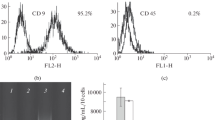
We studied the effect of neural stem cells (NSC) and mesenchymal stem cells (MSC) from mouse adipose tissue on survival, clonogenic activity, and senescence of NSC after exposure to γ-radiation. It was found that survival and clonogenic activity of NSC irradiated in doses of 1 and 2 Gy was enhanced when irradiated cells were co-cultured with non-irradiated NSC and MSC in permeable Transwell inserts. The proportion of senescent NSC (cells with high β-galactosidase activity) increased with increasing irradiation dose. Co-culturing with non-irradiated NSC in 3 days after irradiation in a dose of 1 Gy led to a decrease in the proportion of senescent cells among irradiated NSC. Factors secreted by NSC and MSC can become the basis for the development of means for prevention and treatment of damage to brain cells resulting from radiation therapy of head and neck cancer.
Similar content being viewed by others
References
Mikhailov VM, Kaminskaya EV, Popov BV, Kuzovatov SN, Grinchuk TM, Nikolsky NN, Skripkina NS, Kosyakova GP, Zaichik AM. Characteristics of tumors developed in mdx mice after transplantation of GFP-positive mesenchymal stem cells isolated from bone marrow of transgenic C57BL/6 mice. Cell and Tissue Biology. 2010;4(5):419-423.
Moskaleva EY, Semochkina YP, Rodina AV, Chukalova AA, Posypanova GA. Effects of γ-radiation on mesenchymal stem cells from mouse bone marrow and brain and their ability to induce tumors. Radiats. Biol. Radioekol. 2017;57(3):245-256. doi: https://doi.org/10.7868/S0869803117030018. Russian.
Posypanova GA, Ratushnyak MG, Vysotskaya OV, Glukhov AI, Semochkina YuP, Rodina AV, Moskaleva EYu. Protection of neural stem cells from genotoxic effects by factors secreted by mesenchymal stem cells. Mol. Med. 2018;16(6):28-34. doi: https://doi.org/10.29296/24999490-2018-06-05. Russian.
Posypanova GA, Ratushnyak MG, Semochkina YP, Abisheva AA, Moskaleva EYu. The sensitivity of the cultured murine neural stem cells to the ionizing radiation. Tsitologiya. 2019;61(10):806-816. doi: https://doi.org/10.1134/S0041377119100067. Russian.
Rodina AV, Semochkina YP, Vysotskaya OV, Glukhov AI, Moskaleva EYu. Features of long-term cultured adipose tissuederived mesenchymal stem cells response to γ-irradiation. Radiats. Biol. Radioekol. 2019;59(3):243-254. Russian.
Acharya MM, Christie LA, Hazel TG, Johe KK, Limoli CL. Transplantation of human fetal-derived neural stem cells improves cognitive function following cranial irradiation. Cell Transplant. 2014;23(10):1255-1266. doi: https://doi.org/10.3727/096368913X670200
Acharya MM, Christie LA, Lan ML, Giedzinski E, Fike JR, Rosi S, Limoli CL. Human neural stem cell transplantation ameliorates radiation-induced cognitive dysfunction. Cancer Res. 2011;71(14):4834-4845.
Acharya MM, Martirosian V, Christie LA, Riparip L, Strnadel J, Parihar VK, Limoli CL. Defining the optimal window for cranial transplantation of human induced pluripotent stem cellderived cells to ameliorate radiation-induced cognitive impairment. Stem Cells Transl. Med. 2015;4(1):74-83. doi: https://doi.org/10.5966/sctm.2014-0063
Acharya MM, Rosi S, Jopson T, Limoli CL. Human neural stem cell transplantation provides long-term restoration of neuronal plasticity in the irradiated hippocampus. Cell Transplant. 2015;24(4):691-702. doi: https://doi.org/10.3727/096368914X684600
Baraniak PR, McDevitt TC. Stem cell paracrine actions and tissue regeneration. Regen. Med. 2010;5(1):121-143. doi: https://doi.org/10.2217/rme.09.74
Campisi J, d’Adda di Fagagna F. Cellular senescence: when bad things happen to good cells. Nat. Rev. Mol. Cell Biol. 2007;8(9):729-740. doi: https://doi.org/10.1038/nrm2233
Constantin G, Marconi S, Rossi B, Angiari S, Calderan L, Anghileri E, Gini B, Bach SD, Martinello M, Bifari F, Galiè M, Turano E, Budui S, Sbarbati A, Krampera M, Bonetti B. Adipose-derived mesenchymal stem cells ameliorate chronic experimental autoimmune encephalomyelitis. Stem Cells. 2009;27(10):2624-2635. doi: https://doi.org/10.1002/stem.194
Drago D, Cossetti C, Iraci N, Gaude E, Musco G, Bachi A, Pluchino S. The stem cell secretome and its role in brain repair. Biochimie. 2013;95(12):2271-2285. doi: https://doi.org/10.1016/j.biochi.2013.06.020
Guo S, Wang T, Zhang S, Chen P, Cao Z, Lian W, Guo J, Kang Y. Adipose-derived stem cell-conditioned medium protects fibroblasts at different senescent degrees from UVB irradiation damages. Mol. Cell. Biochem. 2020;463(1-2):67-78. doi: https://doi.org/10.1007/s11010-019-03630-8
Kirkland JL, Tchkonia T. Senolytic drugs: from discovery to translation. J. Intern. Med. 2020;288(5):518-536. doi: https://doi.org/10.1111/joim.13141
Kritsilis M, Rizou SV, Koutsoudaki PN, Evangelou K, Gorgoulis VG, Papadopoulos D. Ageing, cellular senescence and neurodegenerative disease. Int. J. Mol. Sci. 2018;19(10):2937. doi: https://doi.org/10.3390/ijms19102937
Lees JS, Sena ES, Egan KJ, Antonic A, Koblar SA, Howells DW, Macleod MR. Stem cell-based therapy for experimental stroke: a systematic review and meta-analysis. Int. J. Stroke. 2012;7(7):582-588. doi: https://doi.org/10.1111/j.1747-4949.2012.00797.x
Li M, You L, Xue J, Lu Y. Ionizing radiation-induced cellular senescence in normal, non-transformed cells and the involved DNA damage response: a mini review. Front. Pharmacol. 2018;9:522. doi: https://doi.org/10.3389/fphar.2018.00522
Martínez-Cué C, Rueda N. Cellular senescence in neurodegenerative diseases. Front. Cell. Neurosci. 2020;14:16. doi: https://doi.org/10.3389/fncel.2020.00016
Miller RH, Bai L, Lennon DP, Caplan AI. The potential of mesenchymal stem cells for neural repair. Discov. Med. 2010;9(46):236-242.
Mitchell R, Mellows B, Sheard J, Antonioli M, Kretz O, Chambers D, Zeuner MT, Tomkins JE, Denecke B, Musante L, Joch B, Debacq-Chainiaux F, Holthofer H, Ray S, Huber TB, Dengjel J, De Coppi P, Widera D, Patel K. Secretome of adipose-derived mesenchymal stem cells promotes skeletal muscle regeneration through synergistic action of extracellular vesicle cargo and soluble proteins. Stem Cell Res. Ther. 2019;10(1):116. doi: https://doi.org/10.1186/s13287-019-1213-1
Munoz JR, Stoutenger BR, Robinson AP, Spees JL, Prockop DJ. Human stem/progenitor cells from bone marrow promote neurogenesis of endogenous neural stem cells in the hippocampus of mice. Proc. Natl Acad. Sci. USA. 2005;102(50):18171- 18176. doi: https://doi.org/10.1073/pnas.0508945102
Nguyen N, Lee SB, Lee YS, Lee KH, Ahn JY. Neuroprotection by NGF and BDNF against neurotoxin-exerted apoptotic death in neural stem cells are mediated through Trk receptors, activating PI3-kinase and MAPK pathways. Neurochem. Res. 2009;34(5):942-951. doi: https://doi.org/10.1007/s11064-008-9848-9
Niwa O, Barcellos-Hoff MH, Globus RK, Harrison JD, Hendry JH, Jacob P, Martin MT, Seed TM, Shay JW, Story MD, Suzuki K, Yamashita S; ICRP. ICRP Publication 131: Stem cell biology with respect to carcinogenesis aspects of radiological protection. Ann. ICRP. 2015;44(3-4):7-357. doi: https://doi.org/10.1177/0146645315595585
Schneider L, Pellegatta S, Favaro R, Pisati F, Roncaglia P, Testa G, Nicolis SK, Finocchiaro G, d’Adda di Fagagna F. DNA damage in mammalian neural stem cells leads to astrocytic differentiation mediated by BMP2 signaling through JAKSTAT. Stem Cell Reports. 2013;1(2):123-138. doi: https://doi.org/10.1016/j.stemcr.2013.06.004
Wang AS, Dreesen O. Biomarkers of cellular senescence and skin aging. Front. Genet. 2018;9:247. doi: https://doi.org/10.3389/fgene.2018.00247
White RR, Vijg J. Do DNA double-strand breaks drive aging? Mol. Cell. 2016;63(5):729-738. doi: https://doi.org/10.1016/j.molcel.2016.08.004
Willis CM, Nicaise AM, Hamel R, Pappa V, Peruzzotti-Jametti L, Pluchino S. Harnessing the neural stem cell secretome for regenerative neuroimmunology. Front. Cell. Neurosci. 2020;14:590960. doi: https://doi.org/10.3389/fncel.2020.590960
Xu P, **n Y, Zhang Z, Zou X, Xue K, Zhang H, Zhang W, Liu K. Extracellular vesicles from adipose-derived stem cells ameliorate ultraviolet B-induced skin photoaging by attenuating reactive oxygen species production and inflammation. Stem Cell Res. Ther. 2020;11(1):264. doi: https://doi.org/10.1186/s13287-020-01777-6
Author information
Authors and Affiliations
Corresponding author
Additional information
Translated from Kletochnye Tekhnologii v Biologii i Meditsine, No. 3, pp. 147-155, September, 2021
Rights and permissions
About this article
Cite this article
Ratushnyak, M.G., Semochkina, Y.P., Zhirnik, A.S. et al. Improved Survival and Regeneration of Irradiated Mouse Neural Stem Cells after Co-Culturing with Non-Irradiated Mouse Neural Stem Cells or Mesenchymal Stem Cells from the Adipose Tissue. Bull Exp Biol Med 172, 228–235 (2021). https://doi.org/10.1007/s10517-021-05368-0
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10517-021-05368-0