Abstract
Background
We evaluated the risk of metachronous colorectal cancer (mCRC) and explored the optimal extent of colectomy in patients with Lynch syndrome (LS) and first colon cancer (fCC) in Japan, where the extent of colectomy for colon cancer (CC) is shorter than that in Western countries.
Methods
The clinicopathologic and survival data of patients with LS who developed CC were collected from a nationwide database and analyzed retrospectively. The cumulative incidence of mCRC after actual segmental colectomy was compared with that of mCRC when more extensive colectomy was assumed.
Results
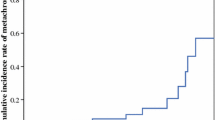
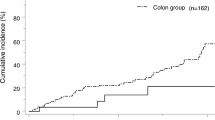
There were 142 eligible patients (65 female). The median age at fCC surgery was 46.5 (range: 14–80) years. The cumulative incidence of 5-, 10-, and 20-year mCRC rate was 13.4%, 20.8%, and 53.6%, respectively. The incidence was higher in the left-sided group (splenic flexure to rectosigmoid colon, n = 54) than in the right-sided group (cecum to transvers colon, n = 88) (66.3% vs. 45.3% in 20 years, P < 0.01). Assuming that all patients would have undergone hemicolectomy or total colectomy, the estimated mCRC risk was 41.5% and 9.4% (P < 0.01, vs. actual procedures), respectively. The 20-year overall survival rate of all the patients was 83.3% without difference by fCC sidedness (P = 0.38).
Conclusions
To reduce the incidence of mCRC, patients with genetically diagnosed LS and fCC, preferentially located in the left-sided colon, may need to undergo more extended colectomy than that usually performed in Japan. However, such extended colectomy should be counterbalanced with favorable overall survival and actual risk of mCRC development.



Similar content being viewed by others
References
Parry S, Win AK, Macrae FA et al (2011) Metachronous colorectal cancer risk for mismatch repair gene mutation carriers: the advantage of more extensive surgery. Gut 60:950–957. https://doi.org/10.1186/1897-4287-9-s1-o1
Stupart DA, Goldberg PA, Baigrie RJ et al (2011) Surgery for colonic cancer in HNPCC: total vs segmental colectomy: surgery for colonic cancer in HNPCC. Colorectal Dis 13:1395–1399. https://doi.org/10.1111/j.1463-1318.2010.02467.x
Kim TJ, Kim ER, Hong SN et al (2017) Survival outcome and risk of metachronous colorectal cancer after surgery in Lynch syndrome. Ann Surg Oncol 24:1085–1092. https://doi.org/10.1245/s10434-016-5633-1
Renkonen-Sinisalo L, Seppälä TT, Järvinen HJ et al (2017) Subtotal colectomy for colon cancer reduces the need for subsequent surgery in Lynch syndrome. Dis Colon Rectum 60:792–799. https://doi.org/10.1097/DCR.0000000000000802
Hiatt MJ, Casey MJ, Lynch HT et al (2018) Efficacy of proximal colectomy for surgical management of right-sided first colorectal cancer in Lynch Syndrome mutation carriers. Am J Surg 216:99–105. https://doi.org/10.1016/j.amjsurg.2017.11.003
Quezada-Diaz FF, Hameed I, von Mueffling A et al (2020) Risk of metachronous colorectal neoplasm after a segmental colectomy in Lynch syndrome patients according to mismatch repair gene status. J Am Coll Surg 230:669–675. https://doi.org/10.1016/j.jamcollsurg.2020.01.005
Giardiello FM, Allen JI, Axilbund JE et al (2014) Guidelines on genetic evaluation and management of Lynch syndrome: a consensus statement by the US Multi-society Task Force on colorectal cancer. Am J Gastroenterol 109:1159–1179. https://doi.org/10.1038/ajg.2014.186
Japanese Society for Cancer of the Colon and Rectum (2019) Japanese classification of colorectal, appendiceal, and anal carcinoma. J Anus Rectum Colon 3:175–195. https://doi.org/10.23922/jarc.2019-018
West NP, Kobayashi H, Takahashi K et al (2012) Understanding optimal colonic cancer surgery: comparison of Japanese D3 resection and European complete mesocolic excision with central vascular ligation. J Clin Oncol 30:1763–1769. https://doi.org/10.1200/JCO.2011.38.3992
Kobayashi H, West NP, Takahashi K et al (2014) Quality of surgery for stage III colon cancer: comparison between England, Germany, and Japan. Ann Surg Oncol 21(Suppl 3):S398-404. https://doi.org/10.1245/s10434-014-3578-9
Shiozawa M, Ueno H, Shiomi A et al (2021) Study protocol for an international prospective observational cohort study for optimal bowel resection extent and central radicality for colon cancer (T-REX study). Jpn J Clin Oncol 51:145–155. https://doi.org/10.1093/jjco/hyaa115
Brierley JD, Gospodarowicz MK, Wittekind C et al (2017) UICC TNM classification of malignant tumours, 8th edn. Wiley-Blackwell, Oxford, p 272
Johnson CH, Peace S, Adamo P et al (2007) The 2007 multiple primary and histology cording rules. National Cancer Institute, Surveillance, Epidemiology and End Results Program. Bethesda, MD. https://seer.cancer.gov/tools/mphrules/mphrules_text.pdf
Haanstra JF, de Vos Tot Nederveen Cappel WH, Gopie JP et al (2012) Quality of life after surgery for colon cancer in patients with Lynch syndrome: partial versus subtotal colectomy: partial versus subtotal colectomy. Dis Colon Rectum 55:653–659. https://doi.org/10.1097/DCR.0b013e31824f5392
Møller P, Seppälä T, Bernstein I et al (2017) Cancer incidence and survival in Lynch syndrome patients receiving colonoscopic and gynaecological surveillance: first report from the prospective Lynch syndrome database. Gut 66:464–472. https://doi.org/10.1136/gutjnl-2015-309675
Dominguez-Valentin M, Sampson JR, Seppälä TT et al (2020) Cancer risks by gene, age, and gender in 6350 carriers of pathogenic mismatch repair variants: findings from the Prospective Lynch Syndrome Database. Genet Med 22:15–25. https://doi.org/10.1038/s41436-019-0596-9
de Vos tot Nederveen Cappel WH, Buskens E, van Duijvendijk P et al (2003) Decision analysis in the surgical treatment of colorectal cancer due to a mismatch repair gene defect. Gut 52:1752–1755. https://doi.org/10.1136/gut.52.12.1752
Seppälä TT, Latchford A, Negoi I et al (2021) European guidelines from the EHTG and ESCP for Lynch syndrome: an updated third edition of the Mallorca guidelines based on gene and gender. Br J Surg 108:484–498. https://doi.org/10.1002/bjs.11902
Acknowledgements
We thank all study participants and staff for their contributions to this project.
Author information
Authors and Affiliations
Contributions
All authors contributed to the study’s conception and design. KC, YM, HI, TY, NT, and KT developed the main concept and designed the study. KC wrote the first draft of the manuscript and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflicts of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Takeshi Nakajima: Affiliation has been moved to Department of Medical Ethics and Medical Genetics, Kyoto University School of Public Health, Kyoto, Japan.
About this article
Cite this article
Chikatani, K., Ishida, H., Mori, Y. et al. Risk of metachronous colorectal cancer after colectomy for first colon cancer in Lynch syndrome: multicenter retrospective study in Japan. Int J Clin Oncol 28, 1633–1640 (2023). https://doi.org/10.1007/s10147-023-02412-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10147-023-02412-z