Abstract
Aim of the study
The rising instances of multidrug-resistant pathogens are rapidly evolving into a global healthcare crisis. Identifying new ways of synthesis of antibiotics is both time-consuming and expensive. Repurposing existing drugs for the treatment of such antimicrobial-resistant pathogens has also been explored.
Methods and results
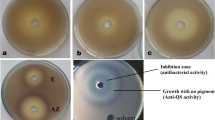
In the current study, ebselen was screened for antibacterial and antibiofilm activity against Serratia marcescens. Various antibacterial studies such as minimum inhibitory concentration (MIC), minimum bactericidal concentration (MBC), time-kill curves, intracellular reactive oxygen species (ROS) quantification, and colony-forming unit assays were performed. The antibiofilm potential was assayed by biofilm inhibition, cell surface hydrophobicity assay, eradication, quantification of extracellular DNA (eDNA), and extracellular polymeric substance (EPS) layer and scanning electron microscopy (SEM) analysis were performed. Anti-quorum sensing assay was validated by quantifying the virulence factors production. Further molecular docking of ebselen with two quorum sensing (QS) specific proteins was also carried out. Antibacterial susceptibility tests showed potent antimicrobial activity of ebselen against S. marcescens with MIC50 of 14 μg/mL. Ebselen’s ability to disturb the redox environment by inducing significant ROS generation led to bacterial death. It also showed concentration-dependent bactericidal activity as indicated by reduced bacterial growth and colony-forming unit propagation. Ebselen was also found to prevent biofilm attachment by altering the cell surface hydrophobicity while also being effective against preformed biofilms as validated by scanning electron microscopy (SEM) analysis. Additionally, ebselen showed reduced virulence factors like urease enzyme activity and prodigiosin pigment production indicating its promising anti-quorum sensing potential. Molecular docking analysis validated the strong binding of ebselen with QS-specific proteins (1Joe and PigG) with binding energies of − 6.6 and − 8.1kj/mol through hydrogen bonds and aromatic interactions. These results show that ebselen has potent antibiofilm potential that can be explored to identify treatment against bacterial infections.









Similar content being viewed by others
Code availability (software application or custom code)
Not applicable for this work.
References
AbdelKhalek A, Abutaleb NS, Mohammad H, Seleem MN (2018) Repurposing ebselen for decolonization of vancomycin-resistant enterococci (VRE). PLoS One 13(6):e0199710
Ashburn TT, Thor KB (2004) Drug repositioning: identifying and develo** new uses for existing drugs. Nat Rev Drug Discov 3:673–683
Azad GK, Tomar RS (2014) Ebselen, a promising antioxidant drug: mechanisms of action and targets of biological pathways. Mol Biol Rep 41:4865–4879
Bodro M, Sabé N, Tubau F, Lladó L, Baliellas C, Roca J, Cruzado JM, Carratalà J (2013) Risk factors and outcomes of bacteremia caused by drug-resistant ESKAPE pathogens in solid-organ transplant recipients. Transpl J 96:843–849
Bowler PG (2018) Antibiotic resistance and biofilm tolerance: a combined threat in the treatment of chronic infections. J Wou Care 27(5):273–277
Bowler P, Murphy C, Wolcott R (2020) Biofilm exacerbates antibiotic resistance: Is this a current oversight in antimicrobial stewardship? Antimicrob Resist Infect Cont 9:162. https://doi.org/10.1186/s13756-020-00830-6
Centres for Disease Control and U.S. Department of Health and Human Services (2013) Antibiotic resistance threats in the United States, Atlanta, GA. http://www.cdc.gov/drugresistance/pdf/ar‐threats‐2013‐508.pdf
Chen C, **ang Y, Yang K-W et al (2018) A protein structure-guided covalent scaffold selectively targets the B1and B2 subclass metallo-β-lactamases. Chem Commun 54:4802–4805
Chew P, Yuen DY, Stefanovic N, Pete J, Coughlan MT, Jandeleit-Dahm KA, Thomas MC, Rosenfeldt F, Cooper ME, de Haan JB (2010) Anti-atherosclerotic and renoprotective effects of ebselen in the diabetic apolipoprotein E/GPx1-double knockout mouse. Diabetes 59:3198–3207
Chiou J, Wan S, Chan KF et al (2015) Ebselen as a potent covalent inhibitor of New Delhi metallo-beta-lactamase (NDM-1). Chem Commun 51:9543–9546
Clinical and Laboratory Standards Institute (2003) Methods for dilution antimicrobial susceptibility tests for bacteria that grow aerobically; Approved standard-sixth edition. CLSI document M7-A6, Wayne, Pa. USA
D’Angelo F, Baldelli V, Halliday N, Pantalone P, Polticelli F, Fiscarelli E, Williams P, Visca P, Leoni L, Rampioni G (2018) Identification of FDA-approved drugs as antivirulence agents targeting the pqs quorum-sensing system of Pseudomonas aeruginosa. Antimicrob Age Chemother 62:e1296–e1318. https://doi.org/10.1128/AAC.01296-18
De Oliveira DMP, Forde BM, Kidd TJ, Harris PNA, Schembri MA, Beatson SA, Paterson DL, Walker MJ (2020) Antimicrobial resistance in ESKAPE pathogens. Clin Microbiol rev 33. https://doi.org/10.1128/CMR.00181-19
Dong C, Zhou J, Wang P, Li T, Zhao Y, Ren X, Lu J, Wang J, Holmgren A (2020) Zou L (2020) Topical therapeutic efficacy of ebselen against multidrug-resistant Staphylococcus aureus LT-1 targeting thioredoxin reductase. Front Microbiol 15(10):3016. https://doi.org/10.3389/fmicb.2019.03016
Foucaud L, Wilson MR, Rown DM, Stone V (2007) Measurement of reactive species production by nanoparticles prepared in biologically relevant media. Toxicol Lett 1:1–9
Founou RC, Founou LL, Essack SY (2017) Clinical and economic impact of antibiotic resistance in develo** countries: a systematic review and meta-analysis. PLoS One 12:e0189621. https://doi.org/10.1371/journal.pone.0189621
Ghodake V, Vishwakarma J, Vavilala SL, Patravale V (2020) Cefoperazone sodium liposomalformulation to mitigate P.aeruginosa biofilm in cystic fibrosis infection: a QbD approach. Int J Pharmaceut 587:119696
Grandclément C, Tannières M, Moréra S, Dessaux Y, Faure D (2016) Quorum quenching: role in nature and applied developments. FEMS Microbiol Rev 40:86–116
Gustafsson TN, Osman H, Werngren J, Hoffner S, Engman L, Holmgren A (2016) Ebselen and analogs as inhibitors of Bacillus anthracis thioredoxin reductase and bactericidal antibacterials targeting Bacillus species, Staphylococcus aureus and Mycobacterium tuberculosis. Biochimica et Biophysica Acta (BBA) - Gen Sub 1860:1265–1271
Jiang Q, Chen J, Yang C, Yin Y, Yao K (2019) Quorum sensing: a prospective therapeutic target for bacterial diseases. Biomed Res Int 2015978. https://doi.org/10.1155/2019/2015978
Khayyat A, Hegazy W, Shaldam MA, Mosbah R, Almalki AJ, Ibrahim TS, Khayat M, Khafagy E, Soliman WE, Abbas HA (2021) Xylitol inhibits growth and blocks virulence in Serratia marcescens. Microorganisms 9. https://doi.org/10.3390/microorganisms9051083
Lewis K (2007) Persister cells, dormancy and infectious disease. Nat Rev Microbiol 5:48–56
LewisOscar F, Nithya C, Bakkiyaraj D, ArunKumar M, Alhari NS (2017) Biofilm inhibitory effect of Spirulina platensis extracts on bacteria of clinical significance. Proc Natl Acad Sci India Sect B Biol Sci 87:537–544
Lu J, Vlamis-Gardikas A, Kandasamy K, Zhao R, Gustafsson TN, Engstrand L, Hoffner S, Engman L, Holmgren A (2013) Inhibition of bacterial thioredoxin reductase: an antibiotic mechanism targeting bacteria lacking glutathione. FASEB J 27:1394–1403
Mellini M, Di Muzio E, D’Angelo F, Baldelli V, Ferrillo S, Visca P, Leoni L, Polticelli F, Miró-Canturri A, Ayerbe-Algaba R, Smani Y (2019) Drug repurposing for the treatment of bacterial and fungal infections. Front Microbiol 10:41. https://doi.org/10.3389/fmicb.2019.00041
Mishra B, Wang G (2017) Individual and combined effects of engineered peptides and antibiotics on Pseudomonas aeruginosa biofilms. Pharmaceuticals 10:E58. https://doi.org/10.3390/ph10030058
Molina-Manso D, del Prado G, Ortiz-Pérez A, Manrubia-Cobo M, Gómez-Barrena E, Cordero-Ampuero J, Esteban J (2013) In vitro susceptibility to antibiotics of staphylococci in biofilms isolated from orthopaedic infections. Int J Antimicr Age 41:521–523
Morones-Ramirez JR, Winkler JA, Spina CS, Collins JJ (2013) Silver enhances antibiotic activity against gram-negative bacteria. Sci Transl Med 5:190ra181
Olsen I (2015) Biofilm-specific antibiotic tolerance and resistance. Eur J Clin Microbiol Infect Dis 34:877–886
O'Neill J (2016) Antimicrobial resistance: Tackling a crisis for the health and wealth of nations. Final report and recommendations. London: HM Government and Wellcome Trust. Review on Antimicrobial Resistance, chaired by Jim O’Neill. http://amr-review.org/Publications.
Patel B, Mishra S, Priyadarsini IK, Vavilala SL (2021) Elucidating the anti-biofilm and anti-quorum sensing potential of selenocystine against respiratory tract infections causing bacteria: in vitro and in silico studies. Biol Chem 402:769–783
Rampioni G, Visca P, Leoni L, Imperi F (2017) Drug repurposing for antivirulence therapy against opportunistic bacterial pathogens. Emer Top Life Sci 1:13–23
Santajit S, Indrawattana N (2016) Mechanisms of antimicrobial resistance in ESKAPE pathogens. BioMed Res Int 2016:2475067
Sethupathy S, Sathiyamoorthi E, Kim Y-G, Lee J-H, Lee J (2020) Antibiofilm and antivirulence properties of indoles against Serratia marcescens. Frnt Microbiol 11:2637. https://doi.org/10.3389/fmicb.2020.584812
Steindl TM, Schuster D, Laggner C, Chuang K, Hoffmann RD, Langer T (2007) Parallel screening and activity profiling with HIV protease inhibitor pharmacophore models. J Chem Inform Model 47:563–571
Streeter K, Katouli M (2016) Pseudomonas aeruginosa: a review of their pathogenesis and prevalence in clinical settings and the environment. Infect Epidemiol Med 2:7. https://doi.org/10.18869/modares.iem.2.1.25
Thangamani S, Younis W, Seleem MN (2015) Repurposing ebselen for treatment of multidrug-resistant staphylococcal infections. Sci Rep 5:11596. https://doi.org/10.1038/srep11596
TrentinDda S, Giordani RB, Zimmer KR, da Silva AG, da Silva MV, Correia MT, Baumvol IJ, Macedo AJ (2011) Potential of medicinal plants from the Brazilian semi-arid region (Caatinga) against Staphylococcus epidermidis planktonic and biofilm lifestyles. J Ethnopharmacol 137:327–335
Vishwakarma J, Vavilala SL (2020) Unraveling the anti-biofilm potential of green algal sulfated polysaccharides against Salmonella enterica and Vibrio harveyi. Appl Microbiol Biotechnol 104:6299–6314
Vishwakarma J, Waghela B, Falcao B, Vavilala SL (2021) Algal polysaccharide’s potential to combat respiratory infections caused by Klebsiella pneumoniae and Serratia marcescens biofilms. Appl Biochem Biotechnol 194:671–693
Wang H, Joseph JA (1999) Quantifying cellular oxidative stress by dichlorofluorescein assay using microplate reader. Free Radic Biol Med 27:612–616
Whitele M, Diggle SP, Greenberg EP (2017) Bacterial quorum sensing: the progress and promise of an emerging research area. Nature 551:313–320
Yanga D, Fana J, Caoa F, Denga Z, Pojmanb JA, Ji L (2019) Immobilization adjusted clock reaction in the urea–urease–H+ reaction system. RSC Adv 9:3514–3519
Zhou J-W, Ruan L-Y, Chen H-J, Luo H-Z, Jiang H, Wang J-S, Jia A-Q (2019) Inhibition of Quorum Sensing and Virulence in Serratia marcescens by Hordenine. J Agri Food Chem 67:784–795
Zou L, Lu J, Wang J, Ren X, Zhang L, Gao Y, Rottenberg ME, Holmgren A (2018) Synergistic antibacterial effect of silver and ebselen against multidrug-resistant gram-negative bacterial infections. EMBO Mol Med 9:1165–1178
Acknowledgements
This work is supported by UM DAE CEBS, Mumbai, India.
Author information
Authors and Affiliations
Contributions
SAS performed and analysed all the invitro experiments; BP performed the in silico experiments; IKP conceived the idea and edited the manuscript; VLS created the concept, idea, design of the research, manuscript writing, and data analysis.
Corresponding authors
Ethics declarations
Ethical approval
Not applicable for this work.
Consent to participate
Not applicable to this work.
Consent for publication
Not applicable to this work.
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Shaikh, S.A., Patel, B., Priyadarsini, I.K. et al. Combating planktonic and biofilm growth of Serratia marcescens by repurposing ebselen. Int Microbiol 26, 693–704 (2023). https://doi.org/10.1007/s10123-022-00301-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10123-022-00301-5