Abstract
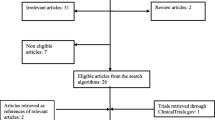
Landmark studies have established trastuzumab in the treatment of HER2-positive breast cancer. The present systematic review and meta-analysis aims to synthesize all available data, so as to evaluate the safety of trastuzumab during pregnancy. This study was performed in accordance with the PRISMA guidelines. All studies that examined the safety of trastuzumab administered during pregnancy, regardless of sample size, were considered eligible. Overall, 17 studies (18 pregnancies; 19 newborns) were included. In 55.6 % of cases, trastuzumab was administered in the metastatic setting. The mean duration of trastuzumab administration was 14.8 weeks. Occurrence of oligohydramnios/anhydramnios (O/A) was the most common (61.1 %) adverse event. 73.3 % of pregnancies exposed to trastuzumab during the second/third trimester were complicated with O/A; the respective rate of pregnancies exposed to trastuzumab exclusively during the first trimester was 0 % (P = 0.043). The mean GA at delivery was 33.8 weeks, and the mean weight of babies at delivery was 2,261 gr. In 52.6 % of cases, a healthy neonate was born. At the long-term evaluation, all children without problems at birth were healthy with a median follow-up of 9 months, while four out of nine children facing troubles at birth were dead within an interval ranging between birth and 5.25 months. All children exposed to trastuzumab in utero exclusively in the first trimester were completely healthy at birth. Trastuzumab should not be administered during pregnancy. However, for women who become accidentally pregnant during trastuzumab administration and wish to continue pregnancy, trastuzumab should be stopped and pregnancy could be allowed to continue.


Similar content being viewed by others
References
Pentheroudakis G, Orecchia R, Hoekstra HJ et al (2010) Cancer, fertility and pregnancy: ESMO clinical practice guidelines for diagnosis, treatment and follow-up. Ann Oncol 21:v266–v273
Mir O, Berveiller P, Ropert S et al (2008) Emerging therapeutic options for breast cancer chemotherapy during pregnancy. Ann Oncol 19:607–613
Howlader N, Noone AM, Krapcho M, et al (2011) SEER cancer statistics review, 1975–2008. National Cancer Institute. Bethesda. http://seer.cancer.gov/csr/1975_2008/, based on November 2010 SEER data submission, posted to the SEER web site
Gianni L, Dafni U, Gelber RD et al (2011) Treatment with trastuzumab for 1 year after adjuvant chemotherapy in patients with HER2-positive early breast cancer: a 4-year follow-up of a randomised controlled trial. Lancet Oncol 12:236–244
Piccart-Gebhart MJ, Procter M, Leyland-Jones B et al (2005) Trastuzumab after adjuvant chemotherapy in HER2-positive breast cancer. N Engl J Med 353:1659–1672
Untch M, Fasching PA, Konecny GE et al (2011) Pathologic complete response after neoadjuvant chemotherapy plus trastuzumab predicts favorable survival in human epidermal growth factor receptor 2-overexpressing breast cancer: results from the TECHNO trial of the AGO and GBG study groups. J Clin Oncol 29:3351–3357
Valero V, Forbes J, Pegram MD et al (2011) Multicenter phase III randomized trial comparing docetaxel and trastuzumab with docetaxel, carboplatin, and trastuzumab as first-line chemotherapy for patients with HER2-gene-amplified metastatic breast cancer (BCIRG 007 study): two highly active therapeutic regimens. J Clin Oncol 29:149–156
Andersson M, Lidbrink E, Bjerre K et al (2011) Phase III randomized study comparing docetaxel plus trastuzumab with vinorelbine plus trastuzumab as first-line therapy of metastatic or locally advanced human epidermal growth factor receptor 2-positive breast cancer: the HERNATA study. J Clin Oncol 29:264–271
NCCN Clinical Practice Guidelines in Oncology (2012) Breast cancer, version 2.2012. www.nccn.com
Azim HA Jr, Azim H, Peccatori FA (2010) Treatment of cancer during pregnancy with monoclonal antibodies: a real challenge. Expert Rev Clin Immunol 6:821–826
Abusief ME, Missmer SA, Ginsburg ES et al (2010) The effects of paclitaxel, dose density, and trastuzumab on treatment-related amenorrhea in premenopausal women with breast cancer. Cancer 116:791–798
Liberati A, Altman DG, Tetzlaff J et al (2009) The PRISMA statement for reporting systematic reviews and meta-analyses of studies that evaluate health care interventions: explanation and elaboration. J Clin Epidemiol 62:e1–e34
Gottschalk I, Berg C, Harbeck N et al (2011) Fetal renal insufficiency following trastuzumab treatment for breast cancer in pregnancy: case report und review of the current literature. Breast Care (Basel) 6:475–478
El-Safadi S, Wuesten O, Muenstedt K (2012) Primary diagnosis of metastatic breast cancer in the third trimester of pregnancy: a case report and review of the literature. J Obstet Gynaecol Res 38:589–592
Mandrawa CL, Stewart J, Fabinyi GC et al (2011) A case study of trastuzumab treatment for metastatic breast cancer in pregnancy: fetal risks and management of cerebral metastases. Aust N Z J Obstet Gynaecol 51:372–376
Goodyer MJ, Ismail JR, O’Reilly SP et al (2009) Safety of trastuzumab (Herceptin) during pregnancy: two case reports. Cases J 2:9329
Beale JM, Tuohy J, McDowell SJ (2009) Herceptin (trastuzumab) therapy in a twin pregnancy with associated oligohydramnios. Am J Obstet Gynecol 201:e13–e14
Weber-Schoendorfer C, Schaefer C (2008) Trastuzumab exposure during pregnancy. Reprod Toxicol 25:390–391; author reply 392
Pant S, Landon MB, Blumenfeld M et al (2008) Treatment of breast cancer with trastuzumab during pregnancy. J Clin Oncol 26:1567–1569
Witzel ID, Müller V, Harps E et al (2008) Trastuzumab in pregnancy associated with poor fetal outcome. Ann Oncol 19:191–192
Sekar R, Stone PR (2007) Trastuzumab use for metastatic breast cancer in pregnancy. Obstet Gynecol 110:507–510
Shrim A, Garcia-Bournissen F, Maxwell C et al (2007) Favorable pregnancy outcome following Trastuzumab (Herceptin) use during pregnancy- Case report and updated literature review. Reprod Toxicol 23:611–613
Bader AA, Schlembach D, Tamussino KF et al (2007) Anhydramnios associated with administration of trastuzumab and paclitaxel for metastatic breast cancer during pregnancy. Lancet Oncol 8:79–81
Waterston AM, Graham J (2006) Effect of adjuvant trastuzumab on pregnancy. J Clin Oncol 24:321–322
Fanale MA, Uyei AR, Theriault RL et al (2005) Treatment of metastatic breast cancer with trastuzumab and vinorelbine during pregnancy. Clin Breast Cancer 6:354–356
Watson WJ (2005) Herceptin (trastuzumab) therapy during pregnancy: association with reversible anhydramnios. Obstet Gynecol 105:642–643
Azim HA Jr, Peccatori FA, Liptrott SJ et al (2009) Breast cancer and pregnancy: how safe is trastuzumab? Nat Rev Clin Oncol 6:367–370
Warraich Q, Smith N (2009) Herceptin therapy in pregnancy: continuation of pregnancy in the presence of anhydramnios. J Obstet Gynaecol 29:147–148
Roberts NJ, Auld BJ (2010) Trastuzamab (Herceptin)-related cardiotoxicity in pregnancy. J R Soc Med 103:157–159
Azim HA Jr, Peccatori FA, Pavlidis N (2010) Treatment of the pregnant mother with cancer: a systematic review on the use of cytotoxic, endocrine, targeted agents and immunotherapy during pregnancy. Part I: solid tumors. Cancer Treat Rev 36:101–109
Zagouri F, Sergentanis TN, Chrysikos D et al (2012) Taxanes for ovarian cancer during pregnancy: a systematic review. Oncology 83:234–238
Zagouri F, Sergentanis TN, Chrysikos D et al (2012) Taxanes for breast cancer during pregnancy: a systematic review. Clin Breast Cancer [Epub Ahead of print]
Pentsuk N, van der Laan JW (2009) An interspecies comparison of placental antibody transfer: new insights into developmental toxicity testing of monoclonal antibodies. Birth Defects Res B Dev Reprod Toxicol 86:328–344
Malek A (2003) Ex vivo human placenta models: transport of immunoglobulin G and its subclasses. Vaccine 21:3362–3364
Kilpatrick SJ (1997) Therapeutic interventions for oligohydramnios: amnioinfusion and maternal hydration. Clin Obstet Gynecol 40:328–336
Vanderheyden T, Kumar S, Fisk NM (2003) Fetal renal impairment. Semin Neonatol 8:279–289
Goodyer PR, Cybulsky A, Goodyer C (1993) Expression of the epidermal growth factor receptor in fetal kidney. Pediatr Nephrol 7:612–615
Jokhi PP, King A, Loke YW (1994) Reciprocal expression of epidermal growth factor receptor (EGF-R) and c-erbB2 by non-invasive and invasive human trophoblast populations. Cytokine 6:433–442
Lee KF, Simon H, Chen H et al (1995) Requirement for neuregulin receptor erbB2 in neural and cardiac development. Nature 378:394–398
Azim HA Jr, Metzger-Filho O, de Azambuja E et al (2012) Pregnancy occurring during or following adjuvant trastuzumab in patients enrolled in the HERA trial (BIG 01–01). Breast Cancer Res Treat 133:387–391
Acknowledgments
FZ is receiving a research grant from HeSMO.
Conflict of interest
The authors have declared no conflicts of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Zagouri, F., Sergentanis, T.N., Chrysikos, D. et al. Trastuzumab administration during pregnancy: a systematic review and meta-analysis. Breast Cancer Res Treat 137, 349–357 (2013). https://doi.org/10.1007/s10549-012-2368-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10549-012-2368-y