Abstract
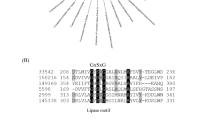
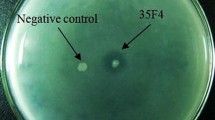
Phospholipase A1 is a hydrolytic enzyme that catalyzes the removal of the acyl group from position 1 of glycerophospholipids to form 2-acyl lysophospholipids. Lysophospholipids are used in foods, cosmetics, and pharmaceuticals as surfactants. Novel forms of phospholipase A1 that function at low temperatures are desirable for use in lipophilic systems in food processing. However, there is currently little variety in the available sources of phospholipase A1. Given this situation, we screened the intestinal contents of marine animals for phospholipase A1–producing bacteria. Colonies that formed a halo on K28CP screening medium and that grew in K28 medium were cultured in liquid K28 medium, and the supernatant was retrieved for analysis. Phosphatidylcholine was added to the culture supernatant, and the product of the reaction was analyzed by using TLC. For culture supernatants that were able to generate lysophosphatidylcholine, synthetic phosphatidylcholines were added, and the site of the reaction was determined by analyzing the fatty acid compositions of the lysophosphatidylcholines generated by GLC. A bacterial isolate from a flatfish, which we named HFKI0020, was found to have phospholipase A1 activity at low temperatures. We determined that the isolate HFKI0020 is closely related to Pseudomonas by using 16S rDNA sequence analysis and by characterizing the isolate with respect to its physiologic and biochemical properties. From the intestinal contents of a marine fish, we successfully isolated a bacterium that secretes phospholipase A1 that is active at low temperatures.


Similar content being viewed by others
References
Alutschul SF, Madden TF, Schäffer AA, Zhang J, Zhang Z, Miller W, Lipman DJ (1997) Gapped BLAST and PSI-BLAST: a new generation of protein database search programs. Nucleic Acids Res 25:3389–3402
Barrow GI, Feltham RKA (1993) Cowan and Steel’s manual for the identification of medical bacteria, 3rd edn. University Press, Cambridge
Bligh EG, Dyer WJ (1959) A rapid method of total lipid extraction and purification. Can J Biochem Physiol 37:911
Cahill MM (1990) Bacterial flora of fishes: a review. NY Microb Ecol 10:21–41
Dekker N, Tommassen J, Lustig AJ, Rosenbusch PH, Verheiji M (1997) Dimerization regulates the enzymatic activity of Escherichia coli outer membrane phospholipase A. J Biol Chem 272:3179–3184
Folch J, Ascoli I, Lees M, Meath JA, LeBaron FN (1951) Preparation of lipid extracts from brain tissue. J Biol Chem 191:883
Givskov M, Molin S (1993) Secretion of Serratia liquefaciens phospholipase from Escherichia coli. Mol Microbiol 8:229–242
Higgs HN, Glomset JA (1996) Purification and properties of a phosphatidic acid-preferring phospholipase A1 from bovine testis. Examination of the molecular basis of its activation. J Biol Chem 271:10874–10883
Jae KS, Jeong J, Joon SR (2005) Phospholipase: occurrence and production in microorganisms, assay for high-throughput screening, and gene discovery from natural and man-made diversity. J Am Chem Soc 82:691–705
Krieg NR, Holt JG (1984) Bergey’s manual of systematic bacteriology, vol 1. Williams & Wilkins, Baltimore
LeaMaster BR, Walsh WA, Brock JA, Fujioka RS (1997) Cold stress-induced changes in the aerobic heterotrophic gastrointestinal tract bacterial flora of red hybrid tilapia. J Fish Biol 50:770–780
Matui T, Taketsugu S, Kodama K, Ishii A, Yamamori K, Shimizu C (1989) Production of tetrodotoxin by the intestinal bacteria of a puffer fish Takifugu niphobles. Nippon Suisan Gakkaishi 55:2199–2203
Miyazawa D, Ikemoto A, Fujii Y, Okuyama H (2003) Partial purification and characterization of phosphatidic acid-specific phospholipase A(1) in porcine platelet membranes. Biochim Biophys Acta 1631:17–25
Oxley AP, Shipton W, Owens L, McKay D (2002) Bacterial flora from the gut of the wild and cultured banana prawn, Penaeus merguiensis. J Appl Microbiol 93:214–223
Ringø E, Olsen RE (1999) The effect of diet on aerobic bacterial flora associated with intestine of Arctic charr (Salvelinus alpinus L.). J Appl Microbiol 86:22–28
Saitou N, Nei M (1987) The neighbor-joining method: a new method for reconstructing phylogenetic trees. Mol Biol Evol 4:406–425
Shen DK, Noodeh AD, Kazemi A, Grillot R, Robson G, Brugere JF (2004) Characterisation and expression of phospholipases B from the opportunistic fungus Aspergillus fumigatus. FEMS Microbiol Lett 239:87–93
Shibata Y, Ono C, Fukui F, Watanabe I, Serizawa N, Gomi K, Yoshikawa H (2001) High-level secretory production of phospholipase A1 by Saccharomyces cerevisiae and Aspergillus oryzae. Biosci Biotechnol Biochem 65:94–101
Soldatova L, Kochoumian L, King TP (1993) Sequence similarity of a hornet (D. maculata) venom allergen phospholipase A1 with mammalian lipases. FEBS Lett 320:145–149
Song KJ, Kim KM, Rhee SJ (1999) Cloning and expression of the gene encoding phospholipase A1 from Serratia sp. MK1 in Escherichia coli. J Biotechnol 72:103–114
Sugita H, Tokuyama K, Deguchi Y (1985) The intestinal microflora of carp Cyprinus carpio, grass carp Ctenopharyngodon idella and tilapia Sarotherodon niloticus. Nippon Suisan Gakkaishi 51:1325–1329
Sugita H, Miyajima C, Iwata J, Arai S, Kubo T, Igarashi S (1988) Intestinal microflora of Japanese coastal fishes. Nippon Suisan Gakkaishi 54:875–882
Sugita H, Noguchi T, Hwang FD, Furuta M, Motokane T, Sonoda T, Hashimoto K, Deguchi Y (1987) Intestinal microflora of coastal puffer fishes. Nippon Suisan Gakkaishi 53:2201–2207
Sugita H, Okano R, Suzuki Y, Iwai D, Mizukami M, Akiyama N, Matuura S (2002) Antibacterial abilities of intestinal bacteria from larval and juvenile Japanese flounder against fish pathogens. Fisheries Sci 68:1004–1011
Watanabe K, Ishikawa C, Ohtsuka I, Kamata M, Tomita M, Yazawa K, Muramatsu H (1997) Lipid and fatty acid compositions of a novel docosahexaenoic acid-producing marine bacterium. Lipid 32:975–978
Wiedmann M, Weilmeier D, Dineen SS, Ralyea R, Boor JK (2000) Molecular and phenotypic characterization of Pseudomonas spp. isolated from milk. Appl Environ Microbiol 66:2085–2095
Yazawa K, Araki K, Ozaki N, Watanabe K, Ishikawa C, Inoue A, Numao N, Kondo K (1988) Production of eicosapentaenoic acid by marine bacteria. Biochemistry 103:5–7
Acknowledgments
The authors thank Q.P. Corporation for supply of egg yolk PC and Techno Suruga Laboratory Co., Ltd. for 16S rDNA sequence analysis.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Nishihara, M., Kamata, M., Koyama, T. et al. New Phospholipase A1-producing Bacteria from a Marine Fish. Mar Biotechnol 10, 382–387 (2008). https://doi.org/10.1007/s10126-007-9074-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10126-007-9074-5