Summary
Purpose
To evaluate the treatment-induced menstrual changes in very young (< 35 years old) breast cancer patients.
Methods and materials
We retrospectively examined the clinical records of 160 patients, ranging in age from 18 to 34 years old (median age, 32 years), treated between June 1992 and December 2002. One hundred twenty patients underwent mastectomy and 40 underwent breast conserving surgery. Postoperatively, 80 patients were treated with alkylating agent-based chemotherapy regimens (CMF) and 80 with anthracycline-based regimens (AD). In addition, 57 patients received adjuvant radiotherapy, and 77 received anti-estrogen therapy. Treatment-induced menstrual changes and present menstrual status were evaluated from hospital records and by one-to-one interviews. The median follow-up period was 54 months (range, 29–156 months).
Results
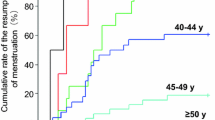
Treatment-induced menstrual change (amenorrhea) was occurred in 59 (36.9%) patients, 25 (31.3%) of those treated with CMF and 34 (42.5%) with AD (p=0.142). Amenorrhea occurred after a median 2 cycles of chemotherapy (range, 1–6 cycles). Menstruation resumed in 49 (83.1%) patients, 20 (80%) of those treated with CMF and 29 (85.3%) with AD (p=0.6). Median time to resumption of menstruation was median 3.5 months (range, 1–18 months) after amenorhrea. Disease recurred in 10 (16.9%) patients who experienced treatment-induced menstrual changes and in 18 (17.8%) of those who did not (p=0.89).
Conclusion
Although the overall incidence of treatment-induced menstrual change in breast cancer patients under age 35 was similar to that reported elsewhere, the rate of recovery from these change is higher. We observed no difference between CMF and AD treated patients in rates of amenorrhea or recovery from these changes.
Similar content being viewed by others
References
Anderson WF, Jatoi I, Devesa SS, Distinct breast cancer incidence and prognostic patterns in the NCI’s SEER program: suggesting a possible link between etiology and outcome Breast Cancer Res Treat 90: 127–137, 2005
Kalantaridou SN, Davis SR, Nelson LM, Premature ovarian failure Endocrinol Metab Clin North Am 27:989–1006, 1998
Morimoto T, Okazaki M, Endo T, Current status and goals of mammographic screening for breast cancer in Japan Breast Cancer 11:73–81, 2004
Walker RA, Lees E, Webb MB, Dearing SJ, Breast carcinomas occurring in young women (<35 years) are different Br J Cancer 74:1796–1800, 1996
Adami HO, Malker B, Holmberg L, Persson I, Stone B, The relation between survival and age at diagnosis in breast cancer N Engl J Med 315:559–563, 1986
Chung M, Chang HR, Bland KI, Wanebo HJ, Younger women with breast carcinoma have a poorer prognosis than older women Cancer 77:97–103, 1996
Kollias J, Elston CW, Ellis IO, Robertson JF, Blamey RW, Early-onset breast cancer-histopathological and prognostic considerations Br J Cancer 75:1318–1323, 1997
NIH Consensus Development Program: Adjuvant Therapy for Breast Cancer. National Institutes of Health Consensus Development Statement, November 1–3, 2000
Early Breast Cancer Trialists’ Collaborative Group: Polychemotherapy for early breast cancer: An overview of the randomised trials. Lancet 352: 930–942, 1998
Early Breast Cancer Trialists’ Collaborative Group: Tamoxifen for early breast cancer: An overview of the randomised trials. Lancet 351: 1451–1467, 1998
Wingo PA, Ries LA, Giovino GA, et al Annual report to the nation on the status of cancer, 1973–1996, with a special section on lung cancer and tobacco smoking J Natl Cancer Inst 91:675–690, 1999
Goodwin PJ, Ennis M, Pritchard KI, Trudeau M, Hood N, Risk of menopause during the first year after breast cancer diagnosis J Clin Oncol 17:2365–2370, 1999
Bines J, Oleske DM, Cobleigh MA, Ovarian function in premenopausal women treated with adjuvant chemotherapy for breast cancer J Clin Oncol 14:1718–1729, 1996
Chapman RM, Sutcliffe SB, Malpas JS, Cytotoxic-induced ovarian failure in women with Hodgkin’s disease. I. Hormone function JAMA 242:1877–1881, 1976
Warne GL, Fairley KF, Hobbs JB, Martin FI, Cyclophosphamide-induced ovarian failure N Engl J Med 289:1159–1162, 1973
Koyama H, Wada T, Nishizawa Y, Iwanaga T, Aoki Y, Cyclophosphamide-induced ovarian failure and its therapeutic significance in patients with breast cancer Cancer 39:1403–1409, 1977
Bonadonna G, Valagussa P, Adjuvant systemic therapy for resectable breast cancer J Clin Oncol 3:259–275, 1985
Goldhirsch A, Gelber RD, Yothers G, et al Adjuvant therapy for very young women with breast cancer: need for tailored treatments J Natl Cancer Inst Monogr 30:44–51, 2001
Nabholtz J, Pienkowski T, Mackey J, et al Phase III trial comparing TAC with FAC in the adjuvant treatment of node positive breast cancer patients: Interim analysis of the BCIRG 001 study Proc Am Soc Clin Oncol 21(abstr 141):361, 2002
Hortobagyi GN, Buzdar AU, Marcus CE, Smith TL, Immediate and long-term toxicity of adjuvant chemotherapy regimens containing doxorubicin in trials at M.D. Anderson Hospital and Tumor Institute NCI Monogr 1:105–109, 1986
Levine MN, Bramwell VH, Pritchard KI, et al Randomized trial of intensive cyclophosphamide, epirubicin, and fluorouracil chemotherapy compared with cyclophosphamide, methotrexate, and fluoruracil in premenopausal women with node-positive breast cancer: National Cancer Institute of Canada Clinical Trials Group J Clin Oncol 16:2651–2658, 1998
Shapiro CL, Manola J, Leboff M, Ovarian failure after chemotherapy is associated with rapid bone loss in women with early stage breast cancer J Clin Oncol 19:3306–3311, 2001
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kil, W.J., Ahn, S.D., Shin, S.S. et al. Treatment-induced menstrual changes in very young (<35 years old) breast cancer patients. Breast Cancer Res Treat 96, 245–250 (2006). https://doi.org/10.1007/s10549-005-9059-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10549-005-9059-x