Abstract
Background
Familial adenomatous polyposis (FAP), an autosomal dominant disorder characterized by multiple colonic polyps, is caused by a germline pathogenic variant of the APC gene. However, this variant is not detected in up to 30% of patients with the adenomatous polyposis phenotype.
Methods
We performed next-generation sequencing (NGS) to identify the causative genes in FAP patients with 10 or more polyps. For patients in whom the APC germline variant was not able to be identified, we screened for APC mosaicism using high-coverage NGS of APC with DNA from leucocytes and/or frozen tissue.
Results
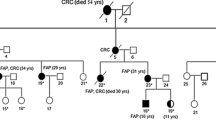
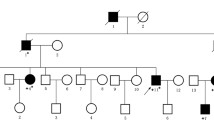
The pathogenic APC germline variant was found in 93.3%, 71.6%, and 17.1% of patients with profuse-type polyposis, sparse-type polyposis, and oligo-polyposis, respectively. The APC germline variant detection rate in patients with FAP-related diseases was 69.7% for fundic gland polyposis, 79.7% for duodenal adenoma, 94.7% for desmoid tumor, and 71.4% for thyroid cancer, with increasing numbers of extracolonic lesions associated with an increasing APC germline variant detection rate. A mosaic test detected nine patients with APC mosaicism. A comparison of APC-associated polyposis with APC mosaicism showed that patients with APC mosaicism had a low frequency of duodenal adenoma and a family history of colonic polyposis.
Conclusions
We determined the detection rate of the APC germline variant by phenotype and identified APC mosaicism. Genetic testing of FAP patients is important because it can help with surgical decision-making, monitoring, and genetic counseling. Furthermore, genetic testing by NGS proved to be an effective method of detecting APC germline variants.




Similar content being viewed by others
Change history
20 December 2021
A Correction to this paper has been published: https://doi.org/10.1007/s10147-021-02106-4
References
Kinzler KW, Nilbert MC, Su LK et al (1991) Identification of FAP locus genes from chromosome 5q21. Science 253(5020):661–665
Miyoshi Y, Ando H, Nagase H et al (1992) Germ-line mutations of the APC gene in 53 familial adenomatous polyposis patients. Proc Natl Acad Sci USA 89:4452–4456
Grover S, Kastrinos F, Steyerberg EW et al (2012) Prevalence and phenotypes of APC and MUTYH mutations in patients with multiple colorectal adenomas. JAMA 308:485–492
Al-Tassan N, Chmiel NH, Maynard J et al (2002) Inherited variants of MYH associated with somatic G:C–>T: a mutations in colorectal tumors. Nat Genet 30:227–232
Jasperson KW, Patel SG, Ahnen DJ. (1998) APC-Associated Polyposis Conditions. 1998 Dec 18 [updated 2017 Feb 2. In: Adam MP, Ardinger HH, Pagon RA, Wallace SE, Bean LJH, Stephens K, Amemiya A, editors. GeneReviews® [Internet]. Seattle (WA): University of Washington, Seattle; 1993–2018. Available from http://www.ncbi.nlm.nih.gov/books/NBK1345/
Nagase H, Miyoshi Y, Horii A et al (1992) Correlation between the location of germ-line mutations in the APC gene and the number of colorectal polyps in familial adenomatous polyposis patients. Cancer Res 52:4055–4057
Miyaki M, Yamaguchi T, Iijima T et al (2008) Difference in characteristics of APC mutations between colonic and extracolonic tumors of FAP patients: variations with phenotype. Int J Cancer 122:2491–2497
Palles C, Cazier JB, Howarth KM et al (2013) Germline mutations affecting the proofreading domains of POLE and POLD1 predispose to colorectal adenomas and carcinomas. Nat Genet 45:136–144
Hes FJ, Nielsen M, Bik EC et al (2008) Somatic APC mosaicism: an underestimated cause of polyposis coli. Gut 57(1):71–76
Aretz S, Stienen D, Friedrichs N et al (2007) Somatic APC mosaicism: a frequent cause of familial adenomatous polyposis (FAP). Hum Mutat 28(10):985–992
Yamaguchi K, Komura M, Yamaguchi R et al (2015) Detection of APC mosaicism by next-generation sequencing in an FAP patient. J Hum Genet 60:227–231
Kohda M, Kumamoto K, Eguchi H et al (2016) Rapid detection of germline mutations for hereditary gastrointestinal polyposis/cancers using HaloPlex target enrichment and high-throughput sequencing technologies. Fam Cancer 15:553–562
Eguchi H, Okazaki Y (2019) Next-generation sequencing for genetic diagnosis of hereditary colorectal cancer and polyposis syndrome. In: Ishida H, Koda K (eds) Recent Advances in the Treatment of Colorectal Cancer. Springer, Singapore, pp 115–125
Lek M, Karczewski KJ, Minikel EV et al (2016) Analysis of protein-coding genetic variation in 60,706 humans. Nature 536:285–291
Tadaka S, Saigusa D, Motoike IN et al (2018) jMorp: Japanese multi omics reference panel. Nucleic Acids Res 46(D1):D551–D557
Pagnamenta AT, Lise S, Harrison V et al (2012) Exome sequencing can detect pathogenic mosaic mutations present at low allele frequencies. J Hum Genet 57:70–72
Judkins T, Leclair B, Bowles K et al (2015) Development and analytical validation of a 25-gene next generation sequencing panel that includes the BRCA1 and BRCA2 genes to assess hereditary cancer risk. BMC Cancer 15:215
Spier I, Drichel D, Kerick M (2016) Low-level APC mutational mosaicism is the underlying cause in a substantial fraction of unexplained colorectal adenomatous polyposis cases. J Med Genet 53(3):172–179. https://doi.org/10.1136/jmedgenet-2015-103468 (Epub 2015 Nov 27 PMID: 26613750)
Takao M, Yamaguchi T, Eguchi H et al (2018) Characteristics of MUTYH variants in Japanese colorectal polyposis patients. Int J Clin Oncol 23(3):497–503. https://doi.org/10.1007/s10147-017-1234-7 (Epub 2018 Jan 12)
Tanakaya K, Kumamoto K, Tada Y et al (2019) A germline MBD4 mutation was identified in a patient with colorectal oligopolyposis and early-onset cancer: a case report. Oncol Rep 42(3):1133–1140
Ito T, Nomizu T, Eguchi H et al (2020) The first case report of polymerase proofreading-associated polyposis in POLD1 variant, c.1433G>A pS478N, in Japan. Jpn J Clin Oncol 50:1080 (published online ahead of print, 2020 Jun 17)
Freidel W, Caspari R, Sengteller M et al (2001) Can APC mutation analysis contribute to therapeutic decisions in familial adenomatous polyposis? Experience from 680 FAP families. Gut 48:515–521
Ciavarella M, Miccoli S, Prossomariti A et al (2018) Somatic APC mosaicism and oligogenic inheritance in genetically unsolved colorectal adenomatous polyposis patients. Eur J Hum Genet 26(3):387–395
Sieber OM, Lamlum H, Crabtree MD et al (2002) Whole-gene APC deletions cause classical familial adenomatous polyposis, but not attenuated polyposis or “multiple” colorectal adenomas. Proc Natl Acad Sci USA 99(5):2954–2958
Waltraut F, Stefan A (2005) Familial adenomatous polyposis: experience from a study of 1164 unrelated german polyposis patients. Hered Cancer Clin Pract 3(3):95–114
Geneviève M, Sabine T, Reinhilde T et al (2005) Large deletions of the APC gene in 15% of mutation-negative patients with classical polyposis (FAP): a Belgian study. Hum Mutat 25(2):125–134
Nielsen M, Hes FJ, Nagengast FM et al (2007) Germline mutations in APC and MUTYH are responsible for the majority of families with attenuated familial adenomatous polyposis. Clin Genet 71(5):427–433
Valle L, de Voer RM, Goldberg Y et al (2019) Update on genetic predisposition to colorectal cancer and polyposis. Mol Aspects Med 69:10–26. https://doi.org/10.1016/j.mam.2019.03.001
Church JM, McGannon E, Hull-Boiner S et al (1992) Gastroduodenal polyps in patients with familial adenomatous polyposis. Dis Colon Rectum 35:1170–1173
Sarre RG, Frost AG, Jagelman DG et al (1987) Gastric and duodenal polyps in familial adenomatous polyposis: a prospective study of the nature and prevalence of upper gastrointestinal polyps. Gut 28:306–314
Bertoni G, Sassatelli R, Nigrisoli E et al (1999) Dysplastic changes in gastric fundic gland polyps of patients with familial adenomatous polyposis. Ital J Gastroenterol Hepatol 31:192–197
Domizio P, Talbot IC, Spigelman AD et al (1990) Upper gastrointestinal pathology in familial adenomatous polyposis: results from a prospective study of 102 patients. J Clin Pathol 43:738–743
Watanabe H, Enjoji M, Yao T et al (1978) Gastric lesions in familial adenomatosis coli: their incidence and histologic analysis. Hum Pathol 9:269–283
Bianchi LK, Burke CA, Bannett AE et al (2008) Fundic gland polyp dysplasia is common in familial adenomatous polyposis. Clin Gastroenterol Hepatol 6(2):180–185. https://doi.org/10.1016/j.cgh.2007.11.018
Heiskanen I, Kellokumpu I, Jarvinen H (1999) Management of duodenal adenomas in 98 patients with familial adenomatous polyposis. Endoscopy 31:412–416
Bulow S, Bjork J, Christensen IJ et al (2004) Duodenal adenomatosis in familial adenomatous polyposis. Gut 53:381–386
Clark SK, Neale KF, Landgrebe JC et al (1999) Desmoid tumours complicating familial adenomatous polyposis. Br J Surg 86(9):1185–1189. https://doi.org/10.1046/j.1365-2168.1999.01222.x
Latchford AR, Sturt NJH, Neale K et al (2006) A 10 year review of surgery for desmoid disease associated with familial adenomatous polyposis. Br J Surg 93(10):1258–1264. https://doi.org/10.1002/bjs.5425
Iwama T, Mishima Y, Utsunomiya J (1993) The impact of familial adenomatous polyposis on the tumorigenesis and mortality at the several organs. Its rational treatment. Ann Surg 217(2):101–108. https://doi.org/10.1097/00000658-199302000-00002
Giardiello FM, Offerhaus GJ, Lee DH et al (1993) Increased risk of thyroid and pancreatic carcinoma in familial adenomatous polyposis. Gut 34(10):1394–1396. https://doi.org/10.1136/gut.34.10.1394
Steinhagen E, Guillem JG, Chang G et al (2012) The prevalence of thyroid cancer and benign thyroid disease in patients with familial adenomatous polyposis may be higher than previously recognized. Clin Colorectal Cancer 11(4):304–308. https://doi.org/10.1016/j.clcc.2012.01.006 (Epub 2012 Mar 15)
Aretz S, Uhlhaas S, Caspari R et al (2004) Frequency and parental origin of de novo APC mutations in familial adenomatous polyposis. Eur J Hum Genet 12:52–58
Schwab AL, Tuohy TMF, Condie M et al (2008) Gonadal mosaicism and familial adenomatous polyposis. Fam Cancer 7:173–177
Tuohy TM, Burt RW (2008) Somatic mosaicism: a cause for unexplained cases of FAP? Gut 57:10–12
Kim B, Won D, Mi J et al (2019) Next-generation sequencing with comprehensive bioinformatics analysis facilitates somatic mosaic APC gene mutation detection in patients with familial adenomatous polyposis. BMC Med Genom 12:103. https://doi.org/10.1186/s12920-019-0553-0
Campbell IM, Shaw CA, Stankiewicz P et al (2015) Somatic mosaicism: implications for disease and transmission genetics. Trends Genet 31(7):382–392. https://doi.org/10.1016/j.tig.2015.03.013 (Epub 2015 Apr 21)
Jansen AML, Goel A (2020) Mosaicism in patients with colorectal cancer or polyposis syndromes: a systematic review. Clin Gastroenterol Hepatol 18:1949–1960
Acknowledgements
We are grateful to all the patients and their families and to Mr. James R. Valera for his assistance with editing the manuscript.
Funding
This study was supported in part by the Program for an Integrated Database of Clinical and Genomic Information of the Japan Agency for Medical Research and Development (AMED) [JP18kk0205004] and a grant-in-aid from the Support Project of the Strategic Research Cancer in Private Universities of the Ministry of Education, Culture, Sports, Science and Technology (MEXT) of Japan, which was awarded to Saitama Medical University Research Center for Genomic Medicine [S1311002]. This study was also supported in part by a grant from the 2018 Ochiai Memorial Award of Saitama Medical University.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
All the authors have no conflicts of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
The original publication was revised due to revision of approval numbers of the Institutional Review Board in Methods section.
Supplementary Information
Below is the link to the electronic supplementary material.
About this article
Cite this article
Takao, M., Yamaguchi, T., Eguchi, H. et al. APC germline variant analysis in the adenomatous polyposis phenotype in Japanese patients. Int J Clin Oncol 26, 1661–1670 (2021). https://doi.org/10.1007/s10147-021-01946-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10147-021-01946-4