid cells: 10 years on. Cell, 174(5), 1054–1066.
Cornel, A. M. (2020). MHC Class I Downregulation in Cancer: Underlying Mechanisms and Potential Targets for Cancer Immunotherapy. Cancers (Basel) 12 (7).
Kärre, K., et al. (1986). Selective rejection of H–2-deficient lymphoma variants suggests alternative immune defence strategy. Nature, 319(6055), 675–678.
Ljunggren, H. G., & Kärre, K. (1985). Host resistance directed selectively against H-2-deficient lymphoma variants. Analysis of the mechanism. Journal of Experimental Medicine, 162(6), 1745–1759.
Brodbeck, T., et al. (2014). Perforin-dependent direct cytotoxicity in natural killer cells induces considerable knockdown of spontaneous lung metastases and computer modelling-proven tumor cell dormancy in a HT29 human colon cancer xenograft mouse model. Molecular Cancer, 13, 244.
Gül, N., et al. (2014). Macrophages eliminate circulating tumor cells after monoclonal antibody therapy. J Clin Invest, 124(2), 812–823.
Xue, D., et al. (2018). Role of regulatory T cells and CD8(+) T lymphocytes in the dissemination of circulating tumor cells in primary invasive breast cancer. Oncol Lett, 16(3), 3045–3053.
Liu, J., et al. (2019). Increased stromal infiltrating lymphocytes are Associated with circulating Tumor cells and metastatic relapse in breast Cancer patients after Neoadjuvant Chemotherapy. Cancer Manag Res, 11, 10791–10800.
Smolkova, B., et al. (2020). Increased stromal infiltrating lymphocytes are Associated with the risk of Disease Progression in Mesenchymal circulating Tumor cell-positive primary breast Cancer patients. International Journal of Molecular Sciences, 21, 24.
Liu, X., et al. (2023). Immune checkpoint HLA-E:CD94-NKG2A mediates evasion of circulating tumor cells from NK cell surveillance. Cancer Cell, 41(2), 272–287. e9.
Lo, H. C., et al. (2020). Resistance to natural killer cell immunosurveillance confers a selective advantage to polyclonal metastasis. Nat Cancer, 1(7), 709–722.
Hope, J. M. (2021). Circulating prostate cancer cells have differential resistance to fluid shear stress-induced cell death. Journal of Cell Science 134 (4).
Barnes, J. M. (2012). Resistance to fluid shear stress is a conserved biophysical property of malignant cells. PLoS One 7 (12), e50973.
Moose, D. L., et al. (2020). Cancer cells resist Mechanical Destruction in circulation via RhoA/Actomyosin-Dependent mechano-adaptation. Cell Rep, 30(11), 3864–3874e6.
Choi, H. Y., et al. (2019). Hydrodynamic shear stress promotes epithelial-mesenchymal transition by downregulating ERK and GSK3β activities. Breast Cancer Research, 21(1), 6.
Chen, X., et al. (2022). YAP1 activation promotes epithelial-mesenchymal transition and cell survival of renal cell carcinoma cells under shear stress. Carcinogenesis, 43(4), 301–310.
Mitchell, M. J., et al. (2015). Lamin A/C deficiency reduces circulating tumor cell resistance to fluid shear stress. American Journal of Physiology. Cell Physiology, 309(11), C736–C746.
Xu, Z. (2022). Fluid shear stress regulates the survival of circulating tumor cells via nuclear expansion. Journal of Cell Science 135 (10).
Maeshiro, M., et al. (2021). Colonization of distant organs by tumor cells generating circulating homotypic clusters adaptive to fluid shear stress. Scientific Reports, 11(1), 6150.
Ortiz-Otero, N., et al. (2020). Cancer associated fibroblasts confer shear resistance to circulating tumor cells during prostate cancer metastatic progression. Oncotarget, 11(12), 1037–1050.
Hagihara, T., et al. (2019). Hydrodynamic stress stimulates growth of cell clusters via the ANXA1/PI3K/AKT axis in colorectal cancer. Scientific Reports, 9(1), 20027.
Osmulski, P. A., et al. (2021). Contacts with macrophages promote an aggressive Nanomechanical phenotype of circulating Tumor cells in prostate Cancer. Cancer Research, 81(15), 4110–4123.
Huang, Q., et al. (2020). Shear stress activates ATOH8 via autocrine VEGF promoting glycolysis dependent-survival of colorectal cancer cells in the circulation. Journal of Experimental & Clinical Cancer Research : Cr, 39(1), 25.
Donato, C., et al. (2020). Hypoxia triggers the intravasation of clustered circulating Tumor cells. Cell Rep, 32(10), 108105.
Labuschagne, C. F., et al. (2019). Cell clustering promotes a metabolic switch that supports metastatic colonization. Cell Metab, 30(4), 720–734e5.
Hong, X., et al. (2021). The Lipogenic Regulator SREBP2 induces transferrin in circulating Melanoma cells and suppresses ferroptosis. Cancer Discovery, 11(3), 678–695.
Sun, B., et al. (2017). Midkine promotes hepatocellular carcinoma metastasis by elevating anoikis resistance of circulating tumor cells. Oncotarget, 8(20), 32523–32535.
Malin, D., et al. (2015). ERK-regulated alphab-crystallin induction by matrix detachment inhibits anoikis and promotes lung metastasis in vivo. Oncogene, 34(45), 5626–5634.
Haemmerle, M., et al. (2017). Platelets reduce anoikis and promote metastasis by activating YAP1 signaling. Nature Communications, 8(1), 310.
**, L., et al. (2015). Glutamate dehydrogenase 1 signals through Antioxidant Glutathione Peroxidase 1 to regulate Redox Homeostasis and Tumor Growth. Cancer Cell, 27(2), 257–270.
**, L., et al. (2018). The PLAG1-GDH1 Axis promotes Anoikis Resistance and Tumor Metastasis through CamKK2-AMPK Signaling in LKB1-Deficient Lung Cancer. Molecular Cell, 69(1), 87–99e7.
Chen, X., et al. (2022). Shear stress enhances anoikis resistance of cancer cells through ROS and NO suppressed degeneration of Caveolin-1. Free Radical Biology and Medicine, 193(Pt 1), 95–107.
Kim, H., et al. (2017). Regulation of anoikis resistance by NADPH oxidase 4 and epidermal growth factor receptor. British Journal of Cancer, 116(3), 370–381.
Du, S., et al. (2018). NADPH oxidase 4 regulates anoikis resistance of gastric cancer cells through the generation of reactive oxygen species and the induction of EGFR. Cell Death and Disease, 9(10), 948.
Tian, T., et al. (2022). CPT1A promotes anoikis resistance in esophageal squamous cell carcinoma via redox homeostasis. Redox Biology, 58, 102544.
Wang, Y. N., et al. (2018). CPT1A-mediated fatty acid oxidation promotes colorectal cancer cell metastasis by inhibiting anoikis. Oncogene, 37(46), 6025–6040.
Sawyer, B. T., et al. (2020). Targeting fatty acid oxidation to promote Anoikis and inhibit ovarian Cancer progression. Molecular Cancer Research, 18(7), 1088–1098.
Koppenol, W. H., et al. (2011). Otto Warburg’s contributions to current concepts of cancer metabolism. Nature Reviews Cancer, 11(5), 325–337.
Stacpoole, P. W. (2017). Therapeutic targeting of the Pyruvate Dehydrogenase Complex/Pyruvate Dehydrogenase Kinase (PDC/PDK) Axis in Cancer. JNCI: Journal of the National Cancer Institute, 109, 11.
Kamarajugadda, S., et al. (2012). Glucose oxidation modulates anoikis and tumor metastasis. Molecular and Cellular Biology, 32(10), 1893–1907.
Kim, J., et al. (2006). HIF-1-mediated expression of pyruvate dehydrogenase kinase: A metabolic switch required for cellular adaptation to hypoxia. Cell Metabolism, 3(3), 177–185.
Maurer, G. D., et al. (2019). Loss of cell-matrix contact increases hypoxia-inducible factor-dependent transcriptional activity in glioma cells. Biochemical and Biophysical Research Communications, 515(1), 77–84.
Zhao, T. (2014). HIF-1-mediated metabolic reprogramming reduces ROS levels and facilitates the metastatic colonization of cancers in lungs. Scientific Reports 4 (1).
Lingwood, D., & Simons, K. (2010). Lipid rafts as a membrane-Organizing Principle. 327 (5961), 46–50.
del Pozo, M. A., et al. (2005). Phospho-caveolin-1 mediates integrin-regulated membrane domain internalization. Nature Cell Biology, 7(9), 901–908.
Li, D. (2023). Cell aggregation activates small GTPase Rac1 and induces CD44 cleavage by maintaining lipid raft integrity. Journal of Biological Chemistry 299 (12).
Han, H. J., et al. (2021). Fibronectin regulates anoikis resistance via cell aggregate formation. Cancer Letters, 508, 59–72.
Peppicelli, S. (2019). Anoikis Resistance as a Further Trait of Acidic-Adapted Melanoma Cells. J Oncol 2019, 8340926.
Corbet, C., et al. (2020). TGFβ2-induced formation of lipid droplets supports acidosis-driven EMT and the metastatic spreading of cancer cells. Nature Communications, 11(1), 454.
Wheatley, S. P., & Altieri, D. C. (2019). Survivin at a glance. Journal of Cell Science, 132, 7.
Végran, F., et al. (2013). Survivin-3B potentiates immune escape in cancer but also inhibits the toxicity of cancer chemotherapy. Cancer Research, 73(17), 5391–5401.
Yie, S. M. (2006). Detection of Survivin-expressing circulating cancer cells in the peripheral blood of breast cancer patients by a RT-PCR ELISA. Clinical & Experimental Metastasis 23 (5–6), 279 – 89.
Yie, S., et al. (2008). Detection of Survivin-expressing circulating Cancer cells (CCCs) in Peripheral blood of patients with gastric and colorectal Cancer reveals high risks of Relapse. Annals of Surgical Oncology, 15(11), 3073–3082.
Yie, S. M., et al. (2009). Clinical significance of detecting survivin-expressing circulating cancer cells in patients with non-small cell lung cancer. Lung Cancer, 63(2), 284–290.
Cao, M., et al. (2009). Detection of survivin-expressing circulating cancer cells in the peripheral blood of patients with esophageal squamous cell carcinoma and its clinical significance. Clinical & Experimental Metastasis, 26(7), 751–758.
Ning, Y., et al. (2015). Cytokeratin-20 and survivin-expressing circulating Tumor cells predict survival in metastatic colorectal Cancer patients by a combined immunomagnetic qRT-PCR Approach. Molecular Cancer Therapeutics, 14(10), 2401–2408.
Lu, J., et al. (2023). Association of survivin positive circulating tumor cell levels with immune escape and prognosis of osteosarcoma. Journal of Cancer Research and Clinical Oncology, 149(15), 13741–13751.
Sun, C., et al. (2018). Regulation and function of the PD-L1 checkpoint. Immunity, 48(3), 434–452.
Mazel, M., et al. (2015). Frequent expression of PD-L1 on circulating breast cancer cells. Molecular Oncology, 9(9), 1773–1782.
Kong, D., et al. (2021). Correlation between PD-L1 expression ON CTCs and prognosis of patients with cancer: A systematic review and meta-analysis. Oncoimmunology, 10(1), 1938476.
Papadaki, M. A. (2020). Clinical Relevance of Immune Checkpoints on Circulating Tumor Cells in Breast Cancer. Cancers (Basel) 12 (2).
Wang, J., et al. (2019). Fibrinogen-like protein 1 is a major Immune Inhibitory ligand of LAG-3. Cell, 176(1–2), 334–347e12.
Yan, Q., et al. (2022). Immune Checkpoint FGL1 expression of circulating Tumor cells is Associated with Poor Survival in Curatively Resected Hepatocellular Carcinoma. Frontiers in Oncology, 12, 810269.
Gruber, I., et al. (2013). Relationship between circulating tumor cells and peripheral T-cells in patients with primary breast cancer. Anticancer Research, 33(5), 2233–2238.
Sun, Y. F., et al. (2021). Dissecting spatial heterogeneity and the immune-evasion mechanism of CTCs by single-cell RNA-seq in hepatocellular carcinoma. Nature Communications, 12(1), 4091.
Palumbo, J. S., et al. (2005). Platelets and fibrin(ogen) increase metastatic potential by impeding natural killer cell-mediated elimination of tumor cells. Blood, 105(1), 178–185.
Placke, T., et al. (2012). Platelet-derived MHC class I confers a pseudonormal phenotype to cancer cells that subverts the antitumor reactivity of natural killer immune cells. Cancer Research, 72(2), 440–448.
Spiegel, A., et al. (2016). Neutrophils suppress intraluminal NK cell-mediated Tumor Cell Clearance and enhance extravasation of disseminated carcinoma cells. Cancer Discovery, 6(6), 630–649.
Gabrilovich, D. I., & Nagaraj, S. (2009). Myeloid-derived suppressor cells as regulators of the immune system. Nature Reviews Immunology, 9(3), 162–174.
Liu, Q., et al. (2016). Myeloid-derived suppressor cells (MDSC) facilitate distant metastasis of malignancies by shielding circulating tumor cells (CTC) from immune surveillance. Medical Hypotheses, 87, 34–39.
Zhou, Q., et al. (2023). Circulating tumor cells PD-L1 expression detection and correlation of therapeutic efficacy of immune checkpoint inhibition in advanced non-small-cell lung cancer. Thorac Cancer, 14(5), 470–478.
Murata, Y., et al. (2018). CD47-signal regulatory protein alpha signaling system and its application to cancer immunotherapy. Cancer Science, 109(8), 2349–2357.
Liu, Y., et al. (2023). Emerging phagocytosis checkpoints in cancer immunotherapy. Signal Transduct Target Ther, 8(1), 104.
Lian, S., et al. (2019). Dual blockage of both PD-L1 and CD47 enhances immunotherapy against circulating tumor cells. Scientific Reports, 9(1), 4532.
Mammadova-Bach, E., et al. (2015). Platelets in cancer. From basic research to therapeutic implications. Hamostaseologie, 35(4), 325–336.
Xu, Y., et al. (2020). Blockade of platelets using tumor-specific NO-Releasing nanoparticles prevents Tumor Metastasis and reverses Tumor Immunosuppression. Acs Nano, 14(8), 9780–9795.
Wang, D., et al. (2022). Role of CD155/TIGIT in Digestive cancers: Promising Cancer Target for Immunotherapy. Frontiers in Oncology, 12, 844260.
Yu, Y., et al. (2022). Engineered drug-loaded cellular membrane nanovesicles for efficient treatment of postsurgical cancer recurrence and metastasis. Science Advances, 8(49), eadd3599.
Wang, H., et al. (2016). NRF2 activation by antioxidant antidiabetic agents accelerates tumor metastasis. Science Translational Medicine, 8(334), 334ra51.
Sayin, V. I., et al. (2014). Antioxidants accelerate lung cancer progression in mice. Science Translational Medicine, 6(221), 221ra15.
Zheng, Y., et al. (2017). Expression of β-globin by cancer cells promotes cell survival during blood-borne dissemination. Nature Communications, 8, 14344.
Wang, Y., et al. (2021). The double-edged roles of ROS in cancer prevention and therapy. Theranostics, 11(10), 4839–4857.
Tasdogan, A., et al. (2021). Redox Regulation in Cancer cells during metastasis. Cancer Discovery, 11(11), 2682–2692.
Regmi, S., et al. (2018). Fluidic shear stress increases the anti-cancer effects of ROS-generating drugs in circulating tumor cells. Breast Cancer Research and Treatment, 172(2), 297–312.
Yang, W. S., et al. (2014). Regulation of Ferroptotic Cancer Cell death by GPX4. Cell, 156(1–2), 317–331.
Que, Z. J., et al. (2021). **fukang regulates integrin/Src pathway and anoikis mediating circulating lung cancer cells migration. Journal of Ethnopharmacology, 267, 113473.
Liu, A., et al. (2021). Silencing ZIC2 abrogates tumorigenesis and anoikis resistance of non-small cell lung cancer cells by inhibiting Src/FAK signaling. Mol Ther Oncolytics, 22, 195–208.
Pang, X. J., et al. (2021). Drug Discovery Targeting Focal Adhesion kinase (FAK) as a Promising Cancer Therapy. Molecules, 26, 14.
Spallarossa, A. (2022). The development of FAK inhibitors: A five-year update. International Journal of Molecular Sciences 23 (12).
Li, J., et al. (2016). Genetic engineering of platelets to neutralize circulating tumor cells. Journal of Controlled Release : Official Journal of the Controlled Release Society, 228, 38–47.
Schell, J. C., et al. (2014). A role for the mitochondrial pyruvate carrier as a Repressor of the Warburg effect and Colon cancer cell growth. Molecular Cell, 56(3), 400–413.
Kim, T. H., et al. (2016). Cancer cells become less deformable and more invasive with activation of beta-adrenergic signaling. Journal of Cell Science, 129(24), 4563–4575.
Sloan, E. K., et al. (2010). The sympathetic nervous system induces a metastatic switch in primary breast cancer. Cancer Research, 70(18), 7042–7052.
Le, C. P., et al. (2016). Chronic stress in mice remodels lymph vasculature to promote tumour cell dissemination. Nature Communications, 7, 10634.
Au, S. H., et al. (2016). Clusters of circulating tumor cells traverse capillary-sized vessels. Proc Natl Acad Sci U S A, 113(18), 4947–4952.
Gkountela, S., et al. (2019). Circulating Tumor Cell Clustering shapes DNA methylation to Enable Metastasis Seeding. Cell, 176(1–2), 98–112e14.
Du, E., et al. (2020). The critical role of the interplays of EphrinB2/EphB4 and VEGF in the induction of angiogenesis. Molecular Biology Reports, 47(6), 4681–4690.
Bowley, T. Y. (2023). Targeting Translation and the Cell Cycle Inversely Affects CTC Metabolism but Not Metastasis. Cancers (Basel) 15 (21).
LeBleu, V. S., et al. (2014). PGC-1α mediates mitochondrial biogenesis and oxidative phosphorylation in cancer cells to promote metastasis. Nature Cell Biology, 16(10), 992–1003.
Elia, I. (2017). Proline metabolism supports metastasis formation and could be inhibited to selectively target metastasizing cancer cells. Nature Communications 8 (1).
Acknowledgements
Figures were created with FigDraw (www.figdraw.com).
Funding
This research was supported by the National Natural Science Foundation of China (82073059, 82103364); the Sichuan Science and Technology Program (2022YFS0204); the Post-doctor Research Project, West China Hospital (2023HXBH127).
Author information
Authors and Affiliations
Contributions
F.B made contributions to the conception and design. S.Z and H.X wrote the manuscript. Y.D and H.H completed the figures. Q.T helped to revise the manuscript. All authors contributed to the article and approved the submitted version.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Competing interests
All authors declare that there are no conflicts of interest regarding the publication of this paper.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Zhou, S., Xu, H., Duan, Y. et al. Survival mechanisms of circulating tumor cells and their implications for cancer treatment. Cancer Metastasis Rev (2024). https://doi.org/10.1007/s10555-024-10178-7
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10555-024-10178-7