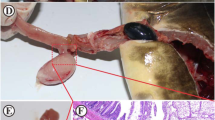
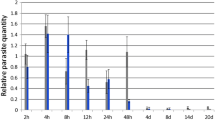
Abstract
Dactylogyrosis is caused by more than 900 reported species of Dactylogyrus (gill fluke), mostly in Cyprinoidei fish leading to low to severe mortality in different culture systems and also in rivers/lakes. Dactylogyrus has been reported worldwide where Cyprinoidei is endemic. In recent years, incidents of infection and infectivity of different species of Dactylogyrus have increased many-fold due to several predisposing factors like intensification of culture, environmental quality, climatic change, poor quality fish seed, etc. These parasites are host choosy, either generalist or specialist. Dactylogyrus infection causes major changes in the anatomy and physiology of gill function. Necrosis, hyperplasia and hypertrophy of gill lamellae are the common pathologies observed during infection, which in turn invites other secondary pathogens leading to heavy mortalities. Currently, very few drugs or chemicals are available for controlling dactylogyrosis. Very few attempts have been made to develop vaccines against this important parasite. Here, we review the innate and adaptive immune responses modulated by Dactylogyrus infection and a hypothetical immunological model has been proposed to understand host-pathogen interaction based on our findings. The information collected and analysed in this review on the biology and host-pathogen interaction will help in the development of prophylactic and other sustainable management measures to reduce the loss associated with dactylogyrosis.




Similar content being viewed by others
Code Availability
As a review, this section may not be applicable.
References
Abbas AK, Lichtman AH, Pober JS (1998) In Immunologia cellulare emolecolare, 9th edn. Piccin Elsevier Srl, Milan, Italy
Achmerow AK (1952) New species of monogenetic trematodes of the fishes of Amur River. Parazitologicheskii Sbornik Zoologicheskogo Institut Akademii Nauk SSSR 14:181–212
Amos SO, Eyiseh TE, Michael ET (2018) Parasitic infection and prevalence in Clarias gariepinus in Lake Gerio, Yola. Adamawa state. MOJ Anat Physiol 5(6):376–381
Bauer ON, Musselius VA, Strelvok Y (1973) Diseases of pond fishes. Israel Program for Scientific Translation. Translated from Russian, Israel Program for Scientific Translations. Jerusalem
Benovics M, Desdevises Y, Šanda R, Vukić J, Scheifler M, Doadrio I, Sousa-Santos C, Simková A (2020) High diversity of fish ectoparasitic monogeneans (Dactylogyrus) in the Iberian Peninsula: a case of adaptive radiation? Parasitology 147:418–430. https://doi.org/10.1017/s0031182020000050
Benovics M, Desdevises Y, Vukić J, Šanda R, Šimková A (2018) The phylogenetic relationships and species richness of host-specific Dactylogyrus parasites shaped by the biogeography of Balkan cyprinids. Sci Rep 8:1–18. https://doi.org/10.1038/s41598-018-31382-w
Benovics M, Kičinjaová ML, Šimková A (2017) The phylogenetic position of the enigmatic Balkan Aulopyge huegelii (Teleostei: Cyprinidae) from the perspective of host-specific Dactylogyrus parasites (Monogenea), with a description of Dactylogyrus omenti n. sp. Parasit Vectors 10:1–13. https://doi.org/10.1186/s13071-017-2491-z
Bethony J, Loukas A, Smout M, Brooker S, Mendez S, Plieskatt J, Goud G, Bottazzi ME, Zhan B, Wang Y, Williamson A (2005) Antibodies against a secreted protein from hookworm larvae reduce the intensity of hookworm infection in humans and vaccinated laboratory animals. FASEB J 19:1743–1745. https://doi.org/10.1096/fj.05-3936fje
Borji H, Naghibi A, Nasiri MR, Ahmadi A (2012) Identification of Dactylogyrus spp. and other parasites of common carp in northeast of Iran. J Parasit Dis 36:234–238. https://doi.org/10.1007/s12639-012-0115-2
Buchmann K (1993) A note on the humoral immune response of infected Anguilla anguilla against the gill monogenean Pseudodactylogyrus bini. Fish Shellfish Immunol 3:397–399. https://doi.org/10.1006/fsim.1993.1039
Buchmann K (1988) Epidemiology of pseudodactylogyrosis in an intensive eel-culture system. Dis Aquat Org 5:81–85. https://doi.org/10.3354/dao005081
Buchmann K, Bresciani J (1997) Parasitic infections in pond-reared rainbow trout Oncorhynchus mykiss in Denmark. Dis Aquat Org 28:125–138. https://doi.org/10.3354/dao028125
Burkart MA, Clark TG, Dickerson HW (1990) Immunization of channel catfish, Ictalurus punctatus Rafinesque, against Ichthyophthirius multifiliis (Fouquet): killed versus live vaccines. J Fish Dis 13:401–410. https://doi.org/10.1111/j.1365-2761.1990.tb00799.x
Bychowsky BE (1957) Monogenetic trematodes, their systematics and phylogeny. Academy of Sciences, Moscow
Chanda M, Paul M, Maity J, Dash G, Gupta SS, Patra BC (2011) Ornamental fish goldfish, Carassius auratus and related parasites in three districts of West Bengal, India. Chron Young Sci 2:51–54. https://doi.org/10.4103/2229-5186.79351
Cone D, Eurell T, Beasley V (1994) A report of Dactylogyrus amphibothrium (Monogenea) on the gills of European ruffe in western Lake Superior. J Parasitol 80:476–478. https://doi.org/10.2307/3283421
Cone DK, Burt MDB (1981) The invasion route of the gill parasite Urocleidus adspectus Mueller, 1936 (Monogenea: Ancyrocephalinae). Can J Zool 59:2166–2171. https://doi.org/10.1139/z81-293
Daghigh Roohi J, Dalimi A, Pourkazemi M, Ghasemi M, Shamsi S (2020) Morphometric and molecular characterization of Dactylogyrus lamellatus isolated from farmed grass carp, Ctenopharyngodon idella (Valenciennes, 1844), in Guilan province. Iran J Fish Sci 19:1051–1061
Dash P, Kar B, Mishra A, Sahoo PK (2014) Effect of Dactylogyrus catlaius (Jain 1961) infection in Labeo rohita (Hamilton 1822): innate immune responses and expression profile of some immune related genes. Indian J Exp Biol 52:267–280
Dash P, Sahoo PK (2015) Ontogeny, tissue distribution and expression analysis of IgZ in rohu, Labeo rohita in response to various stimuli. Vet Immunol Immunopathol 166:70–78. https://doi.org/10.1016/j.vetimm.2015.05.007
Desdevises Y, Morand S, Jousson O, Legendre P (2002) Coevolution between Lamellodiscus (Monogenea: Diplectanidae) and Sparidae (Teleostei): the study of a complex host-parasite system. Evolution 56:2459–2471. https://doi.org/10.1554/0014-3820(2002)056[2459:cblmda]2.0.co;2
Dewi RR, Fadhilla A (2018) The prevalence of parasites in ornamental fish from fish market in Medan. IOP Conference Series: Environ Earth Sci 122:012110. https://doi.org/10.1088/1755-1315/122/1/012110
Diesing KM (1850) Systema helminthum, vol 1. W. Braumüller, Vindobonae, p 679
Dogiel V, Petruschewsky G, Poljansky G (1958) Die Gudlagen der Parasitologie der Fische (Fundamentals of Fish Parasitology). Verlag der Leningrader Staatlichen Universität
Dove AD, Ernst I (1998) Concurrent invaders—four exotic species of Monogenea now established on exotic freshwater fishes in Australia. Int J Parasitol 28:1755–1764. https://doi.org/10.1016/s0020-7519(98)00134-9
El Gharbi S, Renaud F, Lambert AD (1992) Dactylogyrids (Platyhelminthes: Monogenea) of Barbus spp. (Teleostei: Cyprinidae) from the Iberian Peninsula. Res Rev in Parasitol 52:103–116
Farhana H, Arabinda D, Ajmal H, Parthapratim C, Sundaray JK (2018) Mass mortality associated with Dactylogyrus infection in farmed long whiskers catfish, Mystus gulio. J Exp Zool 21:227–231
Feist S, Longshaw M (2008) Histopathology of fish parasite infections-importance for populations. J Fish Biol 73:2143–2160. https://doi.org/10.1111/j.1095-8649.2008.02060.x
Findly RC, Zhao X, Noe J, Camus AC, Dickerson HW (2013) B cell memory following infection and challenge of channel catfish with Ichthyophthirius multifiliis. Dev Comp Immunol 39:302–311. https://doi.org/10.1016/j.dci.2012.08.007
Fu YW, Wang B, Zhang QZ, Xu DH, Lin DJ, Yang XY, Zhu SQ, Pan JY, Deng Q, Liu YM, Zhou SY (2017) Combined effects of Chinese medicine feed and ginger extract bath on co-infection of Ichthyophthirius multifiliis and Dactylogyrus ctenopharyngodonid in grass carp. Parasitol Res 116:2017–2025. https://doi.org/10.1007/s00436-017-5507-x
Futuyma DJ, Moreno G (1988) The evolution of ecological specialization. Annu Rev Ecol Evol Syst 19:207–233
Gado MS, Mahfouz NB, Moustafa EM, Lolo EE (2017) Prevalence of some ectoparasitic diseases in African catfish (Clarias gariepinus) at Kafr El-Sheikh governorate. Int J Fish Aquat Stud 5:576–583
Gharbi K, Matthews L, Bron J, Roberts R, Tinch A, Stear M (2015) The control of sea lice in Atlantic salmon by selective breeding. J R Soc Interface 12(110):20150574. https://doi.org/10.1098/rsif.2015.0574
Ghiorghiasa R, Miron M, Lupu EI, Miron L (2015) Winter parasitic co-infections in common carp (Cyprinus carpio) from a polyculture system. Lucr Stiinţ 58:197–202
Gibson DI, Timofeeva TA, Gerasev PIA (1996) Catalogue of the nominal species of the monogenean genus Dactylogyrus Diesing, 1850 and their host genera. Syst Parasitol 35:3–48. https://doi.org/10.1007/bf00012180
Gonzalez-Lanza MC, Alvarez-Pellitero MP (1982) Description and population dynamics of Dactylogyrus legionensis n. sp. from Barbus barbus bocagei Steind. J Helminthol 56:263–273. https://doi.org/10.1017/s0022149x00034647
Guegan JF, Agnèse JF (1991) Parasite evolutionary events inferred from host phylogeny: the case of Labeo species (Teleostei, Cyprinidae) and their dactylogyrid parasites (Monogenea, Dactylogyridae). Can J Zool 69:595–603. https://doi.org/10.1139/z91-089
Guegan JF, Lambert A (1990) Twelve new species of dactylogyrids (Platyhelminthes, Monogenea) from West African barbels (Teleostei, Cyprinidae), with some biogeographical implications. Syst Parasitol 17:153–181. https://doi.org/10.1007/bf00009552
Gupta G, Chatterjee A, Kumar M, Sardar P, Varghese T, Srivastava PP, Gupta S (2020) Efficacy of single and multiple doses of fenbendazole against gill parasites (Dactylogyrus sp.) of Labeo rohita (Hamilton, 1822) and its physio-metabolic effects on the fish. Aquac Res 51:1190–1199. https://doi.org/10.1111/are.14470
Gusev AV (1962) Class Monogenoidea (Beneden) Byjkovskii, 1937. Monogenetic flukes. In: Bykhovskaya-Pavlovslaya IE et al (eds) Key to the parasites of freshwater fish of the USSR. Akademiya Nauk SSSR, Moscow-Leningrad, pp 770
Gusev AV (1985) Order Dactylogyridea. In: Gusev AV, Dubinina MN, Pugachev ON, Raykova EV, Hotenovskiy IA, Ergens R (eds) In Keys to the parasites of freshwater fish of the USSR fauna. Metazoan parasites, vol 2. Nauka, Leningrad, pp 15–25
Hao B, Liu GL, Hu XG, Wang GX (2012) Bioassay-guided isolation and identification of active compounds from Semen pharbitidis against Dactylogyrus intermedius (Monogenea) in goldfish (Carassius auratus). Vet Parasitol 187:452–458. https://doi.org/10.1016/j.vetpar.2012.01.023
Hossain MD, Hossain MK, Rahman MH, Akter A, Khanom DA (2008) Prevalence of ectoparasites of carp fingerlings at Santaher, Bogra. Univ J Zool Rajshahi Univ 27:17–19. https://doi.org/10.3329/ujzru.v27i0.1947
Humphery-Smith I (1989) The evolution of phylogenetic specificity among parasitic organisms. Parasitol Today 5:385–387. https://doi.org/10.1016/0169-4758(89)90303-7
Ilgova J, Jedlickova L, Dvorakova H, Benovics M, Mikes L, Janda L, Vorel J, Roudnicky P, Potesil D, Zdrahal Z, Gelnar MA (2017) Novel type I cystatin of parasite origin with atypical legumain-binding domain. Sci Rep 7:1–12. https://doi.org/10.1038/s41598-017-17598-2
Ilgova J, Kavanova L, Matiaskova K, Salat J, Kasny M (2020) Effect of cysteine peptidase inhibitor of Eudiplozoon nipponicum (Monogenea) on cytokine expression of macrophages in vitro. Mol Biochem Parasitol 235:111248. https://doi.org/10.1016/j.molbiopara.2019.111248
Kaur N, Kumar R, Kamilya D (2018) Modulation of systemic and mucosal immune responses of Catla catla (Hamilton, 1822) experimentally challenged with gill monogeneans. Fish Shellfish Immunol 74:567–572. https://doi.org/10.1016/j.fsi.2018.01.026
Kearn GC (2011) Monogeneans-the ultimate fish parasites. Monogenea 58:28–32
Kim KH, Hwang YJ, Cho JB, Park SI (2000) Immunization of cultured juvenile rockfish Sebastes schlegeli against Microcotyle sebastis (Monogenea). Dis Aquat Org 40:29–32. https://doi.org/10.3354/dao040029
King SD, Cone DK (2008) Persistence of Dactylogyrus eucalius (Monogenea: Dactylogyridae) on the short-lived host Culaea inconstans (Pisces: Gasterosteiformes). J Parasitol 94:973–975. https://doi.org/10.1645/ge-1495.1
Kishimori JM, Takemura A, Leong JAC (2015) Neobenedenia melleni-specific antibodies are associated with protection after continuous exposure in Mozambique tilapia. J Immunol Res 1-6. https://doi.org/10.1155/2015/635387
Koyun M (2011) Seasonal distribution and ecology of some Dactylogyrus species infecting Alburnus alburnus and Carassius carassius (Osteichthyes: Cyprinidae) from Porsuk River, Turkey. Afr J Biotechnol 10:1154–1159
Krasnovyd V, Vetesník L, Simková A (2020) Distribution of host-specific parasites in hybrids of phylogenetically related fish: the effects of genotype frequency and maternal ancestry? Parasit Vectors 13:1–11. https://doi.org/10.1186/s13071-020-04271-3
Kritsky DC, Heckmann R (2002) Species of Dactylogyrus (Monogenoidea: Dactylogyridae) and Trichodina mutabilis (Ciliata) infesting koi carp, Cyprinus carpio, during mass mortality at a commercial rearing facility in Utah, USA. Comp Parasitol 69:217–218. https://doi.org/10.1654/1525-2647(2002)069[0217:sodmda]2.0.co;2
Kuang J, Yan X, Genders AJ, Granata C, Bishop DJ (2018) An overview of technical considerations when using quantitative real-time PCR analysis of gene expression in human exercise research. PloS One 13:e0196438. https://doi.org/10.1371/journal.pone.0196438
Kumar S, Raman RP, Prasad KP, Srivastava PP, Kumar S, Rajendran KV (2017) Modulation of innate immune responses and induction of oxidative stress biomarkers in Pangasianodon hypophthalmus following an experimental infection with dactylogyrid monogeneans. Fish Shellfish Immunol 63:334–343. https://doi.org/10.1016/j.fsi.2017.02.033
Lari E, Adams RV, Cone DK, Goater CP, Pyle GG (2016) Dactylogyrus olfactorius n. sp. (Monogenea, Dactylogyridae) from the olfactory chamber of the fathead minnow, Pimephales promelas Rafinesque (Cyprinidae). Syst Parasitol 93:575–581. https://doi.org/10.1007/s11230-016-9649-5
Lianda N, Fahrimal Y, Daud R, Rusli R, Aliza D, Adam M (2015) Identification of parasites on Nile tilapia (Oreochromis niloticus) fish collected from Barabung irrigation Darussalam Aceh Besar. J Medika Veterinaria 9(2):3–10
Li JP, Fu YW, Zhang QZ, Xu DH, Liu YM, Zhou SY, Lin DJ (2018) Grass carp which survive Dactylogyrus ctenopharyngodonid infection also gain partial immunity against Ichthyophthirius multifiliis. Dis Aquat Org 129:63–70. https://doi.org/10.3354/dao03223
Liu GL, Hu Y, Chen XH, Wang GX, Ling F (2016) Synthesis and anthelmintic activity of coumarin–imidazole hybrid derivatives against Dactylogyrus intermedius in goldfish. Bioorg Med Chem Lett 26:5039–5043. https://doi.org/10.1016/j.bmcl.2016.08.090
Liu GL, Liu L, Hu Y, Wang GX (2021) Evaluation of the antiparasitic activity of coumarin derivatives against Dactylogyrus intermedius in goldfish (Carassius auratus). Aquaculture 533:736069. https://doi.org/10.1016/j.aquaculture.2020.736069
Long S, Yu K (1958) Parasitic worms of Lake Taihu fishes. I. Monogenea Dactylogyrus (Diesing, Dactylogyridae). Description of Dactylogyrus-two new species from Aristichthys nobilis. Transactions of Shanghai University. Nat Sci 8–10 (In Chinese)
Lu C, Ling F, Ji J, Kang YJ, Wang GX (2013) Expression of immune-related genes in goldfish gills induced by Dactylogyrus intermedius infections. Fish Shellfish Immunol 34:372–377. https://doi.org/10.1016/j.fsi.2012.11.004
Lumanlan SC, Albaladejo JD, Bondad-Reartaso MG, Arthur JR (2004) Fresh-water fishes imported into the Philippines. In: Shariff M, Subashinghe RP, Arthur JR (eds) Diseases in Asian Aquaculture 1. Asian Fisheries Society, Philippines
Luo F, Ling F, Geng T, Liu G, Jiang C, Wang G (2016) Systematic screening identifies synergistic combinations of traditional Chinese medicines and ingredients against Dactylogyrus infections using a goldfish model. Aquaculture 459:198–202. https://doi.org/10.1016/j.aquaculture.2016.03.045
Molnar K (1971) Studies on gill parasitosis of the grass carp (Ctenopharyngodon idella) caused by Dactylogyrus lamellatus Achmerov, 1952. Acta Vet Hung 21:267–289
Monni G, Cognetti-Varriale AM (2002) Antigenicity of Pseudodactylogyrus anguillae and P. bini (Monogenea) in the European eel (Anguilla anguilla, L.) under different oxygenation conditions. Fish Shellfish Immunol 13:125–131. https://doi.org/10.1006/fsim.2001.0387
Mozhdeganloo Z, Heidarpour M (2014) Oxidative stress in the gill tissues of goldfishes (Carassius auratus) parasitized by Dactylogyrus spp. J Parasit Dis 38:269–272
Mueller JF, VanCleave HJ (1932) Parasites of Oneida Lake fishes. Part II. Descriptions of new species and some general taxonomic considerations, especially concerning the trematode family Heterophyidae. Bulletin of the New York State College of Forestry at Syracuse University, 5: [Roosevelt Wildlife Annals, 3], pp 79–137
Murray RKDK, Rranne PAM, Rodwell VW (1990) Harper’s Biochemistry. Harper’s Biochemistry Publisher, Norwalk. https://doi.org/10.1007/s12639-013-0239-z
Neguenga DA (1988) Note on infestation of Oreochromis niloticus with Trichodina sp. and Dactylogyrus sp. In: Pullin RSV, Bhukaswan T, Tonguthai K, Maclean JL (eds) The second international symposium on tilapia in aquaculture, vol 15. ICLARM Con Proc, pp 117–119
Nybelin O (1924) Dactylogyrus vastator n. sp. Arkiv för Zoologi 16(28):1–2
Norton DA, Carpenter MA (1998) Mistletoes as parasites: host specificity and speciation. Trends Ecol Evol 13:101–105. https://doi.org/10.1016/s0169-5347(97)01243-3
Ogawa K (2002) Impacts of diclidophorid monogenean infections on fisheries in Japan. Int J Parasitol 32:373–380. https://doi.org/10.1016/s0020-7519(01)00338-1
Ortega C, Mancera G, Enríquez R, Vargas A, Martínez S, Fajardo R, Avendaño-Herrera R, Navarrete MJ, Romero A (2016) First identification of Francisella noatunensis subsp. orientalis causing mortality in Mexican tilapia Oreochromis spp. Dis Aquat Org 120:205–215. https://doi.org/10.3354/dao02999
Özer A (2002) Co-existence of Dactylogyrus anchoratus Dujardin, 1845 and D. extensus Mueller & Van Cleave, 1932 (Monogenea), parasites of common carp (Cyprinus carpio). Helminthologia 39:45–50
Özer A, Öztürk T (2005) Dactylogyrus cornu Linstow, 1878 (Monogenea) infestations on Vimba (Vimba vimba tenella (Nordmann, 1840)) caught in the Sinop region of Turkey in relation to the host factors. Turkish J Vet Anim Sci 29:1119–1123
Öztürk MO, Altunel FN (2006) Occurrence of Dactylogyrus infection linked to seasonal changes and host fish size on four cyprinid fishes in Lake Manyas, Turkey. Acta Zool Acad Sci Hung 52:407–415
Panjvini F, Abarghuei S, Khara H, Parashkoh HM (2016) Parasitic infection alters haematology and immunity parameters of common carp, Cyprinus carpio, Linnaeus, 1758. J Parasit Dis 40:1540–1543. https://doi.org/10.1007/s12639-015-0723-8
Paperna I (1964) Competitive exclusion of Dactylogyrus extensus by Dactylogyrus vastator (Trematoda, Monogenea) on the gills of reared carp. J Parasitol 50:94–98. https://doi.org/10.2307/3276037
Paperna I (1963b) Dynamics of Dactylogyrus vastator Nybelin (Monogenea) populations on the gills of carp fry in fish ponds. Bamidgeh 15:31–50
Paperna I (1963a) Some observations on the biology and ecology of Dactylogyrus vastator in Israel. Bamidgeh 15:8–28
Park JK, Kim KH, Kang S, Kim W, Eom KS, Littlewood DTJ (2007) A common origin of complex life cycles in parasitic flatworms: evidence from the complete mitochondrial genome of Microcotyle sebastis (Monogenea: Platyhelminthes). BMC Evol Biol 7:1–13
Pathak P (2020) Study of parasitic effect of Dactylogyrus sp. on Indian major carps. J Emerg Technol Innov Res 7:2557–2566
Paul A, Baral B, Parida S, Naveen ND, Sahoo PK (2022) Mixed infections in tropical freshwater fish culture systems: a potential emerging threat for successful aquaculture. Aquac Asia 26:20–24
Paul A, Mohanty J, Rajendran KV, Tripathi G, Sahoo PK (2021) First report of Dactylogyrus scorpius infection in Indian major carp, Labeo rohita from India: host specificity and kinetics of immune gene expression in gills. Aquaculture 536:736453. https://doi.org/10.1016/j.aquaculture.2021.736453
Paul A, Sahoo PK (2022) Prevalence, molecular identification and host preference of Dactylogyrus, an ectoparasite in Indian major carps. J Environ Biol 43:536–543. https://doi.org/10.22438/jeb/43/4/mrn-2017
Pichichero ME (2009) Booster vaccinations: can immunologic memory outpace disease pathogenesis? Pediatrics 124:1633–1641. https://doi.org/10.1542/peds.2008-3645
Pikulkaew S, Meeyam T, Banlunara W (2009) The outbreak of koi herpesvirus (KHV) in koi (Cyprinus carpio koi) from Chiang Mai Province, Thailand. Thai J Vet. Med 39:53–58
Pikulkaew S, Phatwan K, Banlunara W, Intanon M, Bernard JK (2020) First evidence of carp edema virus infection of koi Cyprinus carpio in Chiang Mai Province, Thailand. Viruses 12:1400. https://doi.org/10.3390/v12121400
Pojmanska T (1995) Seasonal dynamics of occurrence and reproduction of some parasites in four cyprinid fish cultured in ponds. II. Monogenea. Acta Parasitol 40:79–84
Pretto T, Manfrin A, Ceolin C, Dalla Pozza M, Zelco S, Quartesan R, Abbadi M, Panzarin V, Toffan A (2013) First isolation of koi herpes virus (KHV) in Italy from imported koi (Cyprinus carpio koi). Bull Eur Assoc Fish Pathol 33:126–133
Prost M (1963) Investigations on the development and pathogenicity of Dactylogyrus anchoratus (Duj., 1845) and D. extensus Mueller et v. Cleave, 1932 for breeding carps. Acta Parasitol Pol 11:17–47
Quyet PV, Yilmaz BH, Yildiz HY (2021) In vitro antiparasitic activity of ginger (Zingiber officinale) bulb and pomegranate (Punica granatum) peel against Monogenean fish parasite., Dactylogyrus sp. Acta Aquat Turc 17:56–63. https://doi.org/10.22392/actaquatr.751913
Rana N, Das BK (2016) Effect of short-term infection of Paradactylogyrus catlaius on selected haematological and non-specific immune parameters in Catla catla (Hamilton, 1822). Indian J Fish 63:70–75. https://doi.org/10.21077/ijf.2016.63.2.51159-09
Rastiannasab A, Afsharmanesh S, Rahimi R, Sharifian I (2016) Alternations in the liver enzymatic activity of common carp, Cyprinus carpio in response to parasites, Dactylogyrus spp. and Gyrodactylus spp. J Parasit Dis 40:1146–1149. https://doi.org/10.1007/s12639-014-0638-9
Riveau G, Deplanque D, Remoue F, Schacht AM, Vodougnon H, Capron M, Thiry M, Martial J, Libersa C, Capron A (2012) Safety and immunogenicity of rSh28GST antigen in humans: phase 1 randomized clinical study of a vaccine candidate against urinary schistosomiasis. PLOS Negl Trop Dis 6:e1704. https://doi.org/10.1371/journal.pntd.0001704
Rubio-Godoy M, Tinsley RC (2002) Trickle and single infection with Discocotyle sagittata (Monogenea: Polyopisthocotylea): effect of exposure mode on parasite abundance and development. Folia Parasitol 49:269–278. https://doi.org/10.14411/fp.2002.051
Sahoo PK, Paul A, Sahoo MK, Pattanayak S, Rajesh Kumar P, Das BK (2020) Incidences of infectious diseases in freshwater aquaculture farms of eastern India: a passive surveillance based study from 2014-2018. J Aquac Res Dev 11:1–5. https://doi.org/10.35248/2155-9546.20.11.579
Sahoo PK, Mishra M, Mohapatra A, Parida S, Mohanty J (2021) Vaccination approach to prevent Argulus siamensis infection-success, challenges and preparedness. Fish Shellfish Immunol Rep 2:100023. https://doi.org/10.1016/j.fsirep.2021.100023
Sarig S (1971) The prevention and treatment of diseases of warmwater fish under subtropical conditions, with special emphasis on intensive fish farming. T.F.H. Publications Inc., Jersey City, N.J., p 127
Schmahl G, Mehlhorn H (1985) Treatment of fish parasites. 1. Praziquantel effective against Monogenea (Dactylogyrus vastator, Dactylogyrus extensus, Diplozoon paradoxum). Zeitschrift für Parasitenkunde 71:727–737
Schmahl G, Mehlhorn H, Haberkorn A (1988) Sym. triazinone (toltrazuril) effective against fish-parasitizing Monogenea. Parasitol Res 75:67–68. https://doi.org/10.1007/bf00931193
Sharif M, Vijiarungam AF (1986) The occurrence of parasites at the fish breeding station in peninsular Malaysia and their control. Penerbit Universiti Pertanian Malaysia, pp 68–73
Simkova A, Desdevise Y, Gelnar M, Morand S (2001) Morphometric correlates of host specificity in Dactylogyrus species (Monogenea) parasites of European Cyprinid fish. Parasitology 123:169–177. https://doi.org/10.1017/s0031182001008241
Simkova A, Matejusová I, Cunningham CO (2006b) A molecular phylogeny of the Dactylogyridae sensu Kritsky & Boeger (1989) (Monogenea) based on the D1-D3 domains of large subunit rDNA. Parasitology 133:43–53. https://doi.org/10.1017/s0031182006009942
Simkova A, Morand S (2008) Co-evolutionary patterns in congeneric monogeneans: a review of Dactylogyrus species and their Cyprinoidei hosts. J Fish Biol 7:2210–2227. https://doi.org/10.1111/j.1095-8649.2008.02064.x
Simkova A, Morand S, Jobet E, Gelnar M, Verneau (2004) Molecular phylogeny of congeneric monogenean parasites (Dactylogyrus): a case of intrahost speciation. Evolution 58: 1001-1018. https://doi.org/10.1554/03-606
Simkova A, Verneau O, Gelnar M, Morand S (2006a) Specificity and specialization of congeneric monogeneans parasitizing cyprinid fish. Evolution 60:1023–1037. https://doi.org/10.1554/05-521.1
Smyth JD, Halton DW (1983) The physiology of trematodes. CUP Archive
Sood N, Pradhan PK, Verma DK, Yadav MK, Dev AK, Swaminathan TR, Sood NK (2018) Candidatus Actinochlamydia pangasiae sp. nov. (Chlamydiales, Actinochlamydiaceae), a bacterium associated with epitheliocystis in Pangasianodon hypophthalmus. J Fish Dis 41:281–290. https://doi.org/10.1111/jfd.12711
Soto E, Hawke JP, Fernandez D, Morales J (2009) Francisella sp., an emerging pathogen of tilapia, Oreochromis niloticus (L.), in Costa Rica. J Fish Dis 32:713–722. https://doi.org/10.1111/j.1365-2761.2009.01070.x
Swaminathan TR, Kumar R, Dharmaratnam A, Basheer VS, Sood N, Pradhan PK, Sanil NK, Vijayagopal P, Jena JK (2016) Emergence of carp edema virus in cultured ornamental koi carp, Cyprinus carpio koi, in India. J Gen Virol 97:3392–3399. https://doi.org/10.1099/jgv.0.000649
Tancredo KR, Marchiori NDC, Pereira SA, Martins ML (2019) Toxicity of formalin for fingerlings of Cyprinus carpio var. koi and in vitro efficacy against Dactylogyrus minutus Kulwièc, 1927 (Monogenea: Dactylogyridae). J Parasit Dis 43:46–53. https://doi.org/10.1007/s12639-018-1056-1
Thilakaratne IDSIP, Rajapaksha G, Hewakopara A, Rajapakse RPVJ, Faizal ACM (2003) Parasitic infections in freshwater ornamental fish in Sri Lanka. Dis Aquat Org 54:157–162. https://doi.org/10.3354/dao054157
Thoney DA, Hargis WJ Jr (1991) Monogenea (Platyhelminthes) as hazards for fish in confinement. Annu Rev Fish Dis 1:133–153. https://doi.org/10.1016/0959-8030(91)90027-h
Tu X, Liu L, Qi X, Chen W, Wang G, Ling F (2016) Characterization of toll-like receptor gene expression in goldfish (Carassius auratus) during Dactylogyrus intermedius infection. Dev Comp Immunol 63:78–83. https://doi.org/10.1016/j.dci.2016.05.019
Vladimirov VL (1971) The immunity of fishes in the case of dactylogyrosis. Parasitologiya 5:51–58
von Gersdorff JL, Kania PW, Rasmussen KJ, Mattsson AH, Schmidt J, Al-Jubury A, Sander A, Salanti A, Buchmann K (2017) Rainbow trout (Oncorhynchus mykiss) immune response towards a recombinant vaccine targeting the parasitic ciliate Ichthyophthirius multifiliis. J Fish Dis 40:1815–1821. https://doi.org/10.1111/jfd.12653
Wang GX, Han J, Feng TT, Li FY, Zhu B (2009) Bioassay-guided isolation and identification of active compounds from Fructus arctii against Dactylogyrus intermedius (Monogenea) in goldfish (Carassius auratus). Parasitol Res 106:247–255. https://doi.org/10.1007/s00436-009-1659-7
Wang GX, Jiang DX, Li J, Han J, Liu YT, Liu XL (2010a) Anthelmintic activity of steroidal saponins from Dioscorea zingiberensis CH Wright against Dactylogyrus intermedius (Monogenea) in goldfish (Carassius auratus). Parasitol Res 107:1365–1371. https://doi.org/10.1007/s00436-010-2010-z
Wang GX, Zhou Z, Jiang DX, Han J, Wang JF, Zhao LW, Li J (2010b) In vivo anthelmintic activity of five alkaloids from Macleaya microcarpa (Maxim) Fedde against Dactylogyrus intermedius in Carassius auratus. Vet Parasitol 171:305–313. https://doi.org/10.1016/j.vetpar.2010.03.032
Woo PT, Leatherland JF, Bruno DW (2006) Fish diseases and disorders. In: Protozoan and Metazoan Infections. CAB International, Oxon, UK, p 3
World Organisation for Animal Health (OIE) (2018) Manual of diagnostic tests for aquatic animals, Chapter 2.3.3. Infection with Gyrodactylus salaris. OIE, Paris, France. https://www.woah.org/fileadmin/Home/eng/Health_standards/aahm/current/chapitre_gyrodactylus_salaris.pdf
Yang BJ, Zou H, Zhou S, Wu SG, Wang GT, Li WX (2016) Seasonal dynamics and spatial distribution of the Dactylogyrus species on the gills of grass carp (Ctenopharyngodon idellus) from a fish pond in Wuhan, China. J Parasitol Res 102:507–513. https://doi.org/10.1645/15-931
Yang JL, Chen HC (2003) Serum metabolic enzyme activities and hepatocyte ultrastructure of common carp after gallium exposure. Zool Stud 42:455–461
Yang Z, Tan X, Zhang Z, Han J, Qu S, Liu T, Wang G (2022) Ononin: a candidate anti-parasitic drug isolated from Spatholobi caulis against infections of Dactylogyrus intermedius (Monogenea). Parasitol Int 88:102535. https://doi.org/10.1016/j.parint.2021.102535
Yusoff SFM, Christianus A, Matori FUAD, Talba MA, Hamid NH, Hayati R (2020) The impact of natural co-infection of Dactylogyrus spp. and Aeromonas hydrophila on behavioural, clinical, and histopathological changes of striped catfish, Pangasianodon hypophthalmus (Sauvage, 1878): a case study. J Sustain Sci Manag 15:74–82
Zahner H, Hobom G, Stim S (1995) The microfilarial sheath and its proteins. Parasitol Today 11:116–120. https://doi.org/10.1016/0169-4758(95)80173-1
Zhang C, Li DL, Chi C, Ling F, Wang GX (2015) Dactylogyrus intermedius parasitism enhances Flavobacterium columnare invasion and alters immune-related gene expression in Carassius auratus. Dis Aquat Org 116:11–21. https://doi.org/10.3354/dao02902
Zhang XP, Li WX, Ai TS, Zou H, Wu SG, Wang GT (2014) The efficacy of four common anthelmintic drugs and traditional Chinese medicinal plant extracts to control Dactylogyrus vastator (Monogenea). Aquaculture 420:302–307. https://doi.org/10.1016/j.aquaculture.2013.09.022
Acknowledgements
The authors wish to thank the Director, ICAR-Central Institute of Freshwater Aquaculture, Kausalyaganga, Bhubaneswar, and ICAR-Central Institute of Fisheries Education, Mumbai, India for providing necessary facilities during this study. Funding support by DoF, MFAHD, Govt. of India under the National Surveillance Programme for Aquatic Animal Diseases is duly acknowledged. The authors are also thankful to Ms. Banya Kar for the English editing of the manuscript.
Author information
Authors and Affiliations
Contributions
AP: conceptualization, investigation, data curation, writing—original draft; PKS: conceptualization, validation, data curation, writing—review and editing
Corresponding author
Ethics declarations
Ethics Approval
As a review, this section may not be applicable.
Consent to Participate
All authors contributed to the study including conception and collections. All authors read and approved the final manuscript.
Consent for Publication
All authors gave their consent for research publication.
Competing Interests
The authors declare no competing interests.
Additional information
Handling Editor: Gavin Burnell
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Paul, A., Sahoo, P.K. A host choosy gill parasite (Dactylogyrus spp.) in fish: an insight into host-parasite interaction for develo** control strategies. Aquacult Int (2024). https://doi.org/10.1007/s10499-024-01393-8
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10499-024-01393-8