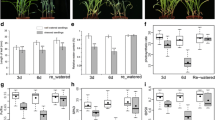
Abstract
Drought stress is one of the major abiotic stresses that limit growth, development and yield of maize crops. To better understand the responses of maize inbred lines with different levels of drought resistance and the molecular mechanism of exogenous glycine betaine (GB) in alleviating drought stress, the responses of two maize inbred lines to drought stress and to the stress-mitigating effects of exogenous GB were investigated. Seedling morphology, physiological and biochemical indexes, root cell morphology and root transcriptome expression profiles were compared between a drought-tolerant inbred line Chang 7-2 and drought-sensitive inbred line TS141. Plants of both lines were subjected to treatments of drought stress alone and drought stress with application of exogenous GB. The results showed that with the increase of drought treatment time, the growth and development of TS141 were inhibited, while those of Chang 7-2 were not significantly different from those of the control (no drought stress and GB). Compared with the corresponding data of the drought-stress group, every index measured from the two inbred lines indicated mitigating effects from exogenous GB, and TS141 produced stronger mitigating responses due to the GB. Transcriptome analysis showed that 562 differentially expressed genes (DEGs) were up-regulated and 824 DEGs were down-regulated in both inbred lines under drought stress. Due to the exogenous GB, 1061 DEGs were up-regulated and 424 DEGs were down-regulated in both lines. In addition, quantitative real-time polymerase chain reaction (qRT-PCR) was used to verify 10 DEGs screened from the different treatments. These results showed that the expression of 9 DEGs were basically consistent with their respective transcriptome expression profiles. The results of this study provide models of potential mechanisms of drought tolerance in maize as well as potential mechanisms of how exogenous GB may regulate drought tolerance.







Similar content being viewed by others
References
Amorim J, Souza K, Duarte C, Duarte I, Sarubbo L (2020) Plant and bacterial nanocellulose: production, properties and applications in medicine, food, cosmetics, electronics and engineering. A Rev Environ Chem Lett 18(3):851–869. https://doi.org/10.1007/s10311-020-00989-9
Anderson N, Chapple C (2014) Perturbing lignin biosynthesis: metabolic changes in response to manipulation of the phenylpropanoid pathway. John Wiley & Sons Ltd. https://doi.org/10.1002/9781118329634.ch2
Ashraf M, Foolad MR (2007) Roles of glycine betaine and proline in improving plant abiotic stress resistance. Environ Exp Bot 59(2):206–216. https://doi.org/10.1016/j.envexpbot.2005.12.006
Audic S, Claverie J (1997) The significance of digital gene expression profiles. Genome Res 7(10):986–995. https://doi.org/10.1101/gr.7.10.986
Avramova V, Nagel KA, AbdElgawad H, Bustos D, DuPlessis M, Fiorani F, Beemster GT (2016) Screening for drought tolerance of maize hybrids by multi-scale analysis of root and shoot traits at the seedling stage. J Exp Bot 67(8):2453–2466. https://doi.org/10.1093/jxb/erw055
Babita M, Maheswari M, Rao LM, Shanker AK, Rao DG (2010) Osmotic adjustment, drought tolerance and yield in castor (Ricinus communis L.) hybrids. Environ Exp Bot 69(3):243–249. https://doi.org/10.1016/j.envexpbot.2010.05.006
Banu MN, Hoque MA, Watanabe-Sugimoto M, Islam MM, Uraji M, Matsuoka K, Nakamura Y, Murata Y (2010) Proline and glycinebetaine ameliorated NaCl stress via scavenging of hydrogen peroxide and methylglyoxal but not superoxide or nitric oxide in tobacco cultured cells. Biosci Biotechnol Biochem 74(10):2043–2049. https://doi.org/10.1271/bbb.100334
Bargmann BO, Laxalt AM, ter Riet B, van Schooten B, Merquiol E, Testerink C, Haring MA, Bartels D, Munnik T (2009) Multiple PLDs required for high salinity and water deficit tolerance in plants. Plant Cell Physiol 50(1):78–89. https://doi.org/10.1093/pcp/pcn173
Benešová M, Holá D, Fischer L, Jedelský PL, Hnilička F, Wilhelmová N, Rothová O, Kočová M, Procházková D, Honnerová J, Fridrichová L, Hniličková H (2012) The physiology and proteomics of drought tolerance in maize: early stomatal closure as a cause of lower tolerance to short-term dehydration? PLoS ONE 7(6):e38017. https://doi.org/10.1371/journal.pone.0038017
Benjamini Y, Hochberg Y (1995) Controlling the false discovery rate: a practical and powerful approach to multiple testing. J Roy Stat Soc 57(1):289–300. https://doi.org/10.1111/j.2517-6161.1995.tb02031.x
Bouvier-Navé P, Benveniste P, Oelkers P, Sturley SL, Schaller H (2000) Expression in yeast and tobacco of plant cDNAs encoding acyl CoA:diacylglycerol acyltransferase. Eur J Biochem 267(1):85–96. https://doi.org/10.1046/j.1432-1327.2000.00961.x
Bowman MJ, Park W, Bauer PJ, Udall JA, Page JT, Raney J, Scheffler BE, Jones DC, Campbell BT (2013) RNA-Seq transcriptome profiling of upland cotton (Gossypium hirsutum L.) root tissue under water-deficit stress. PLoS ONE 8(12):e82634. https://doi.org/10.1371/journal.pone.0082634
Carillo P, Cirillo C, De Micco V, Arena C, De Pascale S, Rouphael Y (2019) Morpho-anatomical, physiological and biochemical adaptive responses to saline water of Bougainvillea spectabilis Willd. trained to different canopy shapes. Agric Water Manag 212:12–22. https://doi.org/10.1016/j.agwat.2018.08.037
Chanda B, **a Y, Mandal MK, Yu K, Sekine KT, Gao QM, Selote D, Hu Y, Stromberg A, Navarre D, Kachroo A, Kachroo P (2011) Glycerol-3-phosphate is a critical mobile inducer of systemic immunity in plants. Nat Genet 43(5):421–427. https://doi.org/10.1038/ng.798
Chen TH, Murata N (2008) Glycinebetaine: an effective protectant against abiotic stress in plants. Trends Plant Sci 13(9):499–505. https://doi.org/10.1016/j.tplants.2008.06.007
Du Z, Zhou X, Ling Y, Zhang Z, Su Z (2010) agriGO: a GO analysis toolkit for the agricultural community. Nucleic Acids Res. https://doi.org/10.1093/nar/gkq310
Dugas DV, Monaco MK, Olsen A, Klein RR, Kumari S, Ware D, Klein PE (2011) Functional annotation of the transcriptome of Sorghum bicolor in response to osmotic stress and abscisic acid. BMC Genom 12:514. https://doi.org/10.1186/1471-2164-12-514
Fan W, Zhang M, Zhang H, Zhang P (2012) Improved tolerance to various abiotic stresses in transgenic sweet potato (Ipomoea batatas) expressing spinach betaine aldehyde dehydrogenase. PLoS ONE 7(5):e37344. https://doi.org/10.1371/journal.pone.0037344
Farmer EE, Goossens A (2019) Jasmonates: what allene oxide synthase does for plants. J Exp Bot 70(13):3373–3378. https://doi.org/10.1093/jxb/erz254
Farooq M, Basra SMA, Wahid A, Cheema MA, Khaliq A (2008) Physiological role of exogenously applied glycinebetaine to improve drought tolerance in fine grain aromatic rice (Oryza sativa L.). J Agronomy Crop Sci 194(5):325–333
Freitas de Campos M, de Carvalho K, de Souza F, Marur C, Protasio Pereira L, Bespalhok Filho J, Esteves Vieira L (2011) Drought tolerance and antioxidant enzymatic activity in transgenic “Swingle” citrumelo plants over-accumulating proline. Environ Exp Bot 72(2):242–250. https://doi.org/10.1016/j.envexpbot.2011.03.009
Galmés J, Flexas J, Savé R, Medrano H (2007) Water relations and stomatal characteristics of Mediterranean plants with different growth forms and leaf habits: responses to water stress and recovery. Plant Soil 290(1):139–155
Gálvez L, González EM, Arrese-Igor C (2005) Evidence for carbon flux shortage and strong carbon/nitrogen interactions in pea nodules at early stages of water stress. J Exp Bot 56(419):2551–2561. https://doi.org/10.1093/jxb/eri249
Gao Y, Li C, Lou K (2011) Effect of glycine betaine on physiological characteristics of cotton seedlings under drought stress. Plant Nutr Fert Sci. https://doi.org/10.3724/SP.J.1011.2011.00353
Genard H, Le Saos J, Billard J-P, Tremolieres A, Boucaud J (1991) Effect of salinity on lipid composition, glycine betaine content and photosynthetic activity in chloroplasts of Suaeda maritima. Plant Physiol Biochem 29(5):421–427
Giardi M, Cona A, Geiken B, Kučera T, Masojídek J, Mattoo A (1996) Long-term drought stress induces structural and functional reorganization of photosystem II. Planta 199(1):118–125. https://doi.org/10.1007/BF00196888
Guo L, Devaiah SP, Narasimhan R, Pan X, Zhang Y, Zhang W, Wang X (2012) Cytosolic glyceraldehyde-3-phosphate dehydrogenases interact with phospholipase Dδ to transduce hydrogen peroxide signals in the Arabidopsis response to stress. Plant Cell 24(5):2200–2212. https://doi.org/10.1105/tpc.111.094946
Gupta B, Huang B (2014) Mechanism of salinity tolerance in plants: physiological, biochemical, and molecular characterization. Int J Genom 2014:701596. https://doi.org/10.1155/2014/701596
Haga K, Iino M (2004) Phytochrome-mediated transcriptional up-regulation of allene oxide synthase in rice seedlings. Plant Cell Physiol 45(2):119–128. https://doi.org/10.1093/pcp/pch025
Hany, Samir, Osman (2015) Enhancing antioxidant-yield relationship of pea plant under drought at different growth stages by exogenously applied glycine betaine and proline. Annals of Agricultural Ence 60(2):389–402
Hong Y, Pan X, Welti R, Wang X (2008) Phospholipase Dalpha3 is involved in the hyperosmotic response in Arabidopsis. Plant Cell 20(3):803–816. https://doi.org/10.1105/tpc.107.056390
Hui H, XU X, Li Q (2003) Exogenous betaine improves photosynthesis of Lycium barbarum under salt stress. Acta Botanica Boreali-occidentalia Sinica 2137–2142
Jiang W, Zhu X, Wang J, Xu-Feng L, Yang Y (2009) Physiological analysis of AtGluRS transgenic Arabidopsis under the treatment of different phytohormones. J Sichuan Univ. https://doi.org/10.3969/j.issn.0490-6756.2009.02.044
Joshi CP, Mansfield SD (2007) The cellulose paradox–simple molecule, complex biosynthesis. Curr Opin Plant Biol 10(3):220–226. https://doi.org/10.1016/j.pbi.2007.04.013
Kakumanu A, Ambavaram MM, Klumas C, Krishnan A, Batlang U, Myers E, Grene R, Pereira A (2012) Effects of drought on gene expression in maize reproductive and leaf meristem tissue revealed by RNA-Seq. Plant Physiol 160(2):846–867. https://doi.org/10.1104/pp.112.200444
Kanehisa M, Araki M, Goto S, Hattori M, Hirakawa M, Itoh M, Katayama T, Kawashima S, Okuda S, Tokimatsu T, Yamanishi Y (2008) KEGG for linking genomes to life and the environment. Nucleic Acids Res. https://doi.org/10.1093/nar/gkm882
Khan SH, Ahmad N, Ahmad F, Kumar R (2010) Naturally occurring organic osmolytes: from cell physiology to disease prevention. IUBMB Life 62(12):891–895. https://doi.org/10.1002/iub.406
Kim D, Langmead B, Salzberg SL (2015) HISAT: a fast spliced aligner with low memory requirements. Nat Methods 12(4):357–360. https://doi.org/10.1038/nmeth.3317
Kim YH, Bae JM, Huh GH (2010) Transcriptional regulation of the cinnamyl alcohol dehydrogenase gene from sweet potato in response to plant developmental stage and environmental stress. Plant Cell Rep 29(7):779–791. https://doi.org/10.1007/s00299-010-0864-2
Knauss S, Rohrmeier T, Lehle L (2003) The auxin-induced maize gene ZmSAUR2 encodes a short-lived nuclear protein expressed in elongating tissues. J Biol Chem 278(26):23936–23943. https://doi.org/10.1074/jbc.M212585200
Kumar S, Reddy A, Sudhakar C (2003) NaCl effects on proline metabolism in two high yielding genotypes of mulberry (Morus alba L.) with contrasting salt tolerance. Plant Sci 165(6):1245–1251. https://doi.org/10.1016/S0168-9452(03)00332-7
Langmead B, Salzberg SL (2012) Fast gapped-read alignment with Bowtie 2. Nat Methods 9(4):357–359. https://doi.org/10.1038/nmeth.1923
Lea P, Sodek L, Parry M, Shewry P, Halford N (2010) Asparagine in plants. Ann Appl Biol 150(1):1–26. https://doi.org/10.1111/j.1744-7348.2006.00104.x
Li B, Dewey C (2011) RSEM: accurate transcript quantification from RNA-Seq data with or without a reference genome. BMC Bioinformatics 12(1):323. https://doi.org/10.1186/1471-2105-12-323
Li D, Zhang T, Wang M, Liu Y, Brestic M, Chen THH, Yang X (2018) Genetic engineering of the biosynthesis of glycine betaine modulates phosphate homeostasis by regulating phosphate acquisition in tomato. Front Plant Sci 9:1995. https://doi.org/10.3389/fpls.2018.01995
Li Y, Beisson F, Koo AJ, Molina I, Pollard M, Ohlrogge J (2007) Identification of acyltransferases required for cutin biosynthesis and production of cutin with suberin-like monomers. Proc Natl Acad Sci USA 104(46):18339–18344. https://doi.org/10.1073/pnas.0706984104
Liu F, Zhang X, Lu C, Zeng X, Li Y, Fu D, Wu G (2015) Non-specific lipid transfer proteins in plants: presenting new advances and an integrated functional analysis. J Exp Bot 66(19):5663–5681. https://doi.org/10.1093/jxb/erv313
Liu Q, Song F (2007) A comparison of root anatomical structure of maize genotypes with different drought tolerance. Agric Res Arid Areas 25(2):86–91
Liu Y, Du H, He X, Huang B, Wang Z (2012) Identification of differentially expressed salt-responsive proteins in roots of two perennial grass species contrasting in salinity tolerance. J Plant Physiol 169(2):117–126. https://doi.org/10.1016/j.jplph.2011.08.019
Lobell DB, Roberts MJ, Schlenker W, Braun N, Little BB, Rejesus RM, Hammer GL (2014) Greater sensitivity to drought accompanies maize yield increase in the U.S. Midwest. Science 344(6183):516–519. https://doi.org/10.1126/science.1251423
Ma X, Wang Y, **e S, Wang C, Wang W (2007) Glycinebetaine application ameliorates negative effects of drought stress in tobacco. Russ J Plant Physiol 54(4):472–479. https://doi.org/10.1134/S1021443707040061
Moussa H, Abdel-Aziz S (2008) Comparative response of drought tolerant and drought sensitive maize genotypes to water stress. Aust J Crop Sci 1(1):31–36. https://doi.org/10.1007/978-0-387-77337-7_19
Nagalakshmi U, Wang Z, Waern K, Shou C, Raha D, Gerstein M, Snyder M (2008) The transcriptional landscape of the yeast genome defined by RNA sequencing. Science 320(5881):1344–1349. https://doi.org/10.1126/science.1158441
Peng W, Wang W, Liang H, Wang J, Wu J, Liu C (2003) The effect of water stress on the microstructure of Glycyrrhica uralensis. J Agric Univ Hebei 26(3):46–48. https://doi.org/10.1007/BF02974893
Peng Y, Zhao X, Ren X, Li J (2013) Effect of drought stress on growth of different plant type maize (Zea mays) in the bell-mouthed period. J Desert Res 1997:1064–1070
Persson S, Paredez A, Carroll A, Palsdottir H, Doblin M, Poindexter P, Khitrov N, Auer M, Somerville CR (2007) Genetic evidence for three unique components in primary cell-wall cellulose synthase complexes in Arabidopsis. Proc Natl Acad Sci USA 104(39):15566–15571. https://doi.org/10.1073/pnas.0706592104
Rahman MS, Miyake H, Takeoka Y (2008) Effects of exogenous glycinebetaine on growth and ultrastructure of salt-stressed rice seedlings (Oryza sativa L.). Plant Product Sci 5(1):33–44
Raja R, Brien H, Seepaul R, Lokhande S, Gajanayake B, Brand D (2010) Exogenous application of glycinebetaine facilitates maize (Zea mays L.) growth under water deficit conditions. Am J Exp Agric 3(1):1–13. https://doi.org/10.9734/AJEA/2013/1730
Riccardi F, Gazeau P, de Vienne D, Zivy M (1998) Protein changes in response to progressive water deficit in maize. Quantitative variation and polypeptide identification. Plant Physiol 117(4):1253–1263. https://doi.org/10.1104/pp.117.4.1253
Saha S, Mina BL, Gopinath KA, Kundu S, Gupta HS (2008) Relative changes in phosphatase activities as influenced by source and application rate of organic composts in field crops. Biores Technol 99(6):1750–1757. https://doi.org/10.1016/j.biortech.2007.03.049
Sakaki T, Kondo N, Yamada M (1990) Pathway for the synthesis of triacylglycerols from monogalactosyldiacylglycerols in ozone-fumigated spinach leaves. Plant Physiol 94(2):773–780. https://doi.org/10.1104/pp.94.2.773
Schiefelbein JW, Benfey PN (1991) The development of plant roots: new approaches to underground problems. Plant Cell 3(11):1147–1154. https://doi.org/10.1105/tpc.3.11.1147
Schuerger AC, Brown CS, Stryjewski EC (1997) Anatomical features of pepper plants (Capsicum annuum L.) grown under red light-emitting diodes supplemented with blue or far-red light. Ann Bot 79(3):273–282. https://doi.org/10.1006/anbo.1996.0341
Shan X, Li Y, Jiang Y, Jiang Z, Hao W, Yuan Y (2013) Transcriptome profile analysis of maize seedlings in response to high-salinity, drought and cold stresses by deep sequencing. Plant Mol Biol Rep 31(6):1485–1491. https://doi.org/10.1007/s11105-013-0622-z
Storey JD, Tibshirani R (2003) Statistical significance for genomewide studies. Proc Natl Acad Sci USA 100(16):9440–9445. https://doi.org/10.1073/pnas.1530509100
Swindell WR, Huebner M, Weber AP (2007) Transcriptional profiling of Arabidopsis heat shock proteins and transcription factors reveals extensive overlap between heat and non-heat stress response pathways. BMC Genomics 8:125. https://doi.org/10.1186/1471-2164-8-125
Timmers J, Vernhettes S, Desprez T, Vincken JP, Visser RG, Trindade LM (2009) Interactions between membrane-bound cellulose synthases involved in the synthesis of the secondary cell wall. FEBS Lett 583(6):978–982. https://doi.org/10.1016/j.febslet.2009.02.035
Timperio AM, Egidi MG, Zolla L (2008) Proteomics applied on plant abiotic stresses: role of heat shock proteins (HSP). J Proteomics 71(4):391–411. https://doi.org/10.1016/j.jprot.2008.07.005
Vanholme R, Cesarino I, Rataj K, **ao Y, Sundin L, Goeminne G, Kim H, Cross J, Morreel K, Araujo P, Welsh L, Haustraete J, McClellan C, Vanholme B, Ralph J, Simpson G, Halpin C, Boerjan W (2013) Caffeoyl shikimate esterase (CSE) is an enzyme in the lignin biosynthetic pathway in Arabidopsis. Science 341(6150):1103–1106. https://doi.org/10.1126/science.1241602
Wang L, Feng Z, Wang X, Wang X, Zhang X (2010) DEGseq: an R package for identifying differentially expressed genes from RNA-seq data. Bioinformatics 26(1):136–138. https://doi.org/10.1093/bioinformatics/btp612
Wei D, Zhang W, Wang C, Meng Q, Li G, Chen THH, Yang X (2017) Genetic engineering of the biosynthesis of glycinebetaine leads to alleviate salt-induced potassium efflux and enhances salt tolerance in tomato plants. Plant Sci 257:74–83. https://doi.org/10.1016/j.plantsci.2017.01.012
**n Z, Zhou X, Pilet P (1997) Level changes of jasmonic, abscisic, and indole-3yl-acetic acids in maize under desiccation stress. J Plant Physiol 151(1):120–124. https://doi.org/10.1016/S0176-1617(97)80047-8
Yu YH, Ginsberg HN (2004) The role of acyl-CoA:diacylglycerol acyltransferase (DGAT) in energy metabolism. Ann Med 36(4):252–261. https://doi.org/10.1080/07853890410028429
Zenda T, Liu S, Wang X, ** H, Liu G, Duan H (2018) Comparative proteomic and physiological analyses of two divergent maize inbred lines provide more insights into drought-stress tolerance mechanisms. Int J Mol Sci. https://doi.org/10.3390/ijms19103225
Zeng W, Chen F, Zhuang Z, Ding Y, Peng Y (2020) Mitigation effect analysis of different exogenous regulatory substances on different drought-tolerance maize inbred lines under drought stress. Agric Res Arid Areas 38(3):55–63
Zhao X, Ma Q, Yang X, Liang C, Fang Y, Wang W, Zou Q (2005) Effects of root-applied glycinebetaine on the composition and function of wheat thylakoid membrane under drought stress. Physiol Mol Biol Plants 31(2):135–142. https://doi.org/10.1360/aps040074
Zou J, Wei Y, Jako C, Kumar A, Selvaraj G, Taylor DC (1999) The Arabidopsis thaliana TAG1 mutant has a mutation in a diacylglycerol acyltransferase gene. Plant J 19(6):645–653. https://doi.org/10.1046/j.1365-313x.1999.00555.x
Acknowledgements
This research was supported by the Gansu Provincial Key Laboratory of Aridland Crop Science, Gansu Agricultural University (No. GHSJ-2020-Z5), the central government guides the special project of local science and technology development, and the Lanzhou Science and Technology Bureau (No. 2020-RC-122).
Supporting Information
Table S1. Primer sequences used for qRT-PCR analysis in this article.
Table S2. Quality control of the total RNA for RNA-Seq.
Table S3. Summary of sequencing data.
Table S4. Summary of genome map** information.
Table S5. DEGs up-regulated in Chang 7-2 seedling roots under D treatment.
Table S6. DEGs down-regulated in Chang 7-2 seedling roots under D treatment.
Table S7. DEGs up-regulated in TS141 seedling roots under D treatment.
Table S8. DEGs down-regulated in TS141 seedling roots under D treatment.
Table S9. DEGs up-regulated in Chang 7-2 seedling roots under T treatment.
Table S10. DEGs down-regulated in Chang 7-2 seedling roots under T treatment.
Table S11. DEGs up-regulated in TS141 seedling roots under T treatment.
Table S12. DEGs down-regulated in TS141 seedling roots under T treatment.
Table S13. Function analysis of significantly up-regulated DEGs in two varieties under drought stress.
Table S14. Function analysis of DEGs that significantly up-regulated in Chang 7-2 and down-regulated in TS141 under drought stress.
Table S15. Function analysis of significantly up-regulated DEGs in two varieties under T treatment.
Table S16. DEGs used for qRT-PCR analysis.
Fig S1 Heat map of correlation analysis between samples.
Fig S2 GO annotation of up-down-regulated genes in two inbred lines under different treatments.
Author information
Authors and Affiliations
Contributions
YP designed the experiments. MB wrote the manuscript. WZ and MB performed the experiments and analyzed data. FC, XJ, ZZ, BJ, JW and LJ participated in the critical reading and discussion of the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
Authors declare that there are no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Bai, M., Zeng, W., Chen, F. et al. Transcriptome expression profiles reveal response mechanisms to drought and drought-stress mitigation mechanisms by exogenous glycine betaine in maize. Biotechnol Lett 44, 367–386 (2022). https://doi.org/10.1007/s10529-022-03221-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10529-022-03221-6