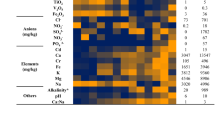
Abstract
Submarine mud volcanoes (MVs) have attracted significant interest in the scientific community for obtaining clues on the subsurface biosphere. On-land MVs, which are much less focused in this context, are equally important, and they may even provide insights also for astrobiology of extraterrestrial mud volcanism. Hereby, we characterized microbial communities of two active methane-see** on-land MVs, Murono and Kamou, in central Japan. 16S rRNA gene profiling of those sites recovered the dominant archaeal sequences affiliated with methanogens. Anaerobic methanotrophs (ANME), with the subgroups ANME-1b and ANME-3, were recovered only from the Murono site albeit a greatly reduced relative abundance in the community compared to those of typical submarine MVs. The bacterial sequences affiliated to Caldatribacteriota JS1 were recovered from both sites; on the other hand, sulfate-reducing bacteria (SRB) of Desulfobulbaceae was recovered only from the Murono site. The major difference of on-land MVs from submarine MVs is that the high concentrations of sulfate are not always introduced to the subsurface from above. In addition, the XRD analysis of Murono shows the absence of sulfate-, sulfur-related mineral. Therefore, we hypothesize one scenario of ANME-1b and ANME-3 thriving at the depth of the Murono site independently from SRB, which is similar to the situations reported in some other methane-see** sites with a sulfate-depleted condition. We note that previous investigations speculate that the erupted materials from Murono and Kamou originate from the Miocene marine strata. The fact that SRB (Desulfobulbaceae) capable of associating with ANME-3 was recovered from the Murono site presents an alternative scenario: the old sea-related juvenile water somehow worked as the source of additional sulfur-related components for the SRB-ANME syntrophic consortium forming at a deeper zone of the site. However, the reason for the differences between Murono and Kamou is still unknown, and this requires further investigation.





Similar content being viewed by others
Data availability
Nucleotide sequence data reported in this paper are available in the DDBJ sequenced Read Archive under the accession numbers DRX221637 to DRX221644 and DRX251432 to DRX251435. Please contact author for any other data requests.
References
Alain K, Holler T, Musat F, Elvert M, Treude T, Krüger M (2006) Microbiological investigation of methane- and hydrocarbon-discharging mud volcanoes in the Carpathian Mountains. Romania Envion Microbiol 8(4):574–590
Aliyev AA, Guliyev IS, Rahmanov RR (2009) Catalogue of mud volcanoes eruptions of Azerbaijan (1810–2007). Nafta-press Baku, Azerbaijan, p 106
Aoyagi K, Kazama T (1980) Transformation changes of clay minerals, zeolite and silica minerals during diagenesis. Sedimentology 27:179–188
Bakken LR, Olsen RA (1989) DNA-content of soil bacteria of different cell size. Soil Biol Biochem 21(6):789–793
Berger S, Welte C, Deppenmeier U (2012) Acetate activation in Methanosaeta thermophila: characterization of the key enzymes pyrophosphatase and acetyl-CoA synthetase, archaea. Hindawi Publishing Corp, Article ID, p 315153
Bhattarai S, Cassarini C, Gonzalez-Gil G, Egger M, Slomp CP, Zhang Y et al (2017) Anaerobic methane-oxidizing microbial community in a coastal marine sediment: anaerobic methanotrophy dominated by ANME-3. Microbiol Ecol 74(3):608–622
Bhattarai S, Cassarini C, Lens PNL (2019) Physiology and distribution of archaeal methanotrophs that couple anaerobic oxidation of methane with sulfate reduction. Microbiol Mol Biol Rev 83:e00074-e118
Bokulich NA, Kaehler BD, Rideout JR, Dillon M, Bolyen E, Knight R et al (2018a) Optimizing taxonomic classification of marker-gene amplicon sequences with QIIME 2’s q2-feature-classifier plugin. Microbiome 6:90
Bokulich NA, Kaehler BD, Rideout JR, Dilon M, Bolyen E, Knight R et al (2018b) Optimizing taxonomic classification of marker-gene amplicon sequences with QIIME 2’s q2-feature-classifier plugin. Microbiome 6:90
Button DK, Robertson BR (2001) Determination of DNA content of aquatic bacteria by flow cytometry. Appl Environ Microbiol 67(4):1636–1645
Caporaso JG, Lauber CL, Walters WA, Berg-Lyons D, Lozupone CA, Turnbaugh PJ et al (2011) Global patterns of 16S rRNA diversity at a depth of millions of sequences per sample. Proc Natl Acad Sci USA 108(Suppl 1):4516–4522
Cheng TW, Chang YH, Tang SL, Tseng CH, Chiang PW, Chang KT et al (2012) Metabolic stratification driven by surface and subsurface interactions in a terrestrial mud volcano. ISME J 6:2280–2290
Etiope G, Nakada R, Tanaka K, Yoshida N (2011) Gas seepage from Tokamachi mud volcanoes, onshore Niigata Basin (Japan): origin, post-genetic alterations and CH4-CO2 fluxes. Appl Geochem 26(3):348–359
Ferrer M, Guazzaroni ME, Richter M, García-Salamanca A, Yarza P, Suárez-Suár A et al (2011) Taxonomic and functional metagenomic profiling of the microbial community in the anoxic sediment of a sub-saline shallow lake (Laguna de Carrizo, Central Spain). Microbial Ecol 62:824–837
Ferry JG and Maupin-Furlow JA (1996) Analysis of genes in the pathway for the fermentation of acetate to methane by Methanosarcina thermophila. Microbial Growth on C1 Compounds, 64–71.
Greene AC, Patel BKC, Yacob S (2009) Geoalkalibacter subterraneus sp. nov., an anaerobic Fe(III)- and Mn(IV)-reducing bacterium from a petroleum reservoir, and emended descriptions of the family Desulfuromonadaceae and the genus Geoalkalibacter. Int J Syst Evol Microbiol 59:781–785
Gründger F, Carrier V, Scenning MM, Panieri G, Vonnahme TR, Klasek S et al (2019) Methane-fuelled biofilms predominantly composed of methanotrophic ANME-1 in Arctic gas hydrate-related sediments. Sci Rep 9:9725
Hosein R, Haque S, Beckles DM (2014) Mud volcanoes of Trinidad as astrobiological analogs for Martian environments. Life 4:566–585
Hoshino T, Toki T, Ijiri A, Morono Y, Machiyama H, Ashi J et al (2017) Atribacteria from the subseafloor sedimentary biosphere disperse to the hydrosphere through submarine mud volcanoes. Fron Microbiol 8:1135
Huang Z, Xu S, Han Y, Gao Y, Wang Y, Song C (2016) Comparative study of Dushanzi and Baiyanggou mud volcano microbial communities in Junggar Basin in **njiang, China. Int Res J Pub Environ Health 3(11):244–256
Iasakov TR, Kanapatskiy TA, Toshchakov SV, Korzhenkov AA, Ulyanova MO, Pimenov NV (2022) The Baltic Sea methane pockmark microbiome: the new insights into the patterns of relative abundance and ANME niche separation. Mar Environ Res 173:105533
Ijiri A, Inagaki F, Kubo Y, Adhikari RR, Hattori S, Hoshino T et al (2018) Deep-biosphere methane production stimulated by geofluids in the Nankai accretionary complex. Sci Adv 4(6):eaao4631
Kakizaki Y, Snyder G, Tanahashi M, Ishida N, Matsumoto R (2018) Concentration and carbon-isotopic change of dissolved gas from Murono mud volcano in Tokamachi City, Niigata Prefecture (central Japan), just before and after the 2014 Kamishiro Fault Earthquake. Jour Geol Soc Japan 124(2):127–140
Kashefi K, Holmes DE, Baross JA, Lovely DR (2003) Thermophily in the Geobacteraceae: Geothermobacter ehrlichii gen. nov., sp. nov., a novel thermophilic member of the Geobacteraceae from the “Bag City” hydrothermal vent. Appl Environ Microbiol 69:2985–2993
Kitamura K, Fujita T, Akada S, Tonouchi A (2011) Methanobacterium kanagiense sp. nov., a hydrogenotrophic methanogen, isolated from rice-field soil. Int J Syst Evol Microbiol 61:1246–1252
Knittel K, Lösekann T, Boetius A, Kort R, Amann R (2005) Diversity and distribution of methanotrophic archaea at cold seeps. Appl Environ Microbiol 71:467–479
Komatsu G, Ori GG (2000) Exobiological implications of potential sedimentary deposits on Mars. Planet Space Sci 48(11):1043–1052
Komatsu G, Ori GG, Cardinale M, Dohm JM, Baker VR, Vaz DA et al (2011) Roles of methane and carbon dioxide in geological processes on Mars. Planet Space Sci 59:169–181
Komatsu G, Okubo CH, Wray JJ, Ojha L, Cardinale M, Murana A et al (2016) Small edifice features in Chryse Planitia, Mars: assessment of a mud volcano hypothesis. Icarus 268:56–75
Komatsu G, Ishimaru R, Miyake N, Ohno S, and Matsui T (2014) Astrobiological potential of mud volcanism on Mars, LPSC XXXXV, Abstract #1085
Kopf AJ (2002) Significance of mud volcanism. Rev Geophys 40(2):1005
Lazer CS, Parkes RJ, Cragg BA, L’Haridon S, Toffin L (2011) Methanogenic diversity and activity in hypersaline sediments of the centre of the Napoli mud volcano. Eastern Mediterranean Sea Environ Microbiol 13(8):2078–2091
Lee DH, Kim JH, Lee YM, Stadnitskaia A, ** YK, Niemann H et al (2018a) Biogeochemical evidence of anaerobic methane oxidation on active submarine mud volcanoes on the continental slope of the Canadian Beaufort Sea. Biogeosciences 15:7419–7433
Lee YM, Hwang K, Lee JI, Kim M, Hwang CY, Noh SC et al (2018b) Genomic insight into the predominance of candidate phylum Atribacteria JS1 lineage in marine sediments. Front Microbiol 9:2909
Li H, Yang Q, Zhou H (2020) Niche differentiation of sulfate- and iron-dependent anaerobic methane oxidation and methylotrophic methanogenesis in deep sea methane seeps. Front Microbiol 6:365
Lin YT, Tu TH, Wei CL, Rumble D, Lin LH, Wang PL (2018) Steep redox gradient and biogeochemical cycling driven by deeply sourced fluids and gases in a terrestrial mud volcano. FEMS Microbiol Ecol 94(11):fiy171. https://doi.org/10.1093/femsec/fiy171
Lloyd KG, Lapham L, Teske A (2006) An anaerobic methane-oxidizing community of ANME-1b archaea in hypersaline Gulf of Mexico sediments. Appl Environ Microbiol 72(11):7218–7230
Lösekann T, Knittel K, Nadalig T, Bernhard F, Niemann H, Boetius A et al (2007) Diverisity and abundance of aerobic and anaerobic methane oxidizers at the Haakon Mosby Mud Volcano, Barents Sea. Appl Environ Microbiol 73:3348–3362
Ma K, Ma A, Zheng G, Ren G, **e F, Zhou H et al (2021) Mineralosphere microbiome leading to changed geochemical properties of sedimentary rocks from Aiqigou Mud Volcano. Northwest China Microorganisms 9:560
Mardanov AV, Kadnikov VV, Beletsky AV, Ravin NV (2020) Sulfur and methane-oxidizing microbial community in a terrestrial mud volcano revealed by metagenomics. Microorganisms 8:1333
Mazzini A and Etiope G (2017) Mud volcanism: an updated review. Earth-Science Reviews, 168.
Merkel AY, Chernyh NA, Pimernov NV, Bonch-Osmolovskaya EA, Slobodkin AI (2021) Diversity and metabolic potential of the terrestrial mud volcano microbial community with a high abundance of archaea mediating the anaerobic oxidation of methane. Life 11:953
Michaelis W, Seifert R, Nauhaus K, Treude T, Thiel V, Blumenberg M et al (2002) Microbial reefs in the Black Sea fueled by anaerobic oxidation of methane. Science 297:1013–1015
Muyzer G, De Waal EC, Uitterlinden AG (1993) Profiling of complex microbial populations by denaturing gradient gel electrophoresis analysis of polymerase chain reaction-amplified genes coding 16S rRNA. Appl Environ Microbiol 59:695–700
Nobu MK, Dodsworth JA, Murugapiran SK, Rinke C, Gies EA, Webster G et al (2016) Phylogeny and physiology of candidate phylum ‘Atribacteria’ (OP9/JS1) inferred from cultivation-independent genomics. ISME J 10:273–286
Oehler DZ, Allen CC (2010) Evidence for pervasive mud volcanism in Acidalia Planitia, Mars. Icarus 208:636–657
Orphan VJ, Hinrichs KU, Ussler W III, Paull CK, Taylor LT, Sylva SP et al (2001) Comparative analysis of methane-oxidizing archaea and sulfate-reducing bacteria in anoxic marine sediments. Appl Environ Microbiol 67(4):1922–1934
Pachiadaki MG, Kallionaki A, Dählmann A, De Lange GJ, Kormas KA (2011) Diversity and spatial distribution of prokaryotic communities along a sediment vertical profile of a deep-sea mud volcano. Microbiol Ecol 62(3):655–668
Probst AJ, Moissl-Eichinger C (2015) “Altiarchaeales”: uncultivated archaea from the subsurface. Life Reviews 5:1381–1395
Raskin L, Stromley JM, Rittmann BE, Stahl DA (1994) Group-specific 16S rRNA hybridization probes to describe natural communities of methanogens. Appl Environ Microbiol 60(4):1232–1240
Robeson II MS, O’Rourke DR, Kaehler BD, Ziemski M, Dillon MR, Foster JT et al (2020) RESCRIPt: reproducible sequence taxonomy reference database management for the masses. bioRxiv 2020.10.05.326504.
Sakai S, Ehara M, Tseng IC, Yamaguchi T, Bräuer SL, Cadillo-Quiroz H et al (2012) Methanolinea mesophiila sp. no., a hydrogenotrophic methanogen isolated from rice field soil, and proposal of the archaeal family Methanoregulaceae fam. nov within the order Methanomicrobiales. Int J Syst Evol Microbiol 62(Pt 6):1389–1395
Saxton MA, Samarkin VA, Schutte CA, Bowles MW, Madigan MT, Cadieux SB et al (2016) Biogeochemical and 16S rRNA gene sequence evidence supports a novel mode of anaerobic methanotrophy in permanently ice-covered Lake Fryxell, Antarctica. Limnol Oceanogr 61:119–130
Shinya T, Tanaka K (2005) Mud volcanoes in Matsudai, Tokamachi City, Niigata Prefecture. J JSNDS 24–1:49–58
Shinya T, Tanaka K (2009) Origin of materials erupting from mud volcano in Tokamachi City, Niigata Prefecture. Central Japan J Geography 118(3):340–349
Sprenger WW, van Belzen MC, Rosenberg J, Hackstein JHP, Keltjens JT (2000) Methanomicrococcus blastticola gen. nov., sp. nov., a methanol- and methylamine-reducing methanogen from the hindgut of the cockroach Periplaneta americana. Int J Syst Evol Microbiol 50:1989–1999
Stoeck T, Epstein S (2003) Novel eukaryotic lineages inferred from small-subunit rRNA analyses of oxygen-depleted marine environments. Appl Environ Microbiol 69(5):2657–2663
Takahashi S, Tomita J, Nishioka K, Hisada T, Nishijima M (2014) Development of a prokaryotic universal primer for simultaneous analysis of Bacteria and Archaea using next-generation sequencing. PLoS ONE 9:e105592
Takai K, Horikoshi K (2000) Rapid detection and quantification of members of the archaeal community by quantitative PCR using fluorogenic probes. Appl Environ Microbiol 66(11):5066–5072
Taylor J, Parkes RJ (1983) The cellular fatty acids of the sulphate-reducing bacteria, Desulfobacter sp., Desulfobulbus sp. and Desulfovibrio desulfuricans. J Gen Microbiol 129:3303–3309
Tu TH, Wu LW, Lin YS, Imachi H, Lin LH, Wang PL (2017) Microbial community composition and functional capacity in a terrestrial ferruginous, sulfate-depleted mud volcano. Front Microbiol 8:2137
Widdel F, Pfennig N (1982) Studies on dissimilatory sulfate-reducing bacteria that decompose fatty acids II. Incomplete oxidation of propionate by Desulfobulbus propionicus gen. nov., sp. nov. Arch Microbiol 131:360–365
Wrede C, Brady S, Rockstroh S, Dreier A, Kokoschka S, Heinzelmann SM et al (2012) Aerobic and anaerobic methane oxidation in terrestrial mud volcanoes in the Northern Apennines. Sediment Geol 263–264:210–219
Wunder LC, Aromokeye DA, Yin X, Richter-Heitmann T, Willis-Poratti G, Schnakenberg A et al (2021) Iron and sulfate reduction structure microbial communities in (sub-) Antarctic sediments. ISME J 15:3587–3604
Yashiro Y, Sakai S, Ehara M, Miyazaki M, Yamaguchi T, Imachi H (2011) Methanoregula formicica sp. nov., a methane-producing archaeon isolated from methanogenic sludge. Int J Syst Evol Microbiol 61(1):53–59
Yakimov MM, Giuliano L, Cappello S, Denaro R, Golyshin PN (2007) Microbial community of a hydrothermal mud vent underneath the deep-sea anoxic brine lake Urania (Eastern Mediterranean). Ori Life Evol Biosph 37(2):177–188
Yanagawa K, Sunamura M, Lever MA, Morono Y, Hiruta A, Ishizaki O et al (2011) Niche separation of methanotrophic archaea (ANME-1 and -2) in methane-seep sediments of the Eastern Japan Sea Offshore Joetsu. Geomicrobiol J 28(2):118–129
Yanagawa K, Breuker A, Schippers A, Nishizawa M, Ijiri A, Hirai M et al (2014) Microbial community stratification controlled by the subseafloor fluid flow and geothermal gradient at the Iheya North hydrothermal field in the Mid-Okinawa Trough (Integrated Ocean Drilling Program Expedition 331). Appl Environ Microbiol 80(19):6126–3615
Acknowledgements
We thank two anonymous reviewers for their constructive comments that helped us in improving the manuscript. The fieldwork was supported by a project focusing on geology and astrobiology of mud volcanoes (Planetary Exploration Research Center, Chiba Institute of Technology). We would like to express our most gratitude to the Waseda University Automobile Club for granting us access to their training circuit for our fieldwork. Makito Kobayashi (University of Tokyo) and Dr. Hiroshi Sakuma (National Institute for Materials Science) contributed to the mineral analysis of this project. Finally, we would like to thank Dr. Shingo Kato (Japan Collection of Microorganisms, RIKEN BioResource Research Center) for his continuous support in this project.
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Material preparation, data collection, and analysis were performed by Nori Miyake, Ryo Ishimaru, and Goro Komatsu. The first draft of the manuscript was written by Nori Miyake and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Competing interests
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Miyake, N., Ishimaru, R., Komatsu, G. et al. Characterization of archaeal and bacterial communities thriving in methane-see** on-land mud volcanoes, Niigata, Japan. Int Microbiol 26, 191–204 (2023). https://doi.org/10.1007/s10123-022-00288-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10123-022-00288-z