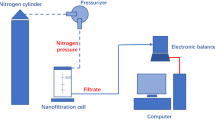
Aspects of membrane separation of borated water streams containing LiCl, NaCl, KCl, CsCl salts at concentrations 2–50 mmol/dm3 were investigated for the purpose of preventing boric acid ingress into drain water, decreasing the amount of radwaste produced, and making ion-exchange filter media more efficient. A Vontron VNF2 (CPR) nanofiltration element was the object of investigation. It was shown that changing the concentration of boric acid in the initial solution does not significantly affect the membrane separation of alkali-metal and ammonium salts, while increasing their concentration decreases the salt-retention of the membrane. The retention of boric acid remains constant both for its individual solutions and in the presence of chloride salts of alkali metals and ammonium. The permeability was calculated and the dimensions of the nanofiltration element as part of a local installation were determined.
Similar content being viewed by others
References
V. A. Vinnitskii, A. F. Nechaev, and A. S. Chugunov, “Dynamics of boric acid sorption by various forms of high-basic anion exchanger AV-17-8 and minimization of its losses in the NPP technological cycle,” Izv. SPbGTI(TU), No. 20(46), 81–84 (2013).
K. Kezia, J. Lee, A. Hill, et al., “Convective transport of boron through a brackish water reverse osmosis membrane,” J. Membrane Sci., 445, 160–169 (2013).
P. Dydo, “Transport model for boric acid, monoborate and borate complexes across thin-fi lm composite reverse osmosis membrane,” Desalination, 311, 69–79 (2013).
L. Han, J. Tian, C. Liu. et al., “Infl uence of pH and NaCl concentration on boron rejection during nanofiltration,” Sept. Pur. Techn., 261, 118248 (2021).
O. Labban, C. Li, T. H. Chong, et al., “Fundamentals of low-pressure nanofiltration: Membrane characterization, modeling, and understanding the multi-ionic interactions in water softening,” J. Membrane Sci., 521, 18–32 (2017).
C.-H. Chu, C. Wang, H.-F. **ao, et al., “Separation of ions with equivalent and similar molecular weights by nanofiltration: sodium chloride and sodium acetate as an example,” Sept. Pur. Techn., 250, 117199 (2020).
K. Meschke, N. Hansen, R. Hofmann, et al., “Influence of process parameters on separation performance of strategic elements by polymeric nanofiltration membranes,” Sept. Pur. Techn., 235, 116186 (2020).
B. Bruggen and J. Kim, “Nanofiltration of aqueous solutions: recent developments and progresses,” in: Advanced Materials for Membrane Preparation, Bentham Sci. Publisher, Netherlands (2012), pp. 228–247.
Membrane Product Catalog. Euro Clear Kft. Water Treatment Equipment Manufacturer and Wholesale Distributor (2020).
V. A. Vinnitskii, A. S. Chugunov, and M. V. Ershov, “Influence of retentate flow on membrane separation of binary solutions of sodium, magnesium and calcium chlorides,” ChemChemTech [Izv. Vys. Ucheb. Zaved., Ser. Khim. Khim. Tekhnol.] 64(10), 46–55 (2021).
A. S. Chugunov and V. A. Vinnitskii, “Local systems for implementing the concept of organizing separate collection and processing of liquid radioactive media of nuclear power plants with VVER,” Radioakt. Otkh., No. 4(17), 44–56 (2021).
Author information
Authors and Affiliations
Corresponding author
Additional information
Translated from Atomnaya Énergiya, Vol. 132, No. 3, pp. 158–162, March, 2022.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Vinnitskii, V.A., Solonovich, G.I. & Chugunov, A.S. Low-Pressure Nanofiltration Systems for Decreasing Boric Acid Ingress Into Drain Water in NPP with VVER. At Energy 132, 163–167 (2022). https://doi.org/10.1007/s10512-023-00920-9
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10512-023-00920-9