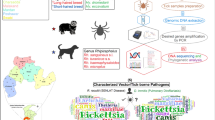
Abstract
An epidemiological study was performed to determine the role of dogs and ticks infesting dogs in the transmission of Q fever in humans and animals from April 2019 to March 2020 in the northeastern hill states of India. In total, 245 pet and stray dogs irrespective of age or sex were sampled, without specific inclusion or exclusion criteria. In total, 478 ticks belonging to three species were detected, namely Rhipicephalus sanguineus, Rhipicephalus (Boophilus) microplus and Hyalomma anatolicum anatolicum. The DNA extracted from blood and tick samples was assayed for molecular characterization of Coxiella burnetii targeting the 16S rRNA and superoxide dismutase (SOD) genes. Amplified PCR products were purified, cloned and custom sequenced. PCR assay showed 3.3% (8/245) of the dogs were positive for Coxiella-like bacteria. Coxiella-like bacterial DNA was detected in adult fully engorged females of R. sanguineus (7.7%, 13/168), R. (B.) microplus (3.3%, 4/123) and H. anatolicum (1.9%, 1/54). Coxiella-like bacterial DNA lacked in adult male or nymphal stage. The infection rate did not vary significantly between seasons, nor according to sex or age of the host. Six nucleotide sequences of 16S rRNA and SOD genes are discussed.








Similar content being viewed by others
References
Astobiza I, Tilburg JJHC, Piñero A, Hurtado A, Garcia- Perez AL, Nabuurs-Fanssen MH, Klassen CHW (2012) Genoty** of Coxiella burnetii from domestic ruminants in northern Spain. BMC Vet Res 8:241. https://doi.org/10.1186/1746-6148-8-241
Badudeiri B (1959) Q fever: a zoonosis. Adv Vet Sci 5:82–154
Baker GC, Smith JJ, Cowan DA (2003) Review and re-analysis of domain-specific 16S primers. J Microbiol Methods 55(3):541–555
Carbonero A, Guzman LT, Montano K, Torralbo A, Arenas-Montes A, Saa LR (2015) Coxiella burnetii seroprevalence and associated risk factors in dairy and mixed cattle farms from Ecuador. Prev Vet Med 118:427–435. doi:https://doi.org/10.1016/j.prevetmed.2015.01.007
Dikshit K, Dikshit J (2014) Weather and climate of North-East India. North–East India: Land, People and Economy. Springer, Dordrecht, pp 149–173
Drancourt M, Berger P, Raoult D (2004) Systematic 16S rRNA gene sequencing of atypical clinical isolates identified 27 new bacterial species associated with humans. J Clin Microbiol 42(5):2197–2202
Duron O, Sidi-Boumedine K, Rousset E, Moutailler S, Jourdain E (2015) The importance of ticks in Q fever transmission: what has (and has not) been demonstrated? Trends Parasitol 31(11):536–552. https://doi.org/10.1016/j.pt.2015.06.014
Fard SN, Khalili MO (2011) PCR-detection of Coxiella burnetii in ticks collected from sheep and goats in Southeast Iran. Iran J Arthropod-Borne Dis 5(1):1–6
Felsenstein J (1985) Confidence limits on phylogenies: an approach using the bootstrap. Evolution 39(4):783–791. https://doi.org/10.1111/j.1558-5646.1985.tb00420.x
Filipe AF, Filomena Magalhães M, Collares-Pereira MJ (2010) Native and introduced fish species richness in Mediterranean streams: the role of multiple landscape influences. Divers Distribut 16(5):773–785
Glazunova O, Roux V, Freylikman O, Sekeyova Z, Fournous G, Tyczka J, Tokarevich N, Kovacova E, Marrie TJ, Raoult D (2005) Coxiella burnetii genoty**. Emerg Infect Dis 11(8):1211–1227. doi: https://doi.org/10.3201/eid1108.041354
Hall TA (1999) BioEdit: a user-friendly biological sequence alignment editor and analysis program for Windows 95/98/NT. Nucl Acids Symp Ser 41:95–98. https://doi.org/10.14601/Phytopathol_Mediterr-14998u1.29
Heinzen RA, Frazier ME, Mallavia LP (1992) Coxiella burnetii superoxide dismutase gene: cloning, sequencing, and expression in Escherichia coli. Infect Immun 60(9):3814–3823
Hornok S, Abichu G, Meli ML, Tánczos B, Sulyok KM, Gyuranecz M, Gönczi E, Farkas R, Hofmann-Lehmann R (2014) Influence of the biotope on the tick infestation of cattle and onthe tick-borne pathogen repertoire of cattle ticks in Ethiopia. PLoS ONE 9(9):e106452. https://doi.org/10.1371/journal.pone.0106452
Huijsmans CJ, Schellekens JJ, Wever PC, Toman R, Savelkoul PH, Janse I, Hermans MH (2011) Single-nucleotide-polymorphism genoty** of Coxiella burnetii during a Q fever outbreak in The Netherlands. Appl Environ Microbiol 77(6):2051–2057
Jang YR, Shin Y, ** CE, Koo B, Park SY, Kim MC, Kim T, Chong YP, Lee SO, Choi SH, Kim YS (2017) Molecular detection of Coxiella burnetii from the formalin-fixed tissues of Q fever patients with acute hepatitis. PLoS ONE 12:0180237. https://doi.org/10.1371/journal.pone.0180237
Jones DT, Taylor WR, Thornton JM (1992) The rapid generation of mutation data matrices from protein sequences. Comput Appl Biosci 8:275–282. https://doi.org/10.1093/bioinformatics/8.3.275
Jourdain E, Duron O, Barry S, González-Acuña D, Sidi-Boumedine K (2015) Molecular methods routinely used to detect Coxiella burnetii in ticks crossreact with Coxiella-like bacteria. Infect Ecol Epidemiol 5:129230. https://doi.org/10.3402/iee.v5.29230
Keshavmurthy R, Singh BB, Kalambhe DG, Aulakh RS, Dhand NK (2019) Prevalence of Coxiella burnetii in cattle and buffalo populations in Punjab, India. Prev Vet Med 166:16–20. doi: https://doi.org/10.1016/J.prevetmed.2019.03.003
Khalili M, Rezaei M, Akhtardanesh B, Abiri Z, Shahheidaripour S (2018) Detection of Coxiella burnetii (Gammaproteobacteria: Coxiellaceae) in ticks collected from infested dogs in Kerman, Southeast of Iran. Persian J Acarol 1(1):93–100. https://doi.org/10.22073/pja.v/7i1.30699
Knobel DL, Maina AN, Cutler SJ, Ogola E, Feikin DR, Junghae M, Halliday JE, Richards AL, Breiman RF, Cleaveland S, Njenga MK (2013) Coxiella burnetii in humans, domestic ruminants and ticks in rural western Kenya. Am J Trop Med Hyg 88(3):513–518
Körner S, Makert GR, Mertens-Scholz K, Henning K, Pfeffer M, Starke A, Nijhof AM, Ulbert S (2020) Uptake and fecal excretion of Coxiella burnetii by Ixodes ricinus and Dermacentor marginatus ticks. Parasites Vectors 13(1):1-1. https://doi.org/10.1186/s13071-020-3956-z
Kumar S, Stecher G, Li M, Knyaz C, Tamura K (2018) MEGA X: molecular evolutionary genetics analysis across computing platforms. MolBiolEvo 35:1547–1549. https://doi.org/10.1093/molbev/msy096
Mares-Guia MA, Rozental T, Guterres A, Gomes R, Almeida DN, Moreira NS, Barreira JD, Favacho AR, Santana AL, Lemos ER (2014) Molecular identification of the agent of Q fever–Coxiella burnetii–in domestic animals in State of Rio de Janeiro, Brazil. Revista da Sociedade Brasileira de Medicina Tropical 47:231–234
McLaughlin HP, Cherney B, Hakovirta JR, Priestley RA, Conley A, Carter A, Hodge D, Pillai SP, Weigel LM, Kersh GJ, Sue D (2017) Phylogenetic inference of Coxiella burnetii by 16S rRNA gene sequencing. PloS ONE 12(12):e0189910
Muramatsu Y, Usaki N, Thongchai C, Kramomtong I, Kriengsak P, Tamura Y (2014) Seroepidemiologic survey in Thailand of Coxiella burnetii infection in cattle and chickens and presence in ticks attached to cattle. Southeast Asian J Trop Med Public Health 45:1167–1172. https://doi.org/10.4269/ajtmh.16-0336
Muskens J, van Engelen E, van Maanen C, Bartels C, Lam TJ (2011) Prevalence of Coxiella burnetii infection in Dutch dairy herds based on testing bulk tank milk and individual samples by PCR and ELISA. Vet Rec 168(3):79. https://doi.org/10.1136/vr.c6106
Pereira F, Carneiro J, Matthiesen R, van Asch B, Pinto N, Gusmao L, Amorim A (2010) Identification of species by multiplex analysis of variable-length sequences. Nucleic Acids Res 38(22):e203. https://doi.org/10.1093/nar/gkq865
Sahu R, Rawool DB, Dhaka P, Yadav JP, Mishra SP et al (2020) Current perspectives on the occurrence of Q fever: highlighting the need for systematic surveillance for a neglected zoonotic disease in Indian subcontinent. Environ Microbiol Rep 13(2):138–158
Satta G, Chis V, Cabras P, Fois F, Masala G (2010) Pathogens and symbionts in ticks: a survey on tick species distribution and presence of tick-transmitted microorganisms in Sardinia, Italy. J Med Microbiol 9(9):e106452. https://doi.org/10.1099/jmm.0.021543-0
Seo MG, Ouh IO, Lee SH, Kim JW, Rhee MH, Kwon OD, Kim TH, Kwak D (2017) Prevalence of Coxiella burnetii in cattle at South Korean national breeding stock farms. PLoS ONE 12(5):e0177478. https://doi.org/10.1371/journal.pone.0177478
Shitta KB, Aron GA, James-Rugu NN, Effanga EO (2011) Seasonal variation in the abundance and distribution of Ixodid Ticks on Mongrel, Alsatian and Mixed breeds of dogs (Canis familiaris) in Jos, Plateau State north-central Nigeria. World J Sci Technol 1(4):24–29
Simmon KE, Croft AC, Pett CA (2006) Application of SmartGene IDNS software to partial 16S rRNA gene sequences for a diverse group of bacteria in a clinical laboratory. J Clin Microbiol 44:4400–4406. https://doi.org/10.1128/JCM.01364-06
Sonenshine DE, Roe RM (2014) Biology of Ticks, 2nd edn. Oxford University Press, New York
Špitalská E, Kocianova E (2013) Detection of Coxiella burnetii in ticks collected in Slovakia and Hungary. Eur J Epidemiol 18(3):263–266. https://doi.org/10.1023/A:1023330222657
Spyridaki I, Gikas A, Kofteridis D, Psaroulaki A, Tselentis Y (1998) Q fever in the Greek island of Crete: detection, isolation, and molecular identification of eight strains of Coxiella burnetii from clinical samples. J Clin Microbiol 36(7):2063–2067. https://doi.org/10.1128/JCM.36.7.2063-2067.1998
Spyridaki I, Psaroulaki A, Loukaides F, Antoniou M, Hadjichristodolou C, Tselentis Y (2002) Isolation of Coxiella burnetii by a centrifugation shell vial assay from ticks collected in Cyprus: detection by nested polymerase chain reaction (PCR) and by PCR-restriction fragment length polymorphism analyses. Am J Trop Med Hyg 66(1):86–90. https://doi.org/10.4269/ajtmh.2002.66.86
Stein A, Raoult D (1992) Detection of Coxiella burnetti by DNA amplification using polymerase chain reaction. J Clin Microbiol 30(9):2462-2466. https://doi.org/10.1128/jcm.30.9.2462-2466
Stephen S, Sangeetha B, Antony PX (2014) Seroprevalence of coxiellosis (Q fever) in sheep & goat in Puducherry & neighbouring Tamil Nadu. Indian J Med Res 140(6):785–787
Tamura K, Nei M (1993) Estimation of the number of nucleotide substitutions in the control region of mitochondrial DNA. Hum chimpanzees MolBiolEvol 10:512–526. https://doi.org/10.1093/oxfordjournals.molbev.a040023
Taylor MA, Coop RL, Wall RL (2016) Veterinary parasitology, 4th edn. Wiley-Blackwell, New York
Ullah Q, Jamal H, Quereshi ZI, Saquib M, Neubauer H (2019) Seroepidemiology of Q fever (Coxiellosis) in small ruminants kept at Government Livestock Farms of Punjab, Pakistan. Pakistan J Zool 51(1):135–140.
Varela-Castro L, Zuddas C, Ortega N, Serrano E, Salinas J, Castellà J, Castillo-Contreras R, Carvalho J, Lavín S, Mentaberre G (2018) On the possible role of ticks in the eco-epidemiology of Coxiella burnetii in a Mediterranean ecosystem. Ticks Tick-Borne Dis 9(3):687–694. https://doi.org/10.1016/j.ttbdis.2018.02.014
Wasihun P, Doda D (2013) Study on prevalence and identification of ticks in Humbo district, Southern Nations, Nationalities, and people’s region (SNNPR). Ethiopia J Vet Med Anim Health 5(3):73–80
Acknowledgements
The authors duly acknowledge the Dean, College of Veterinary Sciences and Animal Husbandry, Central Agricultural University, Imphal, for providing the necessary support to conduct the experiment under the supervision of the Department of Veterinary Parasitology.
Funding
The authors declare that there was no funding for this project.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they don’t have any conflict of interest.
Ethical approval
All animal experiments were carried out strictly as per the guidelines issued by ARRIVE and Committee for the Purpose of Control and Supervision of Experiments on Animals (CPCSEA) and were approved by the Institutional Animal Ethics Committee with reference No.CAU/CVSc/IAEC/no. 8641, dated Selesih, the 25th January 2019. This study did not involve endangered or protected species.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Patra, G., Ghosh, S., Polley, S. et al. Molecular detection and genetic characterization of Coxiella-like endosymbionts in dogs and ticks infesting dogs in Northeast India. Exp Appl Acarol 86, 549–566 (2022). https://doi.org/10.1007/s10493-022-00711-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10493-022-00711-0