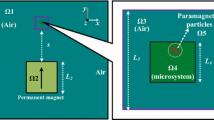
Abstract
Magnetophoresis is one of the most popular particle manipulation schemes used in microfluidic devices. Predicting the performance of arbitrary magnetophoretic module via simulation is crucial for the realization of high-performance, purpose-built magnetophoretic devices. However, the simulation process may take a long time, slowing down the simulation throughput. This work presents a novel algorithm to rapidly predict the trajectory of magnetic particles in microfluidic devices with arbitrary magnetophoretic structures under continuous fluidic flow conditions. Based on the Eulerian–Lagrangian approach, by employing numerically calculated magnetic force and fluidic velocity datasets obtained by finite element analysis software, we used an exponential integrator inspired numerical method to solve for the motion of magnetic particles in the system with the assumption that the magnetic and fluidic forces are constants within a small period of time. The results showed that, for our application, the order of truncation error of our algorithm is comparable with the 4th Order Runge–Kutta method and commercial Runge–Kutta based solver ODE45 from MATLAB. Furthermore, our algorithm is inherently stable, thus enabling the use of arbitrary time step** values without the need of adaptive step size adjustment, which significantly reduces simulation time when compared to the 4th Order Runge–Kutta method and ODE45. In our simulations, a maximum of 40 times reduction in simulation time can be achieved. The proposed algorithm and the performed simulations are validated experimentally.














Similar content being viewed by others
Data availability
Data and material are available from the corresponding author upon request.
Code availability
Code snippets are available from the corresponding author upon request.
References
Aharoni A (1998) Demagnetizing factors for rectangular ferromagnetic prisms. J Appl Phys 83:3432–3434. https://doi.org/10.1063/1.367113
Ai Y, Sanders CK, Marrone BL (2013) Separation of Escherichia coli bacteria from peripheral blood mononuclear cells using standing surface acoustic waves. Anal Chem 85:9126–9134. https://doi.org/10.1021/ac4017715
Alam M, Golozar M, Darabi J (2018) Modelling and simulation of particle-particle interaction in a magnetophoretic bio-separation chip. Phys Fluids 30:042001. https://doi.org/10.1063/1.5022582
Ali-Cherif A, Begolo S, Descroix S, Viovy JL, Malaquin L (2012) Programmable magnetic tweezers and droplet microfluidic device for high-throughput nanoliter multi-step assays. Angew Chem 124:10923–10927. https://doi.org/10.1002/ange.201203862
Amini H, Lee W, Di Carlo D (2014) Inertial microfluidic physics. Lab Chip 14:2739–2761. https://doi.org/10.1039/C4LC00128A
Augustsson P, Persson J, Ekström S, Ohlin M, Laurell T (2009) Decomplexing biofluids using microchip based acoustophoresis. Lab Chip 9:810–818. https://doi.org/10.1039/B811027A
Butcher JC (1996) A history of Runge-Kutta methods. Appl Numer Math 20:247–260. https://doi.org/10.1016/0168-9274(95)00108-5
Chan KY, Ye WW, Zhang Y, **ao LD, Leung PH, Li Y, Yang M (2013) Ultrasensitive detection of E. coli O157: H7 with biofunctional magnetic bead concentration via nanoporous membrane based electrochemical immunosensor. Biosens Bioelectron 41:532–537. https://doi.org/10.1016/j.bios.2012.09.016
Chen H, Chen C, Bai S, Gao Y, Metcalfe G, Cheng W, Zhu Y (2018) Multiplexed detection of cancer biomarkers using a microfluidic platform integrating single bead trap** and acoustic mixing techniques. Nanoscale 10:20196–20206. https://doi.org/10.1039/C8NR06367B
Cheng R, Zhu T, Mao L (2014) Three-dimensional and analytical modeling of microfluidic particle transport in magnetic fluids. Microfluid Nanofluid 16:1143–1154. https://doi.org/10.1007/s10404-013-1280-z
Chu H, Doh I, Cho YH (2009) A three-dimensional (3D) particle focusing channel using the positive dielectrophoresis (pDEP) guided by a dielectric structure between two planar electrodes. Lab Chip 9:686–691. https://doi.org/10.1039/B812213J
Derks RJ, Dietzel A, Wimberger-Friedl R, Prins MW (2007) Magnetic bead manipulation in a sub-microliter fluid volume applicable for biosensing. Microfluid Nanofluid 3:141–149. https://doi.org/10.1007/s10404-006-0112-9
Do J, Ahn CH (2008) A polymer lab-on-a-chip for magnetic immunoassay with on-chip sampling and detection capabilities. Lab Chip 8:542–549. https://doi.org/10.1039/B715569G
Dormand JR, Prince PJ (1980) A family of embedded Runge-Kutta formulae. J Comput Appl Math 6:19–26. https://doi.org/10.1016/0771-050X(80)90013-3
Feng Z, Zhi S, Guo L, Wei M, Zhou Y, Lei C (2018) A novel integrated microfluidic platform based on micro-magnetic sensor for magnetic bead manipulation and detection. Microfluid Nanofluid 22:1–9. https://doi.org/10.1007/s10404-018-2107-8
Forbes TP, Forry SP (2012) Microfluidic magnetophoretic separations of immunomagnetically labeled rare mammalian cells. Lab Chip 12:1471–1479. https://doi.org/10.1039/C2LC40113D
Furlani EP (2007) Magnetophoretic separation of blood cells at the microscale. J Phys D Appl Phys 40:1313. https://doi.org/10.1088/0022-3727/40/5/001
Furlani EP, Ng KC (2006) Analytical model of magnetic nanoparticle transport and capture in the microvasculature. Phys Rev E 73:061919. https://doi.org/10.1103/PhysRevE.73.061919
Furlani EP, Sahoo Y, Ng KC, Wortman JC, Monk TE (2007) A model for predicting magnetic particle capture in a microfluidic bioseparator. Biomed Microdevice 9:451–463. https://doi.org/10.1007/s10544-007-9050-x
Gajasinghe RW, Toprak A, Senveli SU, Yildirim YA, Jones M, Ince T, Tigli O (2016) Rare cell immobilization on MEMS scale sensors using micro-electromagnets. IEEE Sens J 16:7572–7580. https://doi.org/10.1109/JSEN.2016.2600944
Gerber R, Takayasu M, Friedlaender F (1983) Generalization of HGMS theory: the capture of ultra-fine particles. IEEE Trans Magn 19:2115–2117. https://doi.org/10.1109/TMAG.1983.1062795
Gijs MA (2004) Magnetic bead handling on-chip: new opportunities for analytical applications. Microfluid Nanofluid 1:22–40. https://doi.org/10.1007/s10404-004-0010-y
Golozar M, Molki M, Darabi J (2017) Computational and performance analysis of a continuous magnetophoretic bioseparation chip with alternating magnetic fields. Microfluid Nanofluid 21:73. https://doi.org/10.1007/s10404-017-1909-4
Gómez-Pastora J, Karampelas IH, Bringas E, Furlani EP, Ortiz I (2019) Numerical analysis of bead magnetophoresis from flowing blood in a continuous-flow microchannel: implications to the bead-fluid interactions. Sci Rep 9:1–3. https://doi.org/10.1038/s41598-019-43827-x
Hamada R, Takayama H, Shonishi Y, Mao L, Nakano M, Suehiro J (2013) A rapid bacteria detection technique utilizing impedance measurement combined with positive and negative dielectrophoresis. Sens Actuators, B Chem 181:439–445. https://doi.org/10.1016/j.snb.2013.02.030
Hamilton ES, Ganjalizadeh V, Wright JG, Schmidt H, Hawkins AR (2020) 3D Hydrodynamic focusing in microscale optofluidic channels formed with a single sacrificial layer. Micromachines 11:349. https://doi.org/10.3390/mi11040349
Hochbruck M, Ostermann A (2010) Exponential integrators. Acta Numer 19:209–286. https://doi.org/10.1017/S0962492910000048
Inglis DW, Riehn R, Austin RH, Sturm JC (2004) Continuous microfluidic immunomagnetic cell separation. Appl Phys Lett 85:5093–5095. https://doi.org/10.1063/1.1823015
Katsikis G, Cybulski JS, Prakash M (2015) Synchronous universal droplet logic and control. Nat Phys 11:588–596. https://doi.org/10.1038/nphys3341
Khashan SA, Furlani EP (2012) Effects of particle–fluid coupling on particle transport and capture in a magnetophoretic microsystem. Microfluid Nanofluid 12:565–580. https://doi.org/10.1007/s10404-011-0898-y
Khashan SA, Furlani EP (2013) Coupled particle–fluid transport and magnetic separation in microfluidic systems with passive magnetic functionality. J Phys D Appl Phys 46:125002. https://doi.org/10.1088/0022-3727/46/12/125002
Khashan SA, Furlani EP (2014) Scalability analysis of magnetic bead separation in a microchannel with an array of soft magnetic elements in a uniform magnetic field. Sep Purif Technol 125:311–318. https://doi.org/10.1016/j.seppur.2014.02.007
Kim M, Jung T, Kim Y, Lee C, Woo K, Seol JH, Yang S (2015) A microfluidic device for label-free detection of Escherichia coli in drinking water using positive dielectrophoretic focusing, capturing, and impedance measurement. Biosens Bioelectron 74:1011–1015. https://doi.org/10.1016/j.bios.2015.07.059
Kye HG, Park BS, Lee JM, Song MG, Song HG, Ahrberg CD, Chung BG (2019) Dual-neodymium magnet-based microfluidic separation device. Sci Rep 9:1–10. https://doi.org/10.1038/s41598-019-45929-y
Modak N, Datta A, Ganguly R (2010) Numerical analysis of transport and binding of a target analyte and functionalized magnetic microspheres in a microfluidic immunoassay. J Phys D Appl Phys 43:485002. https://doi.org/10.1088/0022-3727/43/48/485002
Pamme N, Eijkel JC, Manz A (2006) On-chip free-flow magnetophoresis: separation and detection of mixtures of magnetic particles in continuous flow. J Magn Magn Mater 307:237–244. https://doi.org/10.1016/j.jmmm.2006.04.008
Park C, Lee J, Kim Y, Kim J, Lee J, Park S (2017) 3D-printed microfluidic magnetic preconcentrator for the detection of bacterial pathogen using an ATP luminometer and antibody-conjugated magnetic nanoparticles. J Microbiol Methods 132:128–133. https://doi.org/10.1016/j.mimet.2016.12.001
Phurimsak C, Tarn MD, Peyman SA, Greenman J, Pamme N (2014) On-chip determination of C-reactive protein using magnetic particles in continuous flow. Anal Chem 86:10552–10559. https://doi.org/10.1021/ac5023265
Prozorov R, Kogan VG (2018) Effective demagnetizing factors of diamagnetic samples of various shapes. Phys Rev Appl 10:014030. https://doi.org/10.1103/PhysRevApplied.10.014030
Ramadan Q, Poenar DP, Yu C (2009) Customized trap** of magnetic particles. Microfluid Nanofluid 6:53–62. https://doi.org/10.1007/s10404-008-0296-2
Ramadan Q, Samper V, Poenar D, Yu C (2006) Magnetic-based microfluidic platform for biomolecular separation. Biomed Microdevice 8:151–158. https://doi.org/10.1007/s10544-006-7710-x
Royet D, Hériveaux Y, Marchalot J, Scorretti R, Dias A, Dempsey NM, Bonfim M, Simonet P, Frénéa-Robin M (2017) Using injection molding and reversible bonding for easy fabrication of magnetic cell trap** and sorting devices. J Magn Magn Mater 427:306–313. https://doi.org/10.1016/j.jmmm.2016.10.102
Shampine LF, Reichelt MW (1997) The MATLAB ODE Suite. SIAM J Sci Comput 18:1–22. https://doi.org/10.1137/S1064827594276424
Sun X, Feng Z, Zhi S, Lei C, Zhang D, Zhou Y (2017) An integrated microfluidic system using a micro-fluxgate and micro spiral coil for magnetic microbeads trap** and detecting. Sci Rep 7:1–8. https://doi.org/10.1038/s41598-017-13389-x
Yamada M, Seki M (2005) Hydrodynamic filtration for on-chip particle concentration and classification utilizing microfluidics. Lab Chip 5:1233–1239. https://doi.org/10.1039/B509386D
Zhang J, Li W, Alici G (2017) Inertial microfluidics: Mechanisms and applications. Adv Mechatron MEMS Devices II:563–593. https://doi.org/10.1007/978-3-319-32180-6_25
Funding
This project is supported by a grant from the Innovation and Technology Commission (Project No. ITS/200/19FP).
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception. KCET contributed to the study design. Simulations, data collection and analysis were performed by KCET. The first draft of the manuscript was written by KCET and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors have no competing interests to declare that are relevant to the content of this article.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Tjon, K.C.E., Yuan, J. A novel algorithm for rapid estimation of magnetic particle trajectory in arbitrary magnetophoretic devices under continuous fluid flow. Microfluid Nanofluid 26, 82 (2022). https://doi.org/10.1007/s10404-022-02586-4
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10404-022-02586-4